Advanced Racemization Technology for High-Purity Agrochemical Intermediates and Commercial Scale-Up
Advanced Racemization Technology for High-Purity Agrochemical Intermediates and Commercial Scale-Up
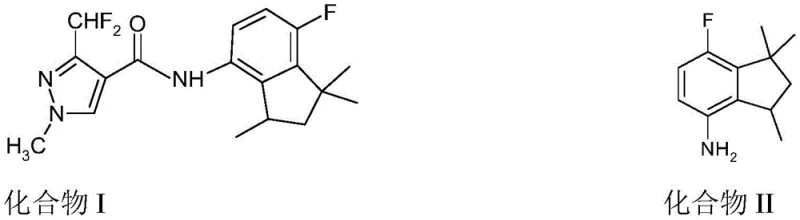
The pharmaceutical and agrochemical industries constantly face the challenge of maximizing atom economy and minimizing waste, particularly when dealing with chiral molecules where only one enantiomer possesses the desired biological activity. Patent CN108777958B, published in March 2021, introduces a groundbreaking methodology specifically designed to address the inefficiency associated with the production of 7-fluoro-1,1,3-trimethyl-2,3-dihydro-1H-inden-4-amine, a critical intermediate for potent fungicidal compounds. This patent details a robust five-step chemical sequence that successfully converts the biologically inactive S-enantiomer of this aminoindan derivative back into its useful racemic form. By implementing this recycling strategy, manufacturers can effectively reclaim what was previously considered waste material, thereby enhancing the overall sustainability and economic viability of the supply chain for high-value agricultural chemicals.
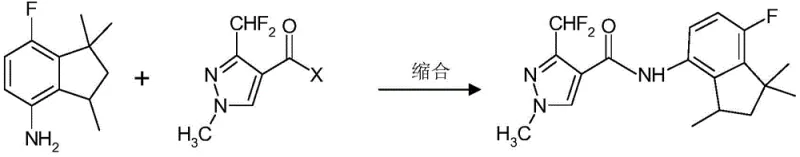
In the context of modern fungicide development, the target molecule, often referred to as Compound I in related literature, relies heavily on the stereochemical purity of its indane scaffold. The traditional synthesis often yields a mixture or requires resolution, leaving the undesired enantiomer as a byproduct with limited utility. The innovation presented in this patent offers a systematic solution to this bottleneck. It provides a reliable agrochemical intermediate supplier with the technical blueprint to transform the unwanted S-isomer into a racemate, which can then be re-separated to isolate the active R-enantiomer. This approach not only aligns with green chemistry principles by reducing waste but also offers substantial potential for cost optimization in the manufacturing of next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral agrochemical intermediates like 7-fluoro-1,1,3-trimethyl-2,3-dihydro-1H-inden-4-amine has been plagued by inherent inefficiencies in stereoselective synthesis or resolution processes. In many conventional pathways, the synthesis results in a racemic mixture, necessitating a resolution step to isolate the biologically active R-enantiomer. The remaining S-enantiomer, which exhibits little to no fungicidal activity, is typically discarded or sold at a fraction of the cost for non-critical applications. This represents a significant loss of valuable raw materials and processing energy, effectively capping the maximum theoretical yield of the active ingredient at 50% unless asymmetric synthesis is employed, which often involves expensive chiral catalysts and complex reaction conditions. Furthermore, the disposal of large quantities of chiral waste imposes additional environmental burdens and regulatory compliance costs on manufacturing facilities.
The Novel Approach
The methodology disclosed in CN108777958B fundamentally shifts this paradigm by introducing a practical racemization cycle. Instead of discarding the S-enantiomer, the process subjects it to a specific sequence of chemical transformations that erase its stereochemical information, regenerating the racemic mixture. This allows the recovered material to be fed back into the resolution or separation loop, theoretically enabling nearly 100% conversion of the starting material into the desired active R-enantiomer over multiple cycles. This novel approach eliminates the need for discarding half of the production output, thereby drastically improving the mass balance of the entire synthesis. For procurement managers and supply chain heads, this translates to a more secure and efficient supply of high-purity agrochemical intermediates, as the effective yield per batch of starting material is significantly enhanced without requiring proportional increases in raw material procurement.
Mechanistic Insights into the Five-Step Racemization Cycle
The core of this invention lies in a cleverly designed five-step reaction sequence that temporarily modifies the molecular structure to allow for the loss of chirality before restoring the original scaffold. The process begins with the acylation of the S-enantiomer amine, protecting the nitrogen atom and modifying the electronic properties of the ring system to facilitate subsequent oxidation. This is followed by an oxidation step, typically using potassium permanganate, which introduces a hydroxyl group at the 3-position of the indane ring. This hydroxylation is crucial as it sets up the molecule for the pivotal dehydration step. By removing water under acidic conditions, a double bond is formed within the five-membered ring, creating an indene derivative. It is at this stage that the chiral center at the 3-position is eliminated, as the sp3 hybridized carbon becomes sp2 hybridized, resulting in a planar, achiral structure.
