Advanced Synthetic Route for Cholic Acid Intermediate A5 and Commercial Scalability
The pharmaceutical industry is currently witnessing a significant paradigm shift in the production of bile acid derivatives, driven by the urgent need for safer and more sustainable supply chains. Patent CN115626944A introduces a groundbreaking chemical synthesis method for producing Cholic Acid Intermediate A5, a critical precursor in the manufacture of Cholic Acid (CA). This innovation addresses the longstanding limitations of traditional extraction methods, which rely heavily on animal sources and carry inherent biological risks. By leveraging a robust oxidation strategy involving chromium-based reagents and N-hydroxyphthalimide, this technology enables the production of high-purity intermediates from plant-derived starting materials. The structural integrity and stereochemistry of the final Cholic Acid molecule, as depicted below, are meticulously preserved throughout this multi-step synthetic sequence, ensuring therapeutic efficacy.
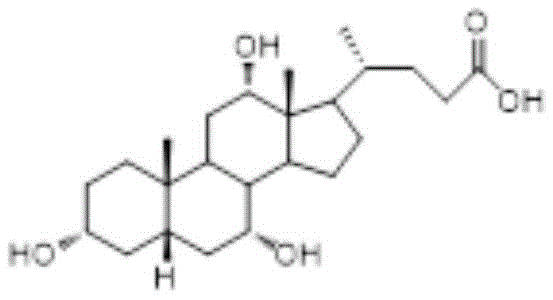
Furthermore, the transition from biological extraction to total chemical synthesis represents a major milestone for regulatory compliance and supply chain stability. The ability to synthesize complex steroid skeletons like Cholic Acid from common plant sources mitigates the volatility associated with animal by-product availability. This patent not only details the preparation of Intermediate A5 but also outlines a comprehensive downstream pathway to the final API, demonstrating a fully integrated solution for manufacturers. For global procurement teams, this signals a move towards a more reliable pharma intermediate supplier landscape, where quality is defined by chemical precision rather than biological variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial production of Cholic Acid has been dominated by extraction and refinement processes utilizing the internal organs of cattle or sheep. While this method has served the market for decades, it is fraught with significant drawbacks that modern pharmaceutical standards can no longer tolerate. The primary concern is the inevitable risk of virus infection and prion transmission, such as BSE (Bovine Spongiform Encephalopathy), which poses a severe public health threat and complicates regulatory approval in stringent markets. Additionally, the supply of animal viscera is subject to seasonal fluctuations, agricultural diseases, and changing slaughterhouse regulations, leading to unpredictable pricing and inconsistent raw material quality. From a processing standpoint, extracting pure bile acids from complex biological matrices requires extensive purification steps to remove proteins, lipids, and other biological contaminants, resulting in lower overall yields and higher waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN115626944A offers a sophisticated chemical synthesis route that bypasses biological sources entirely. The core of this innovation lies in the efficient conversion of Compound A4 to Intermediate A5 through a controlled oxidation reaction. By employing a dual-oxidant system comprising chromium oxide (or pyridinium chlorochromate) and N-hydroxyphthalimide in solvents like acetone or acetonitrile, the process achieves high selectivity under mild conditions (20-25°C). This approach not only eliminates the biosafety risks associated with animal tissues but also provides a consistent, reproducible chemical profile that is essential for GMP manufacturing. The use of plant-source compounds as the ultimate origin of the carbon skeleton ensures a sustainable and ethically sound supply chain, aligning with the growing demand for green chemistry in the fine chemical intermediates sector.
