Revolutionizing Tadalafil Intermediate Production: A Deep Dive into One-Pot Reductive Amination Technology
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, with Tadalafil remaining a cornerstone molecule due to its superior pharmacokinetic profile. A significant technological breakthrough in the manufacturing of this vital active pharmaceutical ingredient is detailed in patent CN110627786B, which discloses a highly efficient preparation method for a key tadalafil intermediate. This innovation addresses long-standing challenges in the industry by utilizing easily obtainable raw materials, specifically piperonyl acid acyl chloride, to achieve high-purity outcomes under mild reaction conditions. Unlike traditional multi-step syntheses that often suffer from low overall yields and complex purification requirements, this novel approach streamlines the entire synthesis process into what can be effectively regarded as a one-step reaction. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms. The ability to produce high-purity intermediates with reduced operational complexity directly translates to enhanced supply chain stability and reduced time-to-market for generic formulations. As we analyze the technical specifics, it becomes clear that this methodology offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that manufacturers can meet the rigorous quality standards demanded by global regulatory bodies while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tadalafil intermediates has been plagued by significant inefficiencies that hinder large-scale industrial production. Prior art methodologies predominantly rely on D-tryptophan methyl ester or its hydrochloride salt reacting with piperonal or its analogues through a series of cumbersome synthetic steps. These conventional routes are inherently problematic because they require the isolation of multiple different intermediates, each step introducing potential yield losses and impurity profiles that are difficult to manage. Specifically, the use of piperonal as a starting material presents logistical challenges, as it is not always easy to obtain as a tubular medicament, creating bottlenecks in the supply chain for reliable agrochemical intermediate supplier networks and pharma manufacturers. Furthermore, alternative routes disclosed in earlier literature, such as those involving the condensation with piperonyl acid acyl chloride followed by Lawesson's reagent treatment, suffer from severe drawbacks. These include the generation of various byproducts, unpleasant odors associated with sulfur-containing reagents, and the necessity for cryogenic conditions, such as reactions at -78°C, which are energy-intensive and operationally hazardous. The cumulative effect of these limitations is a process that is neither economically viable nor environmentally sustainable for modern high-volume manufacturing.
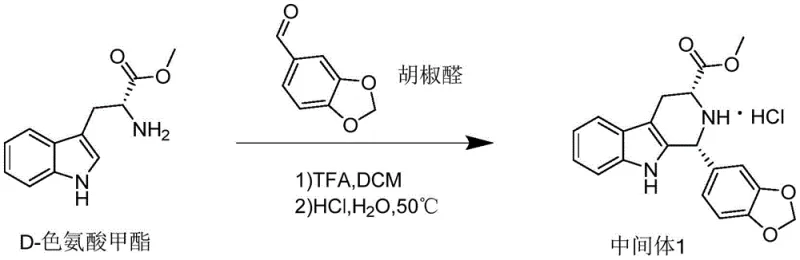
The Novel Approach
In stark contrast to the deficiencies of the prior art, the novel approach disclosed in the patent data offers a streamlined and robust alternative that fundamentally restructures the synthesis pathway. By employing piperonyl acid acyl chloride as the acylating agent in conjunction with D-tryptophan methyl ester hydrochloride, the process bypasses the need for controlled reagents like piperonal while simultaneously simplifying the reaction sequence. This method allows for the direct formation of the target intermediate through a cascade of reactions that occur within a single reaction vessel, effectively eliminating the need for intermediate isolation and purification steps that typically drain resources and time. The reaction conditions are notably mild, operating effectively at temperatures between 30°C and 45°C, which drastically reduces energy consumption compared to the cryogenic requirements of older methods. Moreover, the use of trifluoroacetic acid as a catalytic promoter instead of stoichiometric amounts of harsh Lewis acids minimizes waste generation and simplifies the workup procedure. This technological leap not only enhances the overall yield, achieving figures as high as 86.95% in optimized examples, but also ensures a purity profile that meets stringent pharmaceutical specifications. For procurement managers, this translates to cost reduction in pharmaceutical intermediates manufacturing by reducing raw material waste and processing time, thereby securing a more reliable supply of high-purity tadalafil intermediates.
