Industrial Scale Production of 2-Sulfonylpyridine Derivatives for Pharmaceutical Applications
Introduction to Advanced Pyridine Skeleton Synthesis
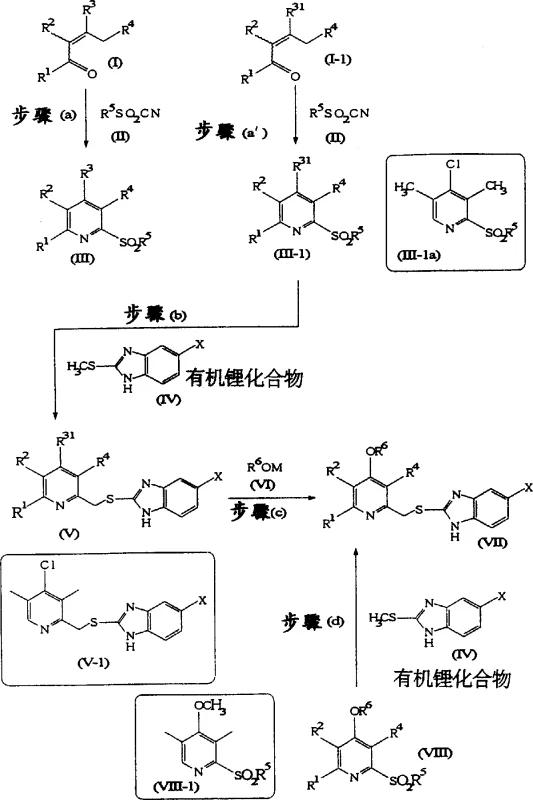
The pharmaceutical and agrochemical industries rely heavily on the efficient production of heterocyclic compounds, particularly those featuring pyridine skeletons which serve as critical backbones for numerous bioactive molecules. Patent CN1233613A introduces a groundbreaking methodology for the industrial production of 2-sulfonylpyridine derivatives and their subsequent conversion into 2-{[(2-pyridyl)methyl]thio}-1H-benzimidazole derivatives. This technology addresses long-standing challenges in synthetic chemistry by replacing unstable starting materials with robust sulfonyl cyanides, thereby enabling high-yield production under generalized conditions. For R&D directors and procurement specialists, this represents a significant shift towards more reliable and cost-effective manufacturing pathways for key intermediates used in drugs such as omeprazole and various agrochemical agents. The ability to synthesize these complex structures in fewer steps not only enhances purity profiles but also drastically simplifies the supply chain logistics required for global distribution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-sulfonylpyridine derivatives has been plagued by significant operational hurdles that hinder industrial scalability and economic efficiency. Traditional methods, such as those described in International Patent Application Publication No. WO96/26188, often necessitate the use of diolefins which are notoriously unstable when exposed to heat, posing serious safety risks and storage difficulties for large-scale facilities. Furthermore, alternative routes involving the reaction of 2-halopyridines with thiolate salts followed by gradual oxidation suffer from low overall yields due to the requirement of multiple discrete reaction steps. These multi-step processes inherently increase the accumulation of impurities and demand extensive purification protocols, leading to substantial material loss and elevated production costs. Additionally, the reliance on 2-halogenated methyl pyridine derivatives as starting materials introduces further complications, as these compounds have short storage periods and must be consumed immediately after synthesis, creating bottlenecks in continuous manufacturing workflows.
The Novel Approach
The innovative strategy outlined in the patent data revolutionizes this landscape by utilizing the reaction of sulfonyl cyanides with α,β-unsaturated carbonyl compounds to effectively produce 2-sulfonylpyridine derivatives. This approach eliminates the need for thermally unstable diolefins, substituting them with chemically robust precursors that can be handled safely under standard industrial conditions. As illustrated in the comprehensive reaction scheme below, the process flows seamlessly from the initial cyclization to the final coupling stage, demonstrating a streamlined pathway that minimizes waste and maximizes throughput. By integrating an organolithium-mediated coupling step, the method achieves the formation of the 2-{[(2-pyridyl)methyl]thio}-1H-benzimidazole skeleton in a single high-yield operation. This consolidation of synthetic steps not only accelerates the production timeline but also ensures a cleaner impurity profile, which is paramount for meeting the stringent quality standards required for active pharmaceutical ingredients.
Mechanistic Insights into Organolithium-Mediated Coupling
The core of this technological advancement lies in the precise execution of the organolithium-mediated coupling reaction, which serves as the pivotal transformation linking the pyridine and benzimidazole moieties. In the presence of organolithium compounds such as n-butyllithium or s-butyllithium, the 2-methylthio-1H-benzimidazole derivatives undergo deprotonation to generate a highly reactive nucleophilic species. This species then attacks the 4-halo-2-sulfonylpyridine derivative, specifically targeting the electrophilic center to displace the sulfonyl group or facilitate the formation of the methylene bridge depending on the specific substitution pattern. The reaction is typically conducted in aprotic solvents like tetrahydrofuran at controlled low temperatures ranging from -50°C to 0°C to prevent side reactions and ensure regioselectivity. The inclusion of tertiary amines such as 1,4-diazabicyclo[2.2.2]octane can further promote the reaction under milder conditions, enhancing the overall efficiency and reproducibility of the process. This mechanistic precision allows for the tolerance of various functional groups, enabling the synthesis of a diverse library of derivatives tailored for specific biological activities.
