Synthesizing High-Purity Cholic Acid Intermediate A3 for Commercial Pharmaceutical Applications
Synthesizing High-Purity Cholic Acid Intermediate A3 for Commercial Pharmaceutical Applications
The pharmaceutical industry is increasingly shifting towards fully synthetic routes for critical bile acid derivatives to mitigate biological safety risks. Patent CN115651049A introduces a robust chemical synthesis method for producing Cholic Acid Intermediate A3 and ultimately Cholic Acid (CA), addressing the significant viral contamination risks inherent in traditional animal organ extraction. This technology provides a scalable, virus-free pathway that ensures consistent quality and supply security for global pharmaceutical manufacturers. By leveraging advanced organic transformations such as Wittig olefination and selective oxidations, this process establishes a new standard for reliable cholic acid intermediate supplier capabilities in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the commercial production of cholic acid has relied heavily on the extraction and refinement of internal organs from cattle or sheep. This biological sourcing method presents unavoidable challenges, primarily the potential risk of virus infection which poses a severe threat to patient safety and regulatory compliance. Furthermore, reliance on animal by-products creates significant volatility in the supply chain, where availability is dictated by slaughterhouse outputs rather than market demand. The purification of natural extracts often requires extensive downstream processing to remove impurities, leading to variable batch-to-batch consistency and complicating the validation processes required for high-purity cholic acid intermediate usage in drug formulations.
The Novel Approach
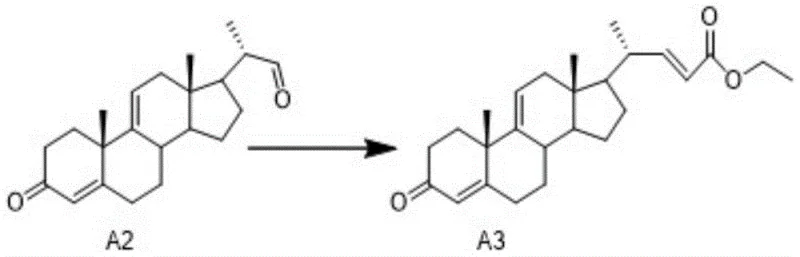
The patented methodology offers a transformative solution by establishing a complete chemical synthesis route starting from readily available precursors like 9α-hydroxy BA. Instead of extraction, the process utilizes precise organic reactions, beginning with the conversion of Compound A2 to Intermediate A3 via a Wittig or Wittig-Horner reaction. This synthetic strategy allows for exact control over molecular structure and stereochemistry, effectively eliminating biological contaminants. The mild reaction conditions described in the patent facilitate easier handling and safer operations, directly contributing to cost reduction in pharmaceutical intermediates manufacturing by streamlining purification steps and reducing the need for complex viral clearance protocols associated with biological materials.
Mechanistic Insights into Wittig and Wittig-Horner Olefination
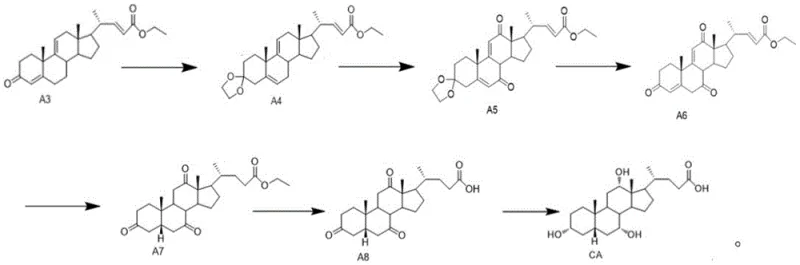
The core innovation of this technology lies in the efficient construction of the side chain at the C17 position of the steroid nucleus. The synthesis of Intermediate A3 is achieved through a Wittig reaction where Compound A2 reacts with ethoxycarbonyl methylene triphenylphosphine in solvents like toluene or tetrahydrofuran under reflux. Alternatively, a Wittig-Horner variation employs triethyl phosphonoacetate with bases such as sodium hydride or potassium tert-butoxide at moderate temperatures ranging from 20°C to 66°C. These reactions proceed through the formation of a phosphorus ylide or phosphonate carbanion, which attacks the aldehyde group of Compound A2 to form the desired alpha,beta-unsaturated ester with high stereoselectivity.
