Advanced Synthesis of 4-tert-butyloxycarbonylpiperazine Naphthalimide for Commercial Pharmaceutical Applications
Advanced Synthesis of 4-tert-butyloxycarbonylpiperazine Naphthalimide for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel scaffolds that offer enhanced biological activity alongside manufacturability, and patent CN110194740B presents a significant breakthrough in this domain with its disclosure of 4-tert-butyloxycarbonylpiperazine-1,8-naphthalimide derivatives. This specific chemical architecture addresses the critical need for high-purity antitumor intermediates that can be reliably sourced for drug development pipelines. The innovation lies in the strategic introduction of a tert-butyloxycarbonylpiperazine functional group at the 4-position of the 1,8-naphthalimide core, a modification that has been shown to significantly enhance bioactivity compared to traditional analogues like aminonaphthene. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital, as it represents a viable pathway for producing complex pharmaceutical intermediates with improved therapeutic potential. The technical robustness of this method ensures that supply chain stakeholders can anticipate consistent quality and scalability, which are paramount for maintaining continuity in the production of next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for naphthalimide derivatives often suffer from cumbersome reaction conditions that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional methodologies frequently rely on harsh reagents or multi-step protection-deprotection sequences that introduce unnecessary impurities and reduce overall yield, thereby escalating the cost of goods sold. Furthermore, the purification of intermediates in older processes often requires extensive chromatographic separation, which is not only time-consuming but also difficult to translate from laboratory benchtop to industrial manufacturing scales without significant loss of material. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and increased vulnerability to raw material price fluctuations. For procurement managers, these technical limitations translate directly into higher costs and reduced reliability, making it challenging to secure a steady flow of high-quality intermediates required for clinical and commercial drug production.
The Novel Approach
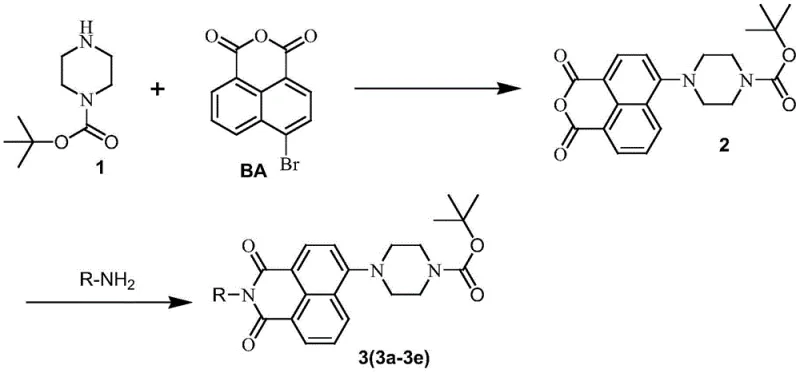
In contrast, the novel approach detailed in the patent data utilizes a streamlined two-step condensation process that drastically simplifies the manufacturing workflow while maintaining exceptional product integrity. By reacting tert-butyloxycarbonylpiperazine directly with 4-bromo-1,8-naphthalic anhydride under controlled heating conditions, the method achieves the formation of the key intermediate with high efficiency and minimal byproduct formation. This route eliminates the need for complex catalytic systems or exotic reagents, relying instead on readily available organic solvents such as ethanol or DMF, which facilitates cost reduction in pharmaceutical intermediates manufacturing. The subsequent amidation step with various amines is equally robust, allowing for the rapid generation of a diverse library of derivatives without compromising on purity or yield. This methodological elegance ensures that the production process is not only chemically sound but also commercially viable for large-scale operations.
Mechanistic Insights into Naphthalimide Derivative Formation
The core of this technological advancement lies in the precise structural modification of the 1,8-naphthalimide scaffold, which serves as the foundation for its enhanced antitumor properties. The introduction of the tert-butyloxycarbonylpiperazine moiety at the 4-position alters the electronic and steric environment of the molecule, facilitating better interaction with biological targets such as DNA or specific enzymes involved in tumor proliferation. This structural feature is critical for R&D teams evaluating the structure-activity relationship (SAR) of new drug candidates, as it provides a versatile handle for further functionalization or optimization. The stability of the Boc group under the reaction conditions ensures that the piperazine ring remains intact throughout the synthesis, preventing unwanted side reactions that could compromise the final product's efficacy. Understanding this mechanistic detail is essential for ensuring that the commercial scale-up of complex naphthalimide derivatives maintains the precise stereochemistry and purity required for regulatory approval.
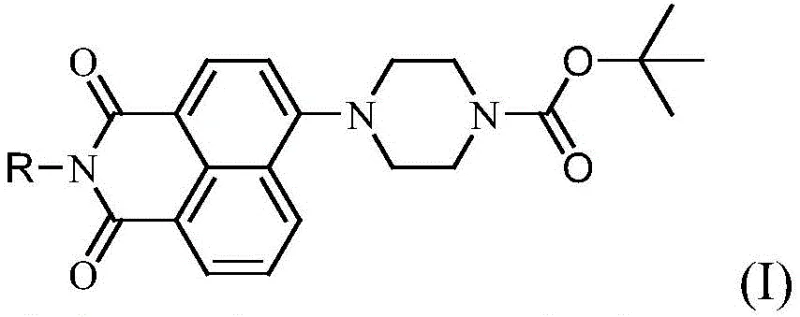
Furthermore, the impurity control mechanism inherent in this synthesis route is designed to meet the stringent purity specifications demanded by the global pharmaceutical market. The use of specific solvent systems and temperature controls during the condensation reactions minimizes the formation of regioisomers and over-reacted byproducts, which are common challenges in naphthalimide chemistry. Post-reaction purification strategies, such as recrystallization from methanol or ethanol, are highly effective in removing trace impurities, resulting in a final product that exhibits consistent quality batch after batch. This level of control is indispensable for supply chain heads who must guarantee the continuity of supply without the risk of quality deviations that could halt production lines. The ability to produce high-purity antitumor intermediates with such reliability positions this technology as a preferred choice for manufacturers aiming to reduce lead time for high-purity pharmaceutical intermediates in their procurement strategies.