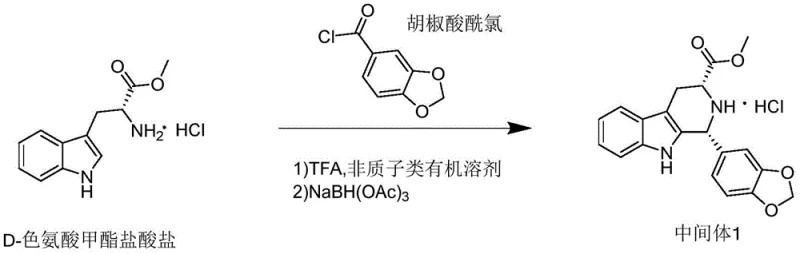
Mechanistic Insights into TFA-Catalyzed Reductive Amination
The core of this technological advancement lies in the intricate mechanistic pathway that facilitates the transformation of simple starting materials into the complex tadalafil intermediate with high fidelity. The reaction mechanism initiates with a Friedel-Crafts acylation reaction between D-tryptophan methyl ester hydrochloride and piperonyl acid acyl chloride, catalyzed by a trace amount of the protonic acid trifluoroacetic acid. This initial step is critical as it activates the aromatic ring for nucleophilic attack, forming a ketone intermediate that is poised for subsequent transformation. Unlike traditional Lewis acid catalysts such as aluminum trichloride, which can generate hydroxide colloids that obscure reactant surfaces and complicate filtration, the protonic acid catalyst remains homogeneous and easy to manage. Following the acylation, the system undergoes a Schiff base reaction where the amine functionality condenses with the carbonyl group to form an unstable imine compound. This imine intermediate is transient and highly reactive, requiring immediate stabilization to prevent decomposition or side reactions. The addition of sodium triacetoxyborohydride serves as a selective reducing agent that targets the imine bond specifically, reducing it to the corresponding amine without affecting other sensitive functional groups within the molecule. This reductive amination step is the final key to locking in the stereochemistry and structural integrity of the intermediate, ensuring that the final product possesses the necessary chiral purity for downstream biological activity.
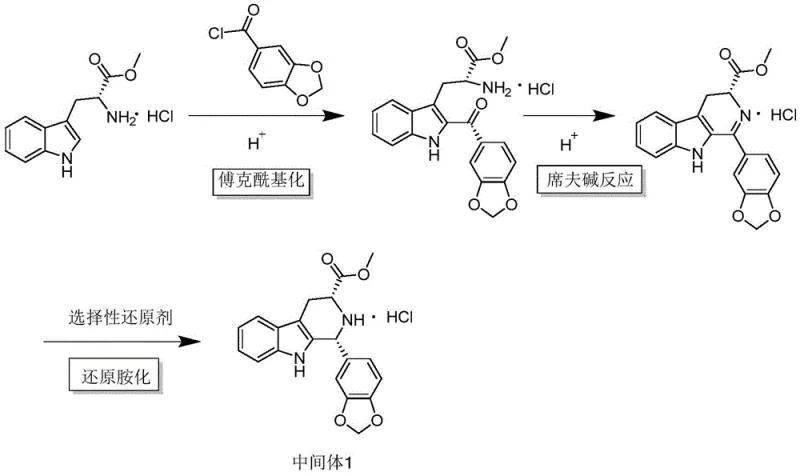
Controlling the impurity profile in this synthesis is paramount for meeting the rigorous standards expected of a high-purity OLED material or pharmaceutical intermediate supplier. The mechanism inherently suppresses the formation of common byproducts through the precise control of reaction parameters, particularly the mass ratio of the hydrochloride salt to the trifluoroacetic acid. Experimental data suggests that maintaining a ratio of 1:0.05 creates an optimal acidic environment that promotes the desired cyclization while minimizing the risk of over-acylation or polymerization. Furthermore, the choice of tetrahydrofuran as the aprotic organic solvent plays a crucial role in solvating the ionic species involved in the reaction, ensuring that the reactants remain in solution and available for collision. The stability of the imine compound is managed by the immediate addition of the reducing agent, which prevents the accumulation of this unstable species and drives the equilibrium towards the final product. This kinetic control is essential for achieving the reported purity levels of over 98%, as it prevents the formation of difficult-to-remove isomers or degradation products. For R&D teams, understanding these mechanistic nuances allows for better troubleshooting and optimization during the technology transfer phase, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds without unexpected deviations in quality.
