Scalable Synthesis of Alpha-Isopropyl-3,4-Dimethoxyphenylacetonitrile for Commercial Verapamil Production
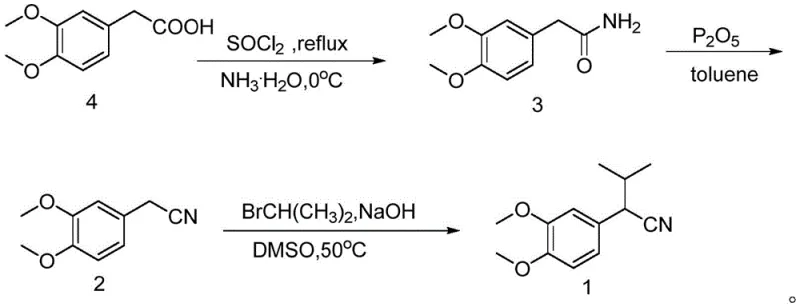
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for critical cardiovascular drug intermediates, particularly for the production of Verapamil, a widely prescribed calcium channel blocker. Patent CN113444020B, published in March 2023, introduces a refined and highly effective methodology for synthesizing alpha-isopropyl-3,4-dimethoxyphenylacetonitrile, a pivotal building block in this therapeutic class. This technical disclosure addresses long-standing challenges in the field, specifically targeting the issues of low overall yields, the use of hazardous reagents, and complex purification processes that have historically plagued the manufacturing of this specific nitrile derivative. By leveraging a strategic three-step sequence starting from the readily available 3,4-dimethoxyphenylacetic acid, the inventors have established a pathway that not only enhances chemical efficiency but also aligns with modern green chemistry principles required by top-tier regulatory bodies.
For R&D directors and process chemists evaluating supply chain partners, the significance of this patent lies in its ability to deliver high-purity intermediates with a drastically simplified impurity profile. The transition from traditional methods to this novel approach eliminates several bottleneck reactions that typically generate difficult-to-remove byproducts. Furthermore, the reliance on stable, commercially available starting materials ensures that the supply chain remains resilient against market fluctuations. As we delve deeper into the mechanistic and operational details of this synthesis, it becomes evident that this technology represents a substantial leap forward in the cost-effective and safe manufacturing of high-value pharmaceutical intermediates, offering a compelling value proposition for procurement teams focused on long-term stability and margin optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-isopropyl-3,4-dimethoxyphenylacetonitrile has been fraught with significant technical and safety hurdles that impact both cost and scalability. Traditional routes often rely on the direct alkylation of phenylacetonitrile derivatives using strong, pyrophoric bases such as sodium hydride (NaH) in solvents like tetrahydrofuran (THF). As highlighted in the comparative data within the patent, these conventional methods suffer from inherently low yields, often hovering around 65.9% for the critical alkylation step, due to competing side reactions and the difficulty in controlling the exothermic nature of the deprotonation. Additionally, the use of NaH presents severe safety risks on an industrial scale, requiring specialized equipment and rigorous safety protocols that drive up operational expenditures. Another common legacy approach involves the dehydration of amides using thionyl chloride, which, while effective in small-scale laboratory settings, frequently results in poor conversion rates (approximately 68.5% yield) and generates corrosive gaseous byproducts that complicate waste management and environmental compliance.
The Novel Approach
In stark contrast, the methodology outlined in CN113444020B offers a transformative solution by replacing these hazardous and inefficient steps with milder, higher-yielding alternatives. The innovation centers on a specific dehydration protocol using phosphorus pentoxide (P2O5) in toluene, which boosts the yield of the nitrile formation step to an impressive 91.3-92.2%, effectively eliminating the bottlenecks associated with thionyl chloride. Furthermore, the final alkylation step utilizes a phase-transfer catalysis system with sodium hydroxide in dimethyl sulfoxide (DMSO) at a moderate temperature of 50°C. This shift not only elevates the yield to nearly 89% but also removes the need for dangerous pyrophoric reagents, thereby simplifying the reactor requirements and significantly enhancing workplace safety. This holistic redesign of the synthetic route ensures a more predictable and economical manufacturing process, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships.
Mechanistic Insights into Phosphorus Pentoxide Dehydration and Phase Transfer Alkylation
The core chemical innovation in this patent lies in the precise manipulation of reaction conditions to favor the desired product while suppressing side pathways. In the second step, the conversion of 2-(3,4-dimethoxyphenyl)acetamide to the corresponding nitrile is driven by the potent dehydrating capability of phosphorus pentoxide. Unlike thionyl chloride, which can lead to chlorination side products or incomplete conversion due to equilibrium issues, P2O5 acts as an irreversible water scavenger in the toluene reflux system. This drives the equilibrium strongly towards the nitrile product, ensuring high conversion rates. The mechanism involves the activation of the amide oxygen by the phosphorus species, facilitating the elimination of water to form the carbon-nitrogen triple bond. This step is crucial for maintaining the integrity of the electron-rich dimethoxy benzene ring, which can be sensitive to harsh acidic conditions found in other dehydration protocols.