Following the formation of Intermediate A3, the pathway involves a sophisticated sequence of functional group manipulations to install the necessary hydroxyl groups with correct stereochemistry. The process includes ketal protection of the C3 ketone, followed by oxidation at the C7 and C12 positions using chromium-based oxidants or pyridinium chlorochromate in the presence of N-hydroxyphthalimide. Subsequent hydrolysis removes the protecting groups, and a critical hydrogenation step using palladium on carbon in an ethanol-pyridine system ensures the formation of the 5α-H isomer with high purity. This meticulous control over oxidation states and stereochemistry is essential for producing bioactive bile acids that meet stringent pharmacopeial standards.
How to Synthesize Cholic Acid Intermediate A3 Efficiently
The synthesis of Cholic Acid Intermediate A3 requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the aldehyde precursor (Compound A2) via oxidation and elimination reactions, followed by the key carbon-carbon bond-forming step. Operators must maintain strict temperature controls during the Wittig reaction, typically refluxing in toluene for 10 to 16 hours or reacting in THF at 20-66°C for shorter durations. Post-reaction workup involves specialized precipitation steps using zinc chloride solutions to remove triphenylphosphine oxide byproducts, ensuring the isolation of a high-quality intermediate suitable for downstream processing.
- Oxidize 9α-hydroxy BA (SM) to Compound A1 using TEMPO/NaClO or Jones reagent.
- Perform elimination on A1 to obtain Compound A2, followed by a Wittig or Wittig-Horner reaction to synthesize Intermediate A3.
- Convert A3 to Cholic Acid via ketal protection, oxidation, hydrolysis, hydrogenation, and final selective reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the transition to this synthetic route offers substantial strategic benefits beyond mere technical feasibility. By decoupling production from the fluctuations of the livestock industry, manufacturers can guarantee a stable and continuous supply of critical intermediates regardless of seasonal or agricultural disruptions. The ability to produce these compounds in a controlled chemical environment significantly reduces the lead time associated with sourcing raw biological materials and performing extensive safety testing for viral pathogens. This reliability is paramount for maintaining uninterrupted production schedules for downstream API manufacturing.
- Cost Reduction in Manufacturing: The synthetic route eliminates the expensive and logistically complex steps required for viral clearance and safety validation of animal-derived products. By using standard chemical reagents and solvents, the process leverages existing infrastructure in fine chemical plants, avoiding the need for specialized biological containment facilities. The removal of transition metal catalysts in certain steps and the use of recyclable solvents further contribute to operational efficiency, driving down the overall cost of goods sold without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing intermediates from a synthetic pathway mitigates the risks associated with animal disease outbreaks or regulatory bans on animal parts, which can abruptly halt traditional supply lines. The raw materials for this synthesis, such as phosphorus reagents and steroid precursors, are commercially available from multiple global suppliers, creating a resilient supply network. This diversification ensures that procurement teams can secure long-term contracts with confidence, knowing that production capacity can be scaled up rapidly to meet surging market demand for bile acid therapeutics.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as moderate temperatures and atmospheric pressure for many steps, are inherently safer and easier to scale from pilot plant to commercial tonnage. The process design incorporates efficient workup procedures, such as aqueous precipitation and filtration, which minimize solvent waste and simplify effluent treatment. This alignment with green chemistry principles not only reduces environmental impact but also ensures compliance with increasingly strict global regulations regarding industrial emissions and waste disposal in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is synthetic Cholic Acid preferred over animal extraction?
A: Synthetic routes eliminate the risk of virus infection associated with extracting bile acids from animal viscera, ensuring higher safety standards for pharmaceutical applications.
Q: What is the key reaction for forming Intermediate A3?
A: The core transformation involves a Wittig reaction or Wittig-Horner reaction between Compound A2 and a phosphorus ylide or phosphonate reagent to extend the side chain.
Q: How is stereochemical purity controlled in this process?
A: The process utilizes specific hydrogenation conditions with palladium catalysts and solvents like pyridine/ethanol to achieve high 5α-H isomer purity in Intermediate A7.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Intermediate A3 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to secure the future of pharmaceutical supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex steroid syntheses like the one described in CN115651049A can be successfully transferred to industrial scale. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of intermediate meets the highest international standards, providing our partners with the confidence needed for regulatory filings and commercial launch.
We invite you to collaborate with us to leverage this innovative technology for your bile acid projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our synthetic capabilities can enhance your product portfolio and optimize your manufacturing costs.