How to Synthesize Tadalafil Intermediate Efficiently
Implementing this synthesis route in a production environment requires a strict adherence to the optimized parameters defined in the patent literature to ensure reproducibility and safety. The process begins with the dissolution of the starting materials in a dry, aprotic solvent under an inert atmosphere to prevent moisture-induced hydrolysis of the acid chloride. Detailed standardized synthesis steps are crucial for maintaining the delicate balance between reaction rate and selectivity, particularly during the addition of the reducing agent. Operators must monitor the reaction progress closely, typically using thin-layer chromatography, to determine the precise endpoint before quenching. The following guide outlines the critical operational phases required to achieve the high yields and purity reported in the technical disclosure.
- Dissolve D-tryptophan methyl ester hydrochloride and piperonyl acid chloride in an aprotic organic solvent such as tetrahydrofuran under nitrogen protection.
- Add a catalytic amount of trifluoroacetic acid and stir the mixture at 30-45°C for 8-12 hours to facilitate Friedel-Crafts acylation and Schiff base formation.
- Introduce sodium triacetoxyborohydride as a selective reducing agent and continue reacting for 2-4 hours to complete the reductive amination and isolate the intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply. The primary benefit stems from the drastic simplification of the manufacturing process, which eliminates multiple unit operations such as intermediate isolation, drying, and re-dissolution. This reduction in processing steps directly correlates to a significant reduction in labor costs, energy consumption, and equipment occupancy time, thereby enhancing the overall throughput of the manufacturing facility. Furthermore, the avoidance of expensive and hazardous reagents like Lawesson's reagent not only lowers the direct material costs but also reduces the burden on waste treatment systems, leading to substantial cost savings in environmental compliance. The use of readily available raw materials, such as piperonyl acid acyl chloride, mitigates the risk of supply chain disruptions that are often associated with controlled or specialty reagents. This reliability is crucial for maintaining consistent production schedules and meeting the demanding delivery timelines of global pharmaceutical clients. Additionally, the mild reaction conditions reduce the need for specialized cryogenic equipment, allowing for production in standard glass-lined or stainless steel reactors, which further lowers the capital expenditure required for technology adoption.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of synthetic steps from multiple stages to a one-pot process fundamentally alters the cost structure of the intermediate. By removing the need for expensive重金属 removal processes and complex purification protocols, manufacturers can achieve a leaner production model. The qualitative improvement in yield, driven by the suppression of byproducts, ensures that more raw material is converted into saleable product, maximizing the return on investment for every batch produced. This efficiency gain is compounded by the reduced solvent usage and shorter cycle times, which collectively drive down the variable cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are not subject to strict regulatory controls or limited availability is a key strategic advantage. The shift away from piperonal to piperonyl acid acyl chloride ensures a more robust supply chain, as the latter is a common chemical building block with multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and price volatility. Moreover, the simplified process is less prone to operational failures, ensuring that production batches are completed on schedule. This predictability is vital for supply chain heads who need to coordinate downstream formulation and packaging activities, reducing the need for safety stock and minimizing inventory holding costs.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is often hindered by safety and environmental concerns, but this method addresses both proactively. The absence of malodorous sulfur reagents and the use of mild temperatures make the process safer for operators and easier to contain within standard ventilation systems. Waste streams are less hazardous, simplifying the treatment process and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturer, making it a more attractive partner for environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and strategic value of the method. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: Why is piperonyl acid chloride preferred over piperonal in this synthesis route?
A: Piperonyl acid chloride avoids the use of controlled reagents like piperonal which can be difficult to source as a tubular medicament. Furthermore, the acyl chloride route enables a one-pot reaction mechanism that significantly reduces processing steps compared to traditional multi-step condensation methods.
Q: What are the critical reaction conditions for maximizing yield in this process?
A: The patent data indicates that maintaining a reaction temperature of 30°C and using tetrahydrofuran as the solvent yields the highest purity and conversion rates. Additionally, the mass ratio of D-tryptophan methyl ester hydrochloride to trifluoroacetic acid should be strictly controlled around 1:0.05 to prevent byproduct formation.
Q: How does this method improve environmental compliance compared to prior art?
A: This method eliminates the need for unpleasant and hazardous reagents like Lawson's reagent used in older synthetic routes. By utilizing mild reaction conditions and avoiding heavy metal catalysts, the process generates less hazardous waste and simplifies the post-treatment purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot reductive amination process are translated into reliable industrial reality. We are committed to delivering high-purity tadalafil intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify every batch. Our capability to manage complex chemical transformations allows us to offer flexible manufacturing solutions that adapt to the specific needs of our clients, whether they require custom synthesis or large-volume supply agreements.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with the highest international standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
