Scalable Synthesis of Water-Soluble BODIPY Photosensitizers for Anticancer Therapy
Scalable Synthesis of Water-Soluble BODIPY Photosensitizers for Anticancer Therapy
In the rapidly evolving landscape of oncological therapeutics, patent CN109575061B introduces a groundbreaking approach to the design and synthesis of water-soluble anticancer photosensitizers that address critical limitations in current photodynamic therapy (PDT) protocols. Traditional photosensitizers often suffer from poor water solubility, uncertain component composition, and limited tissue penetration depth due to short-wavelength absorption, which severely restricts their clinical efficacy and safety profiles. This patented technology leverages the robust fluoroboron dipyrrole (BODIPY) scaffold, strategically modified with multiple carboxyl groups and heavy iodine atoms, to create a next-generation therapeutic agent with enhanced bioavailability and potent singlet oxygen generation capabilities. By shifting the maximum absorption wavelength into the red light region and ensuring amphipathic characteristics through polycarboxyl substitution, this innovation represents a significant leap forward for reliable photosensitizer suppliers aiming to meet the rigorous demands of modern pharmaceutical development.
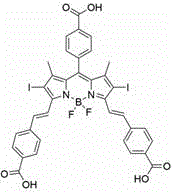
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of photodynamic therapy has relied heavily on first-generation photosensitizers such as hematoporphyrin derivatives, which present substantial challenges for both R&D directors and supply chain managers due to their inherent chemical heterogeneity and suboptimal pharmacokinetic properties. These conventional agents typically consist of complex mixtures with uncertain components, making batch-to-batch consistency difficult to guarantee and regulatory approval processes increasingly arduous for procurement teams. Furthermore, their maximum absorption wavelengths are predominantly located in the short-wavelength visible region, resulting in weak tissue penetration capability that limits treatment to superficial tumors while causing significant skin phototoxicity in patients. The hydrophobic nature of many traditional BODIPY analogs further complicates formulation, as they require organic solvents for dissolution, which are incompatible with biological systems and necessitate complex delivery vehicles that increase manufacturing costs and reduce overall therapeutic index.
The Novel Approach
The novel synthetic strategy outlined in the patent data overcomes these historical barriers by engineering a single-structure BODIPY derivative that combines precise molecular architecture with functional group optimization for biological compatibility. By introducing three carboxyl groups into the molecular framework, the inventors have successfully transformed a typically hydrophobic scaffold into an amphipathic molecule that exhibits superior water solubility without compromising its photophysical stability or fluorescence quantum yield. This structural modification not only facilitates direct administration in aqueous physiological environments but also enhances the interaction between the medicament and biological macromolecules, thereby improving bioavailability and tumor targeting efficiency. Additionally, the strategic expansion of the conjugated system through condensation reactions shifts the absorption spectrum into the red light region, allowing for deeper tissue penetration and minimizing damage to surrounding healthy tissues, which is a paramount concern for clinical adoption.
Mechanistic Insights into Heavy Atom Effect and Conjugation Expansion
The core scientific breakthrough of this synthesis lies in the sophisticated manipulation of electronic states through the heavy atom effect, where the introduction of iodine atoms at the 2,6-positions of the BODIPY core plays a pivotal role in enhancing therapeutic efficacy. From a mechanistic perspective, the presence of these heavy iodine atoms significantly increases the spin-orbit coupling constant, which promotes efficient intersystem crossing from the excited singlet state to the triplet state. This transition is critical because the triplet state is the precursor for energy transfer to molecular oxygen, resulting in the generation of cytotoxic singlet oxygen species that induce tumor cell apoptosis. Without this heavy atom modification, the fluorescence quantum yield might remain high, but the singlet oxygen quantum yield would be insufficient for effective photodynamic killing, rendering the compound merely a fluorescent probe rather than a therapeutic agent.
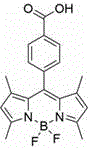
Following the initial core formation, the synthetic pathway proceeds through a carefully controlled iodination step that requires precise stoichiometric balance to avoid over-iodination or decomposition of the sensitive dipyrrole backbone. The use of iodic acid in conjunction with elemental iodine provides a mild yet effective oxidizing environment that facilitates electrophilic substitution specifically at the electron-rich pyrrole positions. This step is crucial for establishing the heavy atom effect described previously, and the resulting intermediate retains the carboxyl functionality necessary for subsequent solubility enhancements. The structural integrity of this iodinated intermediate is paramount, as any degradation at this stage would lead to impurities that are difficult to separate and could compromise the photostability of the final product, highlighting the importance of rigorous process control during this phase of manufacturing.
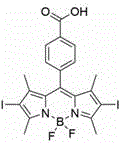
The final mechanistic transformation involves a Knoevenagel-type condensation reaction that expands the pi-conjugated system of the molecule, effectively red-shifting its absorption profile into the therapeutic window of 650 to 800 nanometers. By reacting the iodinated BODIPY core with p-carboxybenzaldehyde in the presence of a base catalyst and acetic acid, the synthesis extends the conjugation length through the formation of vinyl linkages, which lowers the energy gap between the highest occupied molecular orbital and the lowest unoccupied molecular orbital. This electronic tuning ensures that the photosensitizer can be activated by red light, which possesses greater tissue penetration depth compared to blue or green light, thereby enabling the treatment of deeper-seated tumors. Simultaneously, this reaction introduces two additional carboxyl groups, completing the polycarboxyl substitution pattern that defines the molecule's unique solubility profile and distinguishing it from earlier generations of hydrophobic photosensitizers.
