Advanced One-Pot Synthesis of Spiro Indole Derivatives Using Recyclable Basic Ionic Liquids for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds that serve as the backbone for novel therapeutic agents. A significant breakthrough in this domain is documented in Chinese Patent CN110862401B, which discloses a highly efficient method for preparing spiro indole derivatives using a basic ionic liquid one-pot method. This technology addresses critical bottlenecks in the synthesis of spiro-fused heterocycles, which are renowned for their potent biological activities ranging from antibacterial to antiproliferative effects. By leveraging a specialized basic ionic liquid catalyst, this process eliminates the need for toxic organic bases and volatile organic solvents that have traditionally plagued this chemical transformation. The innovation lies not only in the chemical efficiency but also in the operational simplicity, allowing for the direct recycling of the catalytic system without complex regeneration steps. For R&D directors and procurement managers alike, this represents a paradigm shift towards greener manufacturing that does not compromise on yield or purity. The ability to produce high-value pharmaceutical intermediates with reduced environmental impact and simplified downstream processing makes this patent a cornerstone for modern supply chain strategies in the life sciences sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro indole derivatives has been fraught with significant technical and economic challenges that hinder large-scale commercialization. Traditional methodologies often rely on homogeneous organic base catalysts such as piperidine, which, while effective in small-scale laboratory settings, present severe drawbacks in industrial applications. These conventional processes typically require long reaction times and generate substantial amounts of byproducts, leading to low overall yields and complicated purification workflows. Furthermore, the reliance on volatile organic solvents contributes to serious environmental pollution and increases the operational costs associated with solvent recovery and waste disposal. Even newer attempts using eutectic solvents, while an improvement in terms of toxicity, have struggled with issues related to expensive raw material costs and limited catalyst recyclability. The inability to efficiently recover and reuse catalysts in these older methods results in a linear increase in production costs as batch sizes grow, making them economically unviable for the mass production of high-purity pharmaceutical intermediates. Additionally, the complex workup procedures often involving extraction and recrystallization steps further erode profit margins and extend lead times for delivery.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN110862401B introduces a streamlined one-pot synthesis that utilizes a basic ionic liquid to catalyze the reaction between thiochromanone, malononitrile, and isatin. This novel approach drastically reduces reaction times to a mere 22 to 36 minutes under reflux conditions, achieving impressive yields ranging from 84% to 94%. The use of a 95-98% aqueous methanol solution as the reaction medium significantly lowers the environmental footprint compared to pure organic solvents, aligning with global sustainability goals. Crucially, the product separation is remarkably simple; upon cooling, the product precipitates as a solid that can be isolated via suction filtration, bypassing the need for energy-intensive chromatography or extensive extraction protocols. The reaction pathway is depicted below, illustrating the clean conversion of starting materials into the complex spiro-fused structure.
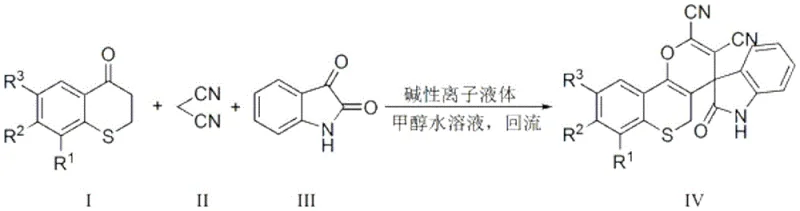
This efficiency is further amplified by the unique properties of the catalyst system, which allows the filtrate to be directly reused for subsequent batches. This capability transforms the economic model of production, turning a consumable cost center into a durable asset that drives down the cost per kilogram over time. For supply chain heads, this means a more predictable and stable production schedule with fewer interruptions for catalyst replenishment or waste treatment.
Mechanistic Insights into Basic Ionic Liquid-Catalyzed Cyclization
The success of this synthetic route is fundamentally rooted in the unique physicochemical properties of the basic ionic liquid catalyst employed in the reaction. Unlike traditional molecular bases, this ionic liquid possesses a high density of active basic sites that are uniformly distributed throughout the reaction medium, facilitating a rapid and selective domino Knoevenagel-Michael reaction sequence. The structure of the catalyst, characterized by its dual cationic and anionic functional groups, creates a microenvironment that stabilizes transition states and accelerates the formation of the carbon-carbon bonds necessary for spiro-ring closure. This structural integrity ensures that the active sites do not leach or degrade easily, which is the primary reason for the catalyst's exceptional recyclability observed over multiple cycles. The mechanism involves the initial activation of the methylene group in malononitrile by the basic sites, followed by a condensation with the carbonyl group of the thiochromanone, and subsequently a Michael addition to the isatin moiety. This cascade occurs seamlessly within the ionic liquid matrix, minimizing side reactions that typically lead to polymeric impurities or regioisomers.
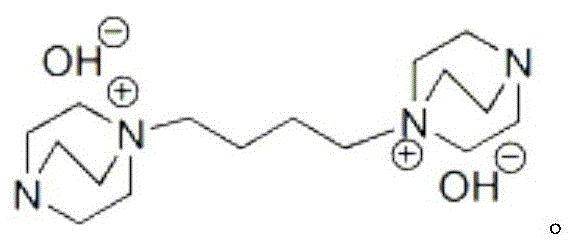
From an impurity control perspective, the use of this specific catalyst in an aqueous methanol system promotes the precipitation of the target product while keeping most polar byproducts and unreacted starting materials in the solution phase. This differential solubility acts as a built-in purification step, ensuring that the crude solid obtained after filtration already possesses high purity levels. For R&D teams focused on regulatory compliance, this inherent selectivity reduces the burden on analytical quality control and simplifies the validation of the manufacturing process. The robustness of the catalytic cycle means that even with substrate variations, such as the introduction of chloro or fluoro substituents on the thiochromanone ring, the reaction kinetics remain favorable, ensuring consistent product quality across different batches. This mechanistic reliability is essential for scaling up from gram-scale discovery to multi-ton commercial production without the risk of unexpected process deviations.