Impurity control is rigorously maintained throughout the synthesis through the careful selection of reaction parameters and purification techniques. The initial cyclization step generates water as a byproduct, which is effectively removed via azeotropic distillation using solvents like toluene or through the use of molecular sieves, preventing hydrolysis of sensitive intermediates. In the subsequent coupling stages, the use of high-purity organolithium reagents and strictly anhydrous conditions minimizes the formation of desulfonylated byproducts or homocoupling artifacts. The final products, such as the 2-{[(4-chloro-2-pyridyl)methyl]thio}-1H-benzimidazole derivatives shown below, can be isolated through standard extraction and crystallization procedures, yielding materials with purity levels exceeding 98%. This high degree of chemical fidelity is essential for downstream applications where trace impurities could compromise the efficacy or safety of the final drug product.
![Chemical structure of 2-{[(4-halo-2-pyridyl)methyl]thio}-1H-benzimidazole derivatives (Formula V)](/insights/img/2-sulfonylpyridine-synthesis-pharma-intermediate-supplier-20260309005153-05.png)
How to Synthesize 2-Sulfonylpyridine Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression that balances chemical reactivity with operational simplicity, making it ideal for transfer from laboratory bench to commercial plant. The process begins with the condensation of readily available α,β-unsaturated carbonyl compounds with sulfonyl cyanides in the presence of a catalyst such as lithium perchlorate or tributyl borate. Following the isolation of the 2-sulfonylpyridine core, the material is subjected to lithiation conditions to facilitate coupling with benzimidazole precursors. Detailed standardized operating procedures regarding stoichiometry, temperature ramps, and quenching protocols are critical for maintaining batch-to-batch consistency. The following guide outlines the fundamental steps required to execute this synthesis effectively, ensuring that technical teams can replicate the high yields reported in the patent literature while adhering to safety and quality guidelines.
- React sulfonyl cyanide with alpha,beta-unsaturated carbonyl compounds in the presence of a solvent and catalyst to form 2-sulfonylpyridine derivatives.
- Perform organolithium-mediated coupling between the 2-sulfonylpyridine derivative and 2-methylthio-1H-benzimidazole to form the thioether skeleton.
- Optionally substitute halogen groups with alkoxyl groups using metal alkoxides to finalize the benzimidazole derivative structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. By shifting away from unstable and hazardous raw materials like diolefins, manufacturers can significantly reduce the costs associated with specialized storage and handling infrastructure. The streamlined nature of the synthesis, which combines multiple transformations into fewer operational units, translates to a drastic reduction in processing time and energy consumption. Furthermore, the use of common industrial solvents such as toluene and ethers ensures that the supply chain remains resilient against fluctuations in specialty chemical availability. This robustness allows for more predictable lead times and enhances the overall reliability of supply for downstream API manufacturers who depend on consistent intermediate delivery.
- Cost Reduction in Manufacturing: The elimination of multi-step oxidation sequences and the use of stable starting materials fundamentally lower the cost of goods sold. By avoiding the need for expensive transition metal catalysts or complex purification trains required by older methods, the process achieves substantial cost savings. The high yields reported, such as the 89% yield in Example 1, indicate minimal material waste, which further optimizes the economic efficiency of the production line. Additionally, the ability to recycle solvents and recover byproducts contributes to a leaner manufacturing model that maximizes return on investment for capital equipment.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents like sulfonyl cyanides and crotonaldehyde mitigates the risk of supply disruptions. Unlike methods requiring freshly prepared halomethyl pyridines with short shelf lives, this process utilizes intermediates that can be stockpiled safely, providing a buffer against market volatility. This stability ensures that production schedules can be maintained without interruption, securing the continuity of supply for critical pharmaceutical programs. The simplified logistics also reduce the administrative burden on procurement teams, allowing them to focus on strategic sourcing rather than crisis management.
- Scalability and Environmental Compliance: The reaction conditions, typically involving reflux temperatures between 80°C and 120°C, are well within the capabilities of standard glass-lined or stainless steel reactors found in most chemical plants. This ease of scale-up means that production can be ramped from pilot batches to multi-ton campaigns without significant re-engineering. Moreover, the reduction in step count inherently lowers the volume of chemical waste generated, aligning with modern environmental sustainability goals. The process avoids the generation of heavy metal waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent regulatory frameworks regarding industrial discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers highlight the versatility of the method across different substitution patterns and its compatibility with existing manufacturing infrastructure.
Q: What are the advantages of using sulfonyl cyanides over diolefins?
A: Sulfonyl cyanides offer superior thermal stability compared to unstable diolefins used in conventional methods, significantly reducing safety risks and storage costs during industrial production.
Q: How does the organolithium step improve yield?
A: The direct organolithium-mediated coupling allows for a one-step formation of the 2-{[(2-pyridyl)methyl]thio}-1H-benzimidazole skeleton, bypassing multiple oxidation steps and minimizing byproduct formation.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process utilizes standard reflux conditions (80°C to 120°C) and common solvents like toluene, making it highly adaptable for large-scale commercial production from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Sulfonylpyridine Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring this advanced synthesis technology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-sulfonylpyridine derivatives meets the highest international standards. Our commitment to quality assurance means that you can rely on us as a partner who understands the critical nature of API intermediates in the global pharmaceutical supply chain.
We invite you to engage with our technical procurement team to discuss how this novel methodology can be integrated into your supply strategy. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our capabilities align with your development timelines and quality objectives. Together, we can optimize the production of these vital chemical building blocks.