How to Synthesize 4-tert-butyloxycarbonylpiperazine-1,8-naphthalimide Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity, allowing manufacturing teams to implement the process with standard equipment and common chemical feedstocks. The procedure begins with the condensation of the starting materials in a suitable organic solvent, followed by a straightforward workup involving cooling and filtration to isolate the intermediate. This intermediate is then subjected to a second reaction with the desired amine component, where reaction progress is easily monitored via thin-layer chromatography to ensure complete conversion. The detailed standardized synthesis steps see the guide below, which outlines the specific parameters for temperature, solvent volume, and reaction time to achieve optimal results. This clarity in process definition reduces the risk of operational errors and ensures that the technical transfer from R&D to production is seamless and efficient.
- Condense tert-butyloxycarbonylpiperazine with 4-bromo-1,8-naphthalic anhydride in organic solvent at 80-120°C to form the intermediate anhydride.
- React the intermediate with specific amines (e.g., piperonylethylamine) in alcohol or aprotic solvent at 60-80°C.
- Purify the crude target compound via recrystallization or silica gel column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial benefits that directly address the pain points of cost, reliability, and scalability faced by procurement and supply chain teams. The elimination of expensive transition metal catalysts and the use of common organic solvents significantly lower the raw material costs, contributing to a more competitive pricing structure for the final intermediate. Additionally, the simplicity of the post-treatment process, which often involves mere filtration and recrystallization rather than complex chromatography, reduces the consumption of consumables and labor hours, further driving down manufacturing expenses. These factors combine to create a robust economic case for adopting this technology, enabling companies to achieve significant cost savings without compromising on the quality or performance of the drug substance. For organizations looking to optimize their supply chain, this process represents a strategic opportunity to enhance margin and operational efficiency.
- Cost Reduction in Manufacturing: The process design inherently minimizes waste and energy consumption by utilizing efficient reaction conditions and recoverable solvents, which translates to lower operational expenditures. By avoiding the use of precious metal catalysts that require expensive removal steps, the overall cost of production is drastically reduced, allowing for more flexible pricing strategies in a competitive market. This economic efficiency is crucial for maintaining profitability while supplying high-quality intermediates to the pharmaceutical sector.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 4-bromo-1,8-naphthalic anhydride and Boc-piperazine ensures that the supply chain is not vulnerable to shortages of exotic reagents. This accessibility guarantees a steady flow of raw materials, reducing the risk of production delays and ensuring that delivery commitments to downstream clients are consistently met. For supply chain heads, this reliability is a key metric in vendor selection and risk management strategies.
- Scalability and Environmental Compliance: The synthesis route is inherently scalable, having been demonstrated to work effectively with standard heating and stirring equipment that is common in chemical manufacturing facilities. Furthermore, the use of less hazardous solvents and the generation of minimal waste align with modern environmental compliance standards, reducing the burden of waste disposal and regulatory reporting. This alignment with green chemistry principles not only mitigates environmental risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthalimide derivatives, based on the detailed patent specifications. These insights are intended to clarify the feasibility of the synthesis route and its potential impact on drug development timelines. By understanding the specific advantages and operational requirements, stakeholders can make informed decisions about integrating this technology into their existing portfolios. The answers provided reflect the rigorous data and experimental results documented in the intellectual property, ensuring accuracy and relevance for technical audiences.
Q: What are the key advantages of this naphthalimide synthesis route?
A: The patented method offers a short preparation period, simple post-treatment, and low cost while yielding derivatives with high purity and stable quality compared to conventional methods.
Q: What is the biological activity profile of these derivatives?
A: In vitro tests demonstrate that specific derivatives exhibit superior antitumor activity against cell lines like MGC-803 and HepG2 compared to reference compounds like aminonaphthene.
Q: Which solvents are recommended for the condensation reaction?
A: The process utilizes alcohol solvents like ethanol or aprotic solvents such as DMF and DMSO, allowing flexibility in process optimization for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-tert-butyloxycarbonylpiperazine-1,8-naphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative chemical pathways into commercial reality, and we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of naphthalimide chemistry, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards required by global regulatory bodies. We are committed to providing a seamless partnership that supports your R&D and commercialization goals, leveraging our state-of-the-art facilities to deliver high-quality intermediates consistently. Our dedication to excellence ensures that your supply chain remains robust and resilient, allowing you to focus on advancing your therapeutic candidates without concern for material availability.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis tailored to your volume needs. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process and accelerate your time to market. Let us demonstrate our capability to be your trusted partner in the synthesis of complex pharmaceutical intermediates, driving value and efficiency into your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