How to Synthesize Spiro Indole Derivatives Efficiently
The implementation of this synthesis protocol is designed to be straightforward and adaptable to standard reactor configurations found in most fine chemical manufacturing facilities. The process begins with the precise weighing of thiochromanone derivatives, malononitrile, and isatin in equimolar ratios, ensuring stoichiometric balance for maximum atom economy. These reagents are introduced into a reaction vessel containing the basic ionic liquid catalyst and the aqueous methanol solvent, where the mixture is subjected to heating under reflux. The detailed standardized operating procedures, including specific temperature ramping rates and agitation speeds optimized for heat transfer, are critical for maintaining the narrow reaction window of 22 to 36 minutes. Following the reaction completion, indicated by TLC analysis, the mixture is cooled to ambient temperature to induce crystallization of the product.
- Combine thiochromanone, malononitrile, and isatin in a 1: 1:1 molar ratio with 6-8% molar weight of basic ionic liquid catalyst in a reaction vessel containing 95-98% aqueous methanol.
- Heat the mixture to reflux under atmospheric pressure for 22 to 36 minutes to facilitate the domino Knoevenagel-Michael reaction.
- Cool the reaction to room temperature, filter the precipitated solid, wash with aqueous methanol, and dry under vacuum to obtain the high-purity spiro indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this basic ionic liquid technology offers profound advantages that extend far beyond simple chemical yield improvements. The primary economic driver is the drastic reduction in raw material costs achieved through the elimination of expensive, single-use catalysts and the minimization of solvent consumption. By utilizing a catalyst that can be recycled at least seven times without significant loss of activity, the effective cost of the catalytic system per unit of product drops precipitously, directly improving the gross margin of the final API intermediate. Furthermore, the use of aqueous methanol as a solvent replaces costly and hazardous pure organic solvents, reducing both procurement expenses and the regulatory burden associated with volatile organic compound (VOC) emissions. This shift not only lowers immediate operational expenditures but also future-proofs the supply chain against tightening environmental regulations that often penalize traditional synthetic routes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the removal of expensive transition metal catalysts and the simplification of the workup procedure. Since the product precipitates directly from the reaction mixture, the need for resource-intensive purification techniques like column chromatography is completely eliminated, resulting in substantial savings in silica gel, solvents, and labor hours. The ability to recycle the mother liquor containing the ionic liquid catalyst means that the facility does not need to continuously purchase fresh catalyst for every batch, creating a compounding cost saving effect over the lifecycle of the product. Additionally, the high atom economy of the one-pot reaction ensures that the majority of the input mass is converted into valuable product rather than waste, optimizing the utilization of every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, the raw materials required for this synthesis—thiochromanone, malononitrile, and isatin—are commodity chemicals that are widely available from multiple global suppliers. This diversity in the supply base mitigates the risk of shortages or price spikes that can occur when relying on proprietary or niche reagents. The robustness of the reaction conditions, which tolerate atmospheric pressure and moderate temperatures, means that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment. This compatibility with existing infrastructure allows for rapid technology transfer and scale-up, ensuring that production timelines can be met consistently even during periods of high market demand. The simplified workflow also reduces the potential for human error during operation, leading to fewer failed batches and more reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method make it exceptionally well-suited for large-scale industrial application. The reduction in solvent volume and the use of a less toxic aqueous methanol mixture significantly lower the volume of hazardous waste generated per kilogram of product. This reduction in waste stream complexity simplifies the effluent treatment process, lowering the costs associated with wastewater management and environmental compliance reporting. The scalability is further supported by the exothermic nature of the reaction being easily manageable under reflux conditions, preventing thermal runaways that can jeopardize safety in large reactors. As global pharmaceutical companies increasingly mandate green manufacturing practices from their suppliers, adopting this technology provides a competitive advantage by aligning production capabilities with the sustainability goals of major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this basic ionic liquid catalyzed synthesis. These insights are derived directly from the experimental data and embodiments provided in the patent literature, ensuring that the information reflects the actual performance capabilities of the technology. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Can the basic ionic liquid catalyst be recycled in this synthesis process?
A: Yes, the patent data confirms that the filtrate containing the basic ionic liquid catalyst can be directly reused for subsequent batches without additional treatment. The catalyst maintains high activity for at least 7 cycles with negligible yield reduction.
Q: What are the optimal reaction conditions for maximizing yield?
A: The optimal conditions involve using a 95-98% aqueous methanol solution as the solvent, maintaining a reflux temperature for 22 to 36 minutes, and utilizing a catalyst loading of 6-8% relative to the thiochromanone substrate.
Q: Does this method support diverse substrate scopes for drug discovery?
A: Yes, the method is robust across various substituted thiochromanones, including chloro, fluoro, and methyl derivatives, consistently delivering yields between 84% and 94% for complex spiro-fused heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the basic ionic liquid one-pot method for producing high-quality spiro indole derivatives. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, commercial-scale manufacturing process. Our facilities are equipped to handle the specific requirements of ionic liquid chemistry, ensuring that the benefits of catalyst recycling and green solvent usage are fully realized in production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, ensuring full compliance with international regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener route for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your target molecules. Let us help you optimize your supply chain and reduce your manufacturing costs while contributing to a more sustainable pharmaceutical industry.
