Advanced Synthesis of N,N-Dibutyl M-Aminophenol for High-Performance ODB-2 Manufacturing
The global demand for high-performance pressure-sensitive recording materials continues to drive innovation in the synthesis of key chemical intermediates, specifically within the realm of dye chemistry. Patent CN108558710B introduces a transformative preparation method for N,N-dibutyl m-aminophenol, a critical precursor for the widely used black color former ODB-2. This compound is essential for producing thermal paper used in faxes, labels, and high-speed recording applications, where storage stability and deep dyeing properties are paramount. The traditional supply chain has long struggled with the inefficiencies of conventional synthesis routes, which often result in suboptimal yields and complex purification challenges. By leveraging the novel approach detailed in this patent, manufacturers can access a more robust and economically viable pathway to produce high-purity intermediates. This technical breakthrough not only addresses the immediate need for reliable dye intermediate suppliers but also sets a new benchmark for process efficiency in the fine chemicals sector. The structural integrity of the final ODB-2 product is directly dependent on the quality of this aminophenol derivative, making the optimization of its synthesis a strategic priority for R&D teams globally.
The limitations of conventional methods for synthesizing N,N-dibutyl m-aminophenol have historically posed significant barriers to efficient commercial scale-up of complex dye intermediates. Traditional processes typically involve an initial alkali fusion and acid-out reaction on sodium meta-aminophenol to obtain meta-aminophenol, followed by a separate alkylation step. This sequence is fraught with chemical inefficiencies, as the phenolic hydroxyl group in the intermediate meta-aminophenol exhibits considerable reactivity that interferes with the desired alkylation. Consequently, this leads to the formation of ether byproducts which consume valuable alkylating reagents and complicate the downstream purification landscape. The selectivity of the reaction is often poor, resulting in a target product conversion rate that hovers around 45 percent, necessitating extensive and costly post-treatment procedures to isolate the desired compound. Furthermore, the overall process flow generates a large amount of wastewater, creating substantial environmental compliance burdens for manufacturing facilities. These inherent flaws in the legacy technology underscore the urgent need for a redesigned synthetic strategy that can bypass these reactivity issues.
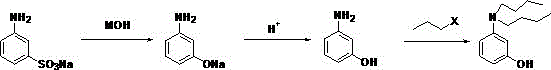
The novel approach presented in the patent fundamentally reorders the synthetic sequence to overcome the reactivity challenges associated with the phenolic hydroxyl group. Instead of alkylating the phenol directly, the process initiates with an alkylation reaction on sodium m-aminobenzenesulfonate, a starting material where the amino group is more nucleophilic and the sulfonate group provides stability. This strategic shift ensures that the alkylation occurs selectively on the nitrogen atom without interference from oxygen-based side reactions, thereby drastically simplifying the impurity profile. The reaction conditions are optimized to operate between 80-200°C for 2-5 hours, allowing for complete conversion while maintaining control over the reaction kinetics. Following alkylation, the intermediate undergoes an alkali fusion reaction at elevated temperatures of 200-350°C to convert the sulfonate group into the desired phenolic structure. This inversion of steps effectively eliminates the formation of ether byproducts that plague the traditional method, leading to a much cleaner reaction mixture. The result is a process that not only improves the yield significantly but also reduces the types of byproducts, lightening the burden of post-treatment and enhancing the overall economic feasibility of the manufacturing operation.
Mechanistic Insights into Alkylation and Alkali Fusion Sequence
A deep mechanistic analysis of this patented route reveals why the stability of the sulfonate group is crucial for achieving high selectivity during the alkylation phase. In the traditional pathway, the free phenolic hydroxyl group acts as a competing nucleophile, attacking the halogenated hydrocarbon to form unwanted ether linkages that are difficult to separate from the target amine. By protecting the phenolic functionality in the form of a sulfonate salt during the initial alkylation, the new method effectively masks this reactive site until the appropriate stage of the synthesis. The alkylation reaction utilizes halogenated hydrocarbons such as chlorobutane or bromobutane in the presence of an acid-binding agent like sodium hydroxide or magnesium oxide. The molar ratios are carefully controlled, typically ranging from 1:2 to 1:4 for the sulfonate to halogenated hydrocarbon, ensuring excess alkylating agent drives the reaction to completion without excessive waste. This precise stoichiometric control, combined with the inherent electronic properties of the sulfonate substrate, ensures that the nitrogen atom is the exclusive site of alkylation. Such mechanistic precision is vital for R&D directors focused on purity and impurity profiles, as it minimizes the generation of structurally similar contaminants that could compromise the performance of the final ODB-2 color former.
The subsequent alkali fusion and acid precipitation steps are engineered to maximize recovery while maintaining stringent purity specifications throughout the production cycle. During the alkali fusion stage, the alkylated sulfonate is subjected to high temperatures in the presence of strong alkaline reagents like potassium hydroxide or sodium hydroxide. This harsh environment is necessary to cleave the sulfonate group and generate the alkylated sodium phenolate intermediate, which is then carefully neutralized. The acid precipitation reaction is conducted under controlled pH conditions, typically adjusting the solution to a pH of 2-7 to precipitate the crude N,N-dibutyl m-aminophenol. This step is critical for removing inorganic salts and residual alkaline materials from the organic product phase. The refining process further includes water washing and heating dehydration at temperatures between 100-250°C to remove trace moisture and volatile impurities. Gas Chromatography analysis of the final product confirms purity levels exceeding 98 percent, demonstrating the efficacy of this purification strategy. For procurement managers, this level of consistency translates to reduced risk of batch rejection and more predictable production scheduling for downstream applications.