How to Synthesize Polycarboxyl-Substituted BODIPY Efficiently
The preparation of this advanced photosensitizer follows a streamlined three-step protocol that balances high yield with operational simplicity, making it highly attractive for commercial scale-up of complex photosensitizers. The process begins with the condensation of readily available starting materials to form the core scaffold, followed by selective halogenation and final conjugation extension, each step designed to minimize side reactions and facilitate straightforward purification via silica gel column chromatography. While the detailed reaction conditions involve specific molar ratios and temperature controls to ensure optimal conversion, the overall route avoids the use of exotic catalysts or extreme pressures, relying instead on standard organic synthesis techniques that are easily transferable from laboratory to pilot plant scales. For a comprehensive breakdown of the specific reagents, equivalents, and workup procedures required to replicate this synthesis with high fidelity, please refer to the standardized guide below.
- React p-carboxybenzaldehyde with 2,4-dimethylpyrrole using TFA, followed by oxidation with DDQ and complexation with BF3·Et2O to form the monocarboxy-substituted BODIPY core (Compound A).
- Introduce heavy atom iodine at the 2,6-positions of Compound A using I2 and HIO3 in absolute ethanol to synthesize the iodofluoroboron dipyrrole derivative (Compound B).
- Perform a condensation reaction between Compound B and p-carboxybenzaldehyde using piperidine and glacial acetic acid in dry acetonitrile to expand the conjugated system and obtain the final polycarboxyl-substituted BODIPY derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for procurement managers and supply chain heads who are tasked with reducing lead time for high-purity photosensitizers while maintaining strict cost controls. The reliance on commodity chemicals such as p-carboxybenzaldehyde and 2,4-dimethylpyrrole as starting materials ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents that often plague the production of fine chemical intermediates. Furthermore, the absence of transition metal catalysts in the key bond-forming steps eliminates the need for expensive and time-consuming metal scavenging processes, which significantly simplifies downstream processing and reduces the overall environmental footprint of the manufacturing operation. This streamlined approach translates directly into substantial cost savings and faster turnaround times, allowing manufacturers to respond more agilely to market demands for anticancer drug ingredients.
- Cost Reduction in Manufacturing: The synthetic pathway is characterized by the use of inexpensive, commercially available raw materials and avoids the necessity for precious metal catalysts, which drastically lowers the bill of materials and operational expenditures. By eliminating complex metal removal steps and utilizing standard purification techniques like silica gel chromatography, the process minimizes waste generation and solvent consumption, leading to a more economically viable production model that supports competitive pricing strategies for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Because the synthesis relies on robust chemical transformations that are less sensitive to minor fluctuations in reaction conditions, the process demonstrates high reproducibility and scalability, ensuring consistent supply continuity for downstream pharmaceutical partners. The use of stable intermediates that can be isolated and stored if necessary provides additional flexibility in production scheduling, allowing manufacturers to build inventory buffers against potential disruptions in the global supply of fine chemicals and maintain reliable delivery schedules for critical oncology therapies.
- Scalability and Environmental Compliance: The method is inherently designed for industrial production, featuring high yields and few side reactions that simplify waste management and align with increasingly stringent environmental regulations. The ability to perform reactions under relatively mild conditions, such as reflux in common solvents like acetonitrile and ethanol, reduces energy consumption and safety hazards associated with high-pressure or cryogenic processes, making it an ideal candidate for green chemistry initiatives and sustainable manufacturing practices within the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this water-soluble BODIPY derivative, based on the specific technological disclosures found in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this photosensitizer into new drug formulations or for procurement specialists assessing the quality standards of potential suppliers. The answers provided reflect the mechanistic realities of the synthesis and the practical benefits observed in biological testing, offering a clear picture of why this molecule stands out in the crowded field of photodynamic therapy agents.
Q: How does the introduction of iodine atoms affect the photosensitizer's performance?
A: The introduction of heavy iodine atoms at the 2,6-positions significantly enhances the intersystem crossing rate, thereby increasing the triplet state and singlet oxygen quantum yield, which is critical for effective photodynamic therapy.
Q: What specific structural features confer water solubility to this BODIPY derivative?
A: The molecule incorporates three carboxyl groups—one on the central phenyl ring and two on the extended styryl arms—which provide strong hydrophilicity and allow for better physiological distribution compared to hydrophobic analogs.
Q: Why is this synthetic route considered superior to traditional porphyrin-based photosensitizers?
A: Unlike porphyrins which often have uncertain components and short-wavelength absorption, this BODIPY derivative offers a single, clear structure with absorption shifted to the red light region, ensuring deeper tissue penetration and easier purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycarboxyl-Substituted BODIPY Derivative Supplier
As a premier contract development and manufacturing organization, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for oncology applications, guaranteeing that every batch of photosensitizer delivered meets the highest international standards for safety and efficacy. We understand the critical nature of supply chain continuity in the pharmaceutical industry and are committed to providing a stable, high-quality source of this advanced intermediate to support your drug development pipelines.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. By engaging with our experts, you can obtain specific COA data and route feasibility assessments that will help you optimize your manufacturing strategy and accelerate your time to market. Let us partner with you to bring this innovative water-soluble anticancer photosensitizer from concept to clinic, leveraging our technical expertise to drive down costs and enhance the therapeutic potential of your photodynamic therapy programs.