Following the formation of the nitrile, the final alkylation step employs a sophisticated phase-transfer catalysis (PTC) mechanism using triethylbenzylammonium chloride (TEBA). In this system, the hydroxide ion from the aqueous sodium hydroxide phase is transported into the organic DMSO phase by the quaternary ammonium cation. This creates a highly reactive, naked anion of the phenylacetonitrile intermediate without the need for extreme bases like NaH. The nucleophilic attack on 2-bromopropane then proceeds efficiently at 50°C. This mechanistic approach minimizes the formation of elimination byproducts (such as propene) and dialkylated impurities, which are common in less controlled alkylation reactions. The result is a cleaner reaction mixture that requires less intensive purification, directly translating to lower solvent consumption and higher throughput in a commercial setting.
How to Synthesize Alpha-Isopropyl-3,4-Dimethoxyphenylacetonitrile Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent data. The process begins with the activation of 3,4-dimethoxyphenylacetic acid using thionyl chloride, followed by immediate ammoniation to secure the amide intermediate with yields exceeding 93%. The subsequent dehydration must be conducted under reflux in toluene with a molar excess of P2O5 to ensure complete water removal. Finally, the alkylation is performed in DMSO with 50% sodium hydroxide and a catalytic amount of TEBA.
- Convert 3,4-dimethoxyphenylacetic acid to 2-(3,4-dimethoxyphenyl)acetamide using thionyl chloride followed by ammoniation.
- Dehydrate the acetamide intermediate to 3,4-dimethoxyphenylacetonitrile using phosphorus pentoxide in toluene.
- Perform alkylation with 2-bromopropane in DMSO using sodium hydroxide and a phase transfer catalyst to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of pyrophoric reagents like sodium hydride removes a major safety liability and reduces the need for specialized storage and handling infrastructure, leading to substantial cost savings in facility maintenance and insurance. Moreover, the use of common industrial solvents such as toluene and DMSO, rather than expensive or highly regulated ethers, simplifies the sourcing of raw materials and ensures consistent availability even during global supply chain disruptions. The higher yields achieved in every step mean that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS) and reducing the volume of chemical waste that needs to be treated and disposed of.
- Cost Reduction in Manufacturing: The shift to a high-yield dehydration process using P2O5 eliminates the losses associated with traditional thionyl chloride methods, where yields often drop below 70%. By improving the yield of this step to over 90%, the overall material efficiency of the plant is significantly enhanced. Additionally, the replacement of expensive and hazardous bases with inexpensive sodium hydroxide in the alkylation step drastically reduces reagent costs. These cumulative efficiencies allow for a more competitive pricing structure for the final API intermediate without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The reliance on 3,4-dimethoxyphenylacetic acid as a starting material leverages a commodity chemical that is widely produced and easily sourced from multiple vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions, particularly the tolerance of the PTC alkylation system to minor variations in temperature and mixing, ensures consistent batch-to-batch quality. This reliability is critical for maintaining uninterrupted production schedules for downstream API manufacturers, preventing costly delays in the formulation of life-saving cardiovascular medications like Verapamil.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the thermal runaway risks associated with NaH additions. The simplified workup procedures, which involve standard extraction and crystallization techniques, reduce the time required for batch turnover. Furthermore, the avoidance of highly toxic cyanide salts (often used in alternative nitrile syntheses) and the reduction of hazardous waste streams align with increasingly stringent environmental regulations. This facilitates smoother regulatory approvals and minimizes the environmental footprint of the manufacturing site, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This route avoids highly toxic reagents and harsh conditions found in prior art. It utilizes a P2O5 dehydration step which achieves significantly higher yields (over 90%) compared to traditional thionyl chloride dehydration methods, and employs a safer phase-transfer alkylation system instead of pyrophoric sodium hydride.
Q: What is the overall yield potential for this pharmaceutical intermediate?
A: The patent data demonstrates individual step yields of approximately 93-95% for acylation, 91-92% for dehydration, and 88-89% for alkylation. This cumulative efficiency represents a substantial improvement over older methods which often suffered from yields below 70% in critical steps.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses common industrial solvents like toluene and DMSO and avoids extremely hazardous reagents like sodium hydride on a large scale. The mild reaction conditions (50°C for alkylation) and simple workup procedures facilitate safe scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Isopropyl-3,4-Dimethoxyphenylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team has thoroughly analyzed the synthetic route described in CN113444020B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this key Verapamil intermediate with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for GMP API synthesis. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions outlined in this patent, ensuring a seamless transition from development to full-scale manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that details how implementing this high-yield process can improve your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the efficiency and reliability of your supply chain for cardiovascular therapeutics.