How to Synthesize N,N-Dibutyl M-Aminophenol Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters and safety protocols to ensure optimal results in a commercial setting. The process begins with the dissolution of sodium m-aminobenzenesulfonate in a solvent, preferably water, to create a homogeneous reaction medium before the addition of alkylating agents. Detailed standardized synthetic steps are essential for replicating the high yields reported in the patent data, particularly regarding temperature control and mixing efficiency during the exothermic alkylation phase. Operators must monitor the reaction progress using HPLC or GC to determine the endpoint accurately, ensuring that raw material consumption is minimized before proceeding to the high-temperature fusion stage. The transition between steps, especially the cooling and layering phases, requires precise handling to prevent emulsification or product loss. Adhering to these operational guidelines allows manufacturing teams to leverage the full potential of this innovative chemistry.
- Conduct alkylation reaction on sodium m-aminobenzenesulfonate with halogenated hydrocarbon at 80-200°C for 2-5 hours.
- Perform alkali fusion reaction on the alkylate solution with alkaline reagent at 200-350°C for 1-3 hours.
- Carry out acid precipitation reaction on alkylated sodium phenolate to obtain crude product.
- Refine the crude product through water washing, dehydration, and drying to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this patented synthesis method offers profound commercial advantages that extend beyond mere technical superiority, addressing critical pain points in cost management and supply chain reliability for fine chemical manufacturing. By eliminating the formation of complex ether byproducts, the process significantly reduces the volume of waste streams that require treatment, leading to substantial cost savings in environmental compliance and waste disposal operations. The simplified post-treatment workflow means that less energy and fewer solvents are consumed during the purification stages, directly contributing to a lower cost of goods sold. For procurement managers evaluating cost reduction in dye intermediates manufacturing, this efficiency gain is a compelling factor that enhances the overall competitiveness of the supply chain. Furthermore, the use of water as a primary solvent aligns with green chemistry principles, reducing the reliance on volatile organic compounds and mitigating safety risks associated with flammable solvents. These operational improvements collectively create a more resilient and sustainable production model that can withstand market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The reversal of the synthetic sequence eliminates the need for expensive purification steps required to remove ether byproducts generated in traditional methods. By improving the reaction selectivity, the consumption of raw materials such as halogenated hydrocarbons is optimized, ensuring that a higher proportion of inputs are converted into saleable product. This increase in atomic efficiency directly lowers the variable cost per kilogram of the final intermediate, providing a clear margin advantage for manufacturers. Additionally, the ability to recycle waste acid and alkali generated during the process further enhances the economic viability of the operation. These factors combine to deliver significant financial benefits without compromising on the quality or performance of the chemical output.
- Enhanced Supply Chain Reliability: The robustness of this new synthetic route ensures a more consistent output of high-purity material, reducing the incidence of off-spec batches that can disrupt downstream production schedules. With a yield improvement from approximately 45 percent to over 65 percent, the effective capacity of existing manufacturing assets is increased without the need for capital expansion. This boost in throughput capability allows suppliers to meet growing demand for ODB-2 precursors more effectively, reducing lead time for high-purity dye intermediates. The simplicity of the process also makes it easier to scale up from pilot plants to full commercial production, ensuring that supply can be ramped up quickly in response to market needs. Such reliability is crucial for supply chain heads who prioritize continuity and risk mitigation in their sourcing strategies.
- Scalability and Environmental Compliance: The process design inherently supports large-scale production due to the use of common industrial reagents and straightforward unit operations like filtration and distillation. The reduction in wastewater discharge and the ability to treat waste streams more easily lighten the regulatory burden on manufacturing facilities, ensuring long-term operational sustainability. This alignment with environmental standards minimizes the risk of production shutdowns due to compliance issues, securing the supply line for customers. Moreover, the lower energy requirements for purification contribute to a reduced carbon footprint, appealing to end-users who are increasingly focused on the sustainability credentials of their supply chain. These attributes make the technology highly attractive for long-term partnerships focused on responsible chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced preparation method for N,N-dibutyl m-aminophenol. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and stakeholders. Understanding these details is essential for making informed decisions about adopting this technology for commercial production. The answers provided reflect the specific improvements in yield, purity, and process efficiency documented in the intellectual property.
Q: How does the new synthesis route improve yield compared to traditional methods?
A: The patented method reverses the reaction order, alkylating the sulfonate first, which increases yield from approximately 45% to over 65% by reducing byproduct formation.
Q: What are the purity specifications achievable with this process?
A: Through optimized refining steps including water washing and controlled dehydration, the process consistently achieves GC purity levels of 98% or higher.
Q: Does this method reduce environmental waste in manufacturing?
A: Yes, the use of water as a primary solvent and the reduction of complex byproducts significantly lighten the burden of post-treatment and wastewater discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dibutyl M-Aminophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of N,N-dibutyl m-aminophenol meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthesis routes effectively, providing you with a dependable source for your dye intermediate needs. By partnering with us, you gain access to a supply chain that prioritizes quality, reliability, and continuous improvement.
We invite you to contact our technical procurement team to discuss how our capabilities can support your specific project requirements and drive value for your organization. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized supply solutions. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to transparency and partnership. Let us collaborate to enhance your production efficiency and secure your supply chain for the future.
