Advanced Synthesis of m-Nitro-beta-hydroxyethyl Sulfone for Reactive Dye Manufacturing
Advanced Synthesis of m-Nitro-beta-hydroxyethyl Sulfone for Reactive Dye Manufacturing
The global demand for high-performance reactive dyes, particularly KN and M types used in textile finishing, necessitates a robust supply chain for critical intermediates like m-nitro-beta-hydroxyethyl sulfone. Recent intellectual property developments, specifically patent CN110194729B, have introduced a transformative preparation method that fundamentally alters the production landscape for this essential dye precursor. By shifting away from traditional sodium sulfite reduction towards a hydrazine hydrate-mediated pathway, this technology addresses long-standing inefficiencies in yield and waste management. For R&D directors and procurement strategists, understanding this shift is vital, as it represents a move towards cleaner, more atom-economical manufacturing protocols that align with modern green chemistry principles. This report analyzes the technical merits of this innovation, providing a roadmap for integrating these superior synthetic routes into commercial supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of m-nitro-beta-hydroxyethyl sulfone has relied heavily on the reduction of m-nitrobenzenesulfonyl chloride using excessive sodium sulfite in an aqueous medium. This legacy process is fraught with significant chemical and operational inefficiencies that burden both the balance sheet and environmental compliance teams. The primary chemical drawback is the susceptibility of the sulfonyl chloride to hydrolysis under the reaction conditions, leading to the formation of m-nitrobenzenesulfonic acid sodium salt as a major byproduct rather than the desired sulfinate. As illustrated in the reaction scheme below, this side reaction competes aggressively with the reduction, capping the overall yield at approximately 79 percent and generating substantial quantities of inorganic salts.
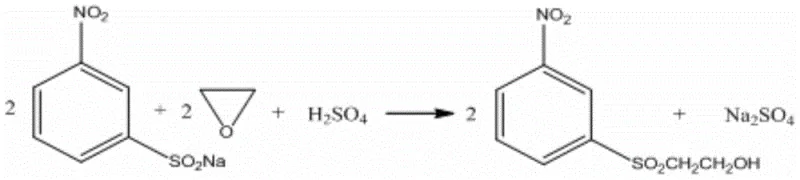
Furthermore, the downstream processing of this traditional method is exceptionally cumbersome and waste-intensive. The reaction mixture requires filtration to separate the crude sodium m-nitrobenzenesulfinate, generating a filtrate laden with high concentrations of sodium sulfate, sodium chloride, and unreacted sulfites, resulting in a Chemical Oxygen Demand (COD) of roughly 62000mg/L. This high-salinity wastewater is difficult and costly to treat, often requiring specialized evaporation or membrane technologies. Additionally, the solid filter cake requires further refining and decolorization steps, generating approximately 70kg of solid waste residues per ton of final product, thereby complicating the waste disposal logistics for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110194729B utilizes hydrazine hydrate as the reducing agent, offering a streamlined, high-efficiency alternative that bypasses the pitfalls of the sulfite route. This novel approach leverages the strong reducing power of hydrazine to convert m-nitrobenzenesulfonyl chloride directly into sodium m-nitrobenzenesulfinate with exceptional selectivity. The reaction is conducted under controlled pH conditions (5.0-7.0) and moderate temperatures (40-70°C), which effectively suppresses the hydrolysis side reactions that plague the traditional method. Consequently, the yield of the intermediate sulfinate skyrockets to over 98.5 percent, representing a massive gain in material efficiency.
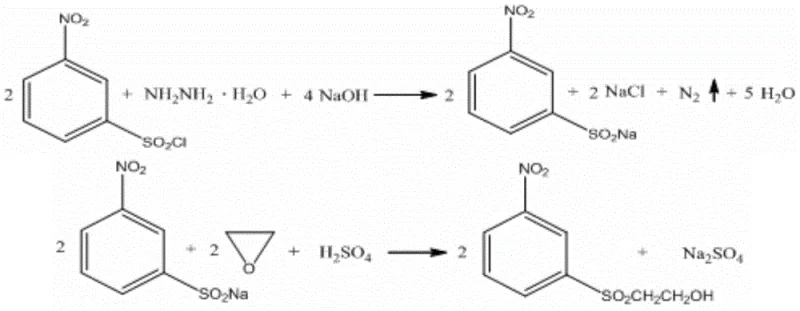
Beyond the impressive yield metrics, the true commercial value of this novel approach lies in its potential for process intensification and waste reduction. Because the sodium m-nitrobenzenesulfinate produced in situ is of such high purity, it can be subjected directly to the subsequent hydroxyethylation step with ethylene oxide without the need for intermediate isolation, filtration, or recrystallization. This telescoping of steps eliminates the generation of the high-salinity filtrate entirely and removes the need for solid waste-generating refining processes. The result is a cleaner manufacturing footprint that reduces high-salinity wastewater discharge by approximately 5 tons and solid waste by 70kg for every ton of product manufactured, significantly lowering the environmental burden and associated treatment costs.
Mechanistic Insights into Hydrazine-Mediated Reduction and Hydroxyethylation
The core of this technological advancement rests on the precise mechanistic control of the reduction phase using hydrazine hydrate. Unlike sulfite ions, which act as nucleophiles susceptible to competing hydrolysis pathways in aqueous media, hydrazine functions as a potent two-electron donor that rapidly reduces the sulfonyl chloride moiety to the sulfinate anion. The reaction kinetics are highly favorable when the pH is meticulously maintained between 5.0 and 7.0 using a liquid base, ensuring that the hydrazine remains in its active form while preventing the acid-catalyzed decomposition of the sensitive sulfonyl chloride starting material. This delicate pH balance is critical; if the environment becomes too acidic, hydrolysis accelerates, but if it becomes too alkaline, unwanted side reactions with the nitro group or over-reduction could theoretically occur, although the patent data suggests excellent chemoselectivity for the sulfonyl group.