Once the chirality is erased, the molecule undergoes deacylation to remove the protecting group, yielding a free amine with a double bond in the ring. The final step involves catalytic hydrogenation, where hydrogen gas is added across the double bond in the presence of a metal catalyst such as palladium on carbon. Since the addition of hydrogen can occur from either face of the planar indene ring with roughly equal probability, the result is a statistical mixture of both R and S configurations, effectively regenerating the racemate. This mechanistic pathway is robust and utilizes standard organic transformations, making it highly reproducible. The ability to toggle between chiral and achiral states through oxidation and reduction provides a powerful tool for managing stereochemistry in complex molecule synthesis, ensuring that no valuable enantiomer is permanently lost to the waste stream.
How to Synthesize 7-Fluoro-1,1,3-trimethyl-2,3-dihydro-1H-inden-4-amine Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for executing this racemization on a laboratory or pilot scale. The process is designed to be operationally simple, relying on common reagents and standard workup procedures such as extraction, filtration, and crystallization. The initial acylation is performed at elevated temperatures to ensure complete conversion, while the oxidation step is carefully controlled at room temperature to prevent over-oxidation. The dehydration and deacylation steps utilize strong mineral acids, which are readily available and cost-effective. Finally, the hydrogenation step employs standard heterogeneous catalysis, allowing for easy removal of the catalyst by filtration. For detailed operational parameters, stoichiometry, and specific purification techniques required to achieve optimal yields and purity, please refer to the standardized synthesis guide below.
- Acylation of the S-enantiomer amine using acetic anhydride to form the corresponding indanamide derivative.
- Oxidation of the indanamide derivative using potassium permanganate to introduce a hydroxyl group at the 3-position.
- Acid-catalyzed dehydration of the hydroxyindanamide to form an indeneamide derivative, eliminating the chiral center.
- Deacylation of the indeneamide using strong acid followed by basification to yield the free amine.
- Catalytic hydrogenation of the indeneamine derivative using Pd-C to restore the saturated indane ring as a racemate.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the supply chain of complex agrochemical intermediates, the implementation of this racemization technology offers profound strategic advantages beyond simple chemical yield. By integrating this recycling loop, companies can significantly reduce their dependency on fresh raw material inputs, as the ineffective S-enantiomer is continuously repurposed. This leads to a drastic simplification of the material flow and a reduction in the total volume of waste requiring treatment or disposal. From a cost perspective, the elimination of waste disposal fees and the maximization of raw material utility contribute to substantial cost savings in agrochemical intermediate manufacturing. Moreover, the use of commodity chemicals like acetic anhydride, potassium permanganate, and sulfuric acid ensures that the supply chain remains resilient against fluctuations in the availability of exotic or specialized reagents.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the theoretical doubling of yield for the active R-enantiomer from a fixed amount of starting material. By recovering the S-enantiomer and converting it back to the racemate, the effective cost per kilogram of the active ingredient is significantly lowered. This efficiency gain is achieved without the need for expensive chiral catalysts or complex asymmetric synthesis setups, relying instead on standard bulk chemicals. Consequently, the overall production cost structure is optimized, allowing for more competitive pricing in the global agrochemical market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Implementing a closed-loop recycling system for chiral intermediates enhances supply security by reducing the volatility associated with raw material sourcing. Since less fresh starting material is required to produce the same amount of active product, the strain on upstream suppliers is alleviated. This buffer capacity is crucial in times of market shortage or logistical disruption. Furthermore, the robustness of the chemical steps, which do not require sensitive anhydrous conditions or cryogenic temperatures for most stages, ensures consistent production throughput. This reliability is essential for meeting the rigorous delivery schedules demanded by downstream formulators and agricultural distributors.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations that are standard in the fine chemical industry, such as stirred tank reactors for acylation and oxidation, and fixed-bed or slurry reactors for hydrogenation. The avoidance of heavy metal catalysts in the early steps and the use of recoverable heterogeneous catalysts in the final step simplify waste management and environmental compliance. By minimizing the generation of chiral waste, the facility's environmental footprint is reduced, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. This sustainability profile enhances the corporate image and facilitates smoother regulatory approvals for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization process. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of adopting this technology. Understanding these aspects is crucial for technical teams evaluating the integration of this route into existing production lines.
Q: How does this racemization process improve overall yield for fungicide production?
A: By converting the inactive S-enantiomer back into a racemic mixture, manufacturers can theoretically double the yield of the active R-enantiomer from a given batch of starting material, significantly reducing raw material waste.
Q: What are the key reagents used in this five-step synthesis route?
A: The process utilizes common industrial reagents including acetic anhydride for acylation, potassium permanganate for oxidation, strong mineral acids like HCl or H2SO4 for dehydration and deacylation, and Pd-C for the final hydrogenation step.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the protocol avoids exotic catalysts and uses standard unit operations such as heating, filtration, and extraction, making it highly adaptable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Fluoro-1,1,3-trimethyl-2,3-dihydro-1H-inden-4-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral management in the production of high-performance agrochemicals. Our team of expert chemists has extensively analyzed the technological potential of the racemization methods described in CN108777958B and is fully prepared to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of intermediate we produce, meeting the exacting standards of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your manufacturing process, reduce waste, and secure a sustainable supply of high-quality chiral intermediates for your next-generation fungicidal products.
