Advanced Synthesis of 1-(6-Chloropyridazin-4-yl) Ethyl-1-Ketone for Commercial Scale-Up
The pharmaceutical industry continuously demands novel heterocyclic building blocks to accelerate the discovery of next-generation therapeutics, particularly those exhibiting potent biological activities such as anti-tumor or herbicidal properties. Addressing this critical need, the recent patent CN114031558A discloses a robust and highly efficient preparation method for 1-(6-chloropyridazin-4-yl) ethyl-1-ketone, a valuable intermediate that has previously lacked a reported synthetic pathway in scientific literature. This innovation represents a significant breakthrough in pyridazine chemistry, offering a streamlined three-step sequence that transforms readily available starting materials into a complex, functionalized scaffold with exceptional yield and purity. By leveraging advanced organometallic catalysis and precise halogenation techniques, this methodology not only fills a gap in chemical space but also provides a reliable foundation for the development of new small molecule drugs. For R&D teams and procurement specialists alike, understanding the nuances of this patented route is essential for securing a stable supply of high-quality intermediates necessary for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of pyridazine rings to introduce ketone moieties at specific positions has been fraught with significant synthetic challenges, often requiring harsh reaction conditions that compromise the integrity of the heterocyclic core. Conventional approaches frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate, thereby drastically reducing the overall process efficiency. Furthermore, many existing methods rely on aggressive reagents or extreme temperatures that can degrade sensitive functional groups, limiting their applicability in the synthesis of complex pharmaceutical candidates. The lack of a dedicated, optimized route for 1-(6-chloropyridazin-4-yl) ethyl-1-ketone meant that researchers were forced to rely on inefficient, multi-step workarounds that increased both the time-to-market and the cost of goods sold. These historical limitations have created a bottleneck in the supply chain for pyridazine-based APIs, necessitating a more sophisticated and controlled chemical strategy.
The Novel Approach
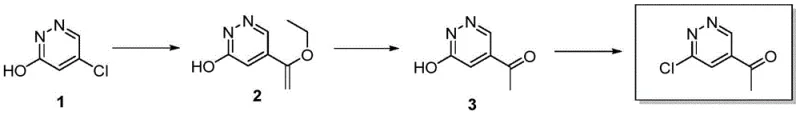
The methodology outlined in patent CN114031558A introduces a paradigm shift by utilizing a strategic Stille coupling reaction to install the carbon framework prior to final functionalization, effectively bypassing the selectivity issues of direct substitution. This novel approach begins with the coupling of 5-chloropyridazin-3-ol with tributyl(1-ethoxyvinyl)tin, a transformation that proceeds under mild nitrogen-protected conditions to yield a stable vinyl ether intermediate with high fidelity. Subsequent hydrolysis and chlorination steps are carefully tuned to operate at moderate temperatures, ensuring that the delicate pyridazine ring remains intact while achieving the desired substitution pattern. By decoupling the carbon-carbon bond formation from the final halogenation, this route minimizes side reactions and simplifies the purification process, resulting in a final product with HPLC purity exceeding 99%. This level of control translates directly into operational excellence, offering a scalable solution that aligns perfectly with the rigorous quality standards of the global pharmaceutical market.
Mechanistic Insights into Pd-Catalyzed Stille Coupling and Chlorination
The cornerstone of this synthesis is the palladium-catalyzed Stille coupling reaction, which facilitates the cross-coupling of the chloropyridazine substrate with the organotin reagent through a well-defined catalytic cycle. In this mechanism, the active Pd(0) species undergoes oxidative addition into the carbon-chlorine bond of the pyridazine ring, forming a reactive organopalladium intermediate that is poised for transmetallation. The tributyl(1-ethoxyvinyl)tin reagent then transfers its vinyl group to the palladium center, a step that is critically dependent on the presence of an organic base such as triethylamine or DIEA to activate the tin species. Following transmetallation, reductive elimination occurs to forge the new carbon-carbon bond, regenerating the Pd(0) catalyst and releasing the 5-(1-ethoxyvinyl)pyridazin-3-ol intermediate. This mechanistic pathway is highly advantageous because it tolerates the hydroxyl group on the pyridazine ring without the need for additional protecting groups, thereby streamlining the synthetic sequence and reducing waste generation.