Following the reduction, the mechanism transitions to a nucleophilic ring-opening of ethylene oxide by the generated sulfinate anion. This hydroxyethylation step is performed in the same reaction vessel, capitalizing on the high concentration of the nucleophile. The process requires a shift in pH control to a slightly neutral-to-basic range of 7.0-7.6, maintained by the addition of dilute sulfuric acid to counteract the basicity introduced by the reagents or byproducts. Maintaining this specific pH window is essential to drive the alkylation to completion while minimizing the polymerization of ethylene oxide, a common side reaction in ethoxylation processes. The temperature is elevated to 50-70°C to provide the necessary activation energy for the ring opening, ensuring that the reaction proceeds to high conversion within a reasonable timeframe of 18 to 22 hours, ultimately delivering the target beta-hydroxyethyl sulfone with purity levels exceeding 96 percent.
How to Synthesize m-Nitro-beta-hydroxyethyl Sulfone Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and thermal management to ensure safety and reproducibility. The process begins with the preparation of the hydrazine solution, followed by the controlled addition of the sulfonyl chloride, and concludes with the pressurized addition of ethylene oxide. Operators must strictly adhere to the specified temperature ramps and pH setpoints to maximize the yield advantages described in the patent literature. While the chemistry is robust, the handling of ethylene oxide and hydrazine requires specialized equipment and safety protocols typical of fine chemical manufacturing. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Reduce m-nitrobenzenesulfonyl chloride using hydrazine hydrate at 40-70°C while maintaining pH 5.0-7.0 to form sodium m-nitrobenzenesulfinate.
- Directly add ethylene oxide to the reaction mixture, controlling temperature at 50-70°C and pH 7.0-7.6 to complete hydroxyethylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrazine-based synthesis route offers compelling strategic advantages that extend far beyond simple yield improvements. The elimination of intermediate isolation steps fundamentally reshapes the cost structure of manufacturing this dye intermediate. By removing the filtration, washing, and refining stages required in the traditional sulfite process, manufacturers can drastically reduce labor hours, utility consumption, and equipment occupancy time. This streamlining translates directly into lower operating expenses (OPEX) and a more competitive pricing structure for the final product, allowing buyers to secure high-quality intermediates at more sustainable price points without compromising on specification integrity.
- Cost Reduction in Manufacturing: The most significant economic driver of this new process is the drastic simplification of the workflow. In the traditional method, the low yield of roughly 79 percent means that over 20 percent of the expensive starting material, m-nitrobenzenesulfonyl chloride, is wasted as hydrolysis byproducts or lost during purification. By boosting the yield to over 98.5 percent, the new method maximizes raw material utilization, effectively lowering the cost of goods sold (COGS) per kilogram of output. Furthermore, the removal of the wastewater treatment burden associated with high-salinity effluents reduces the financial liability related to environmental compliance and waste disposal fees, contributing to substantial long-term cost savings.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, this process offers greater resilience. The traditional method's reliance on multiple batch steps and filtration creates multiple potential choke points where production delays can occur due to equipment fouling or filter blinding. The telescoped nature of the hydrazine route minimizes these mechanical failure points, leading to more predictable batch cycle times and consistent output volumes. Additionally, the raw materials required—hydrazine hydrate and ethylene oxide—are commodity chemicals with well-established global supply chains, reducing the risk of raw material shortages that might affect more exotic reagents used in alternative synthetic pathways.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify globally, the environmental profile of a synthesis route becomes a critical factor in vendor selection. This method's ability to reduce wastewater volume by 5 tons per ton of product and eliminate 70kg of solid waste per ton positions it as a future-proof technology. Facilities adopting this process will find it easier to meet stringent discharge limits and sustainability goals, reducing the risk of regulatory shutdowns or fines. The simplified equipment requirements also mean that scaling up from pilot to commercial production involves less capital expenditure (CAPEX) on complex filtration and drying infrastructure, facilitating faster capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of transitioning from legacy sulfite-based processes to this modern hydrazine-mediated protocol.
Q: How does the hydrazine hydrate method improve yield compared to sodium sulfite reduction?
A: The novel hydrazine-based route achieves yields exceeding 98.5%, a significant improvement over the traditional sodium sulfite method which typically caps at around 79% due to hydrolysis side reactions.
Q: What are the environmental benefits of this new synthesis pathway?
A: This process eliminates the need for intermediate filtration and refining, reducing high-salinity wastewater generation by approximately 5 tons and solid waste by 70kg per ton of product.
Q: Is the intermediate sodium m-nitrobenzenesulfinate isolated in this process?
A: No, the process is designed for telescoping; the sodium m-nitrobenzenesulfinate generated in situ possesses high enough purity to undergo direct hydroxyethylation without isolation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Nitro-beta-hydroxyethyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic methodologies is key to maintaining competitiveness in the global dye intermediate market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110194729B are fully realized in large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of m-nitro-beta-hydroxyethyl sulfone meets the exacting standards required for high-performance reactive dye synthesis.
We invite procurement leaders and technical directors to engage with our team to explore how this optimized route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this high-yield process for your specific volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production needs.