Following the coupling step, the conversion of the vinyl ether to the acetyl group and subsequent chlorination involves precise acid-base chemistry and nucleophilic substitution mechanisms. The hydrolysis step utilizes dilute hydrochloric acid in tetrahydrofuran to cleave the ethoxyvinyl group, generating the ketone functionality through an enol intermediate that tautomerizes to the more stable carbonyl form. The final chlorination employs phosphorus oxychloride (POCl3) as a potent chlorinating agent, which activates the hydroxyl group of the pyridazinone ring via the formation of a dichlorophosphate ester. This activated species is then displaced by a chloride ion in an SNAr-like fashion, effectively replacing the hydroxyl group with a chlorine atom to yield the target 1-(6-chloropyridazin-4-yl) ethyl-1-ketone. The use of acetonitrile as a solvent in this final step ensures optimal solubility and reaction kinetics, allowing the process to proceed efficiently at temperatures between 80°C and 100°C while maintaining high selectivity for the desired product.
How to Synthesize 1-(6-Chloropyridazin-4-yl) Ethyl-1-Ketone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding reagent stoichiometry and temperature control to maximize yield and safety. The process begins with the dissolution of the starting material in DMF, followed by the sequential addition of the tin reagent, base, and palladium catalyst under an inert atmosphere to prevent oxidation of the sensitive catalytic species.
- Perform Stille coupling of 5-chloropyridazin-3-ol with tributyl(1-ethoxyvinyl)tin using Pd(PPh3)2Cl2 catalyst in DMF at 90-140°C.
- Hydrolyze the resulting vinyl ether intermediate using dilute hydrochloric acid in THF at 60-80°C to form the ketone.
- React the hydroxy-ketone with phosphorus oxychloride (POCl3) and organic base in acetonitrile at 80-100°C to obtain the final chlorinated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The reliance on commercially available and relatively inexpensive starting materials, such as 5-chloropyridazin-3-ol and tributyl(1-ethoxyvinyl)tin, ensures a stable and predictable supply chain that is less susceptible to market volatility. Furthermore, the use of common industrial solvents like DMF, THF, and acetonitrile eliminates the need for specialized or hazardous solvent handling infrastructure, thereby reducing capital expenditure and operational complexity for manufacturing partners. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, further enhances the feasibility of scaling this process from gram-scale R&D batches to multi-ton commercial production runs without significant re-engineering.
- Cost Reduction in Manufacturing: The elimination of complex protecting group strategies and the high efficiency of the Stille coupling step significantly reduce the overall number of unit operations required, leading to lower labor and utility costs per kilogram of product. By achieving high yields in each of the three steps, the process minimizes the loss of valuable intermediates, which is a critical factor in optimizing the cost of goods for high-value pharmaceutical intermediates. Additionally, the straightforward purification methods described, such as silica gel chromatography and simple filtration, avoid the need for expensive preparative HPLC or complex distillation setups, further driving down processing expenses.
- Enhanced Supply Chain Reliability: The synthetic route utilizes reagents that are widely sourced from global chemical suppliers, mitigating the risk of single-source dependency and ensuring continuity of supply even during market disruptions. The mild reaction conditions and short reaction times contribute to faster batch cycles, allowing manufacturers to respond more agilely to fluctuating demand from downstream API producers. This reliability is paramount for long-term supply agreements, where consistent quality and on-time delivery are non-negotiable requirements for maintaining regulatory compliance and production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be easily translated from laboratory glassware to large-scale stainless steel reactors without compromising safety or yield. The generation of waste is minimized through high atom economy in the coupling step and the use of recyclable solvents, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. This focus on sustainability not only meets regulatory standards but also appeals to environmentally conscious stakeholders who prioritize eco-friendly supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific pyridazine derivative, based on the detailed disclosures within the patent documentation.
Q: What is the primary advantage of this Stille coupling route for pyridazine functionalization?
A: The primary advantage is the high regioselectivity and mild conditions achieved by using tributyl(1-ethoxyvinyl)tin, which allows for the efficient introduction of the acetyl precursor without damaging the sensitive pyridazine ring structure.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes straightforward purification methods such as silica gel column chromatography and crystallization, consistently achieving HPLC purity levels of 99% in the final step, which is critical for downstream API synthesis.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method employs common industrial solvents like DMF, THF, and acetonitrile, and operates at moderate temperatures (60-140°C), making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(6-Chloropyridazin-4-yl) Ethyl-1-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications, and we are uniquely positioned to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex organometallic reactions like the Stille coupling described in CN114031558A, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards before it leaves our site. We understand that consistency is key in pharmaceutical manufacturing, which is why our team of expert chemists works closely with clients to optimize every parameter of the synthesis, guaranteeing a product that is ready for immediate use in your downstream processes.
We invite you to contact our technical procurement team today to discuss how we can tailor this synthesis to your specific volume requirements and timeline constraints. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to further optimize the supply chain for your specific project needs. We encourage you to reach out now to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will accelerate your drug development programs and secure your position in the competitive global market.