Mechanistic Insights into Chromium-Mediated Oxidation with NHPI
The transformation of Compound A4 into Cholic Acid Intermediate A5 is a pivotal step that defines the success of the entire synthetic route. This reaction involves the selective oxidation of specific positions on the steroid nucleus while preserving the sensitive ketal protection groups and the unsaturated side chain. The mechanism relies on the synergistic action of a primary oxidant, such as chromium oxide or PCC, and a radical mediator, N-hydroxyphthalimide (NHPI). The NHPI generates phthalimide-N-oxyl (PINO) radicals under the reaction conditions, which facilitate hydrogen abstraction from the substrate, thereby lowering the activation energy required for the chromium species to effect the oxidation. This cooperative catalysis allows the reaction to proceed efficiently at ambient temperatures, minimizing thermal degradation and epimerization risks that are common in harsher oxidative environments.
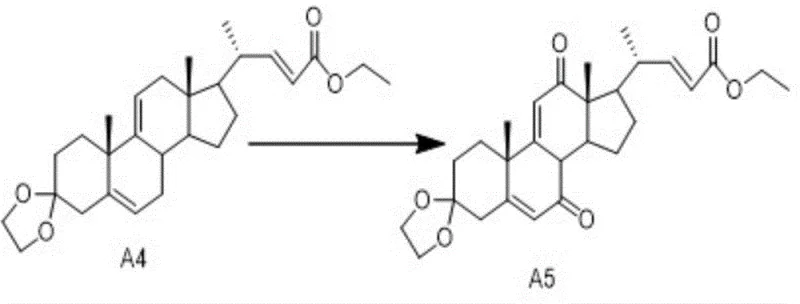
Impurity control is rigorously managed through the specific stoichiometry and addition protocol described in the patent. The oxidant is added in two portions: an initial charge to initiate the reaction followed by a second addition after a preset time (3 to 5 hours) to drive the conversion to completion over a total period of 20 to 25 hours. This staged addition prevents the accumulation of excessive oxidizing power that could lead to over-oxidation or cleavage of the steroid rings. Furthermore, the quenching step using isopropanol effectively neutralizes residual chromium species, simplifying the downstream workup. The result is a crude product with a superior impurity profile, reducing the burden on subsequent purification steps and enhancing the overall yield of the high-purity pharmaceutical intermediate.
How to Synthesize Cholic Acid Intermediate A5 Efficiently
The synthesis of Cholic Acid Intermediate A5 requires precise control over reaction parameters to ensure optimal yield and stereochemical purity. The process begins with the dissolution of the protected steroid precursor (Compound A4) in a polar aprotic solvent, followed by the careful introduction of the oxidant system. Maintaining the temperature within the narrow window of 20-25°C is critical to balance reaction rate and selectivity. The following guide summarizes the key operational phases derived from the patent examples, providing a framework for process chemists to implement this technology in a pilot or production setting.
- Dissolve compound A4 in acetone or acetonitrile and add N-hydroxyphthalimide (NHPI) along with a portion of the chromium oxide or PCC oxidant.
- Maintain the reaction temperature between 20-25°C with stirring for 3 to 5 hours to initiate the oxidation process gently.
- Add the remaining portion of the oxidant and continue stirring for 20 to 25 hours until TLC confirms completion, then quench with isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. The shift from animal extraction to plant-based chemical synthesis fundamentally alters the cost structure and risk profile of Cholic Acid production. By decoupling the supply of this critical intermediate from the livestock industry, manufacturers gain immunity from agricultural shocks and biological contamination scandals. This stability translates directly into more predictable long-term contracts and reduced inventory buffering costs. Moreover, the chemical consistency of the synthetic product reduces the variability in downstream processing, leading to more efficient resource utilization and lower operational expenditures across the value chain.
- Cost Reduction in Manufacturing: The elimination of expensive biological raw materials and the associated complex extraction protocols leads to substantial cost savings. The synthetic route utilizes commodity chemicals and solvents that are readily available in the global market, avoiding the price volatility of specialized animal by-products. Additionally, the mild reaction conditions reduce energy consumption compared to processes requiring extreme temperatures or pressures. The streamlined purification workflow, facilitated by the high selectivity of the oxidation step, further decreases the cost of goods sold by minimizing solvent usage and waste disposal fees.
- Enhanced Supply Chain Reliability: Relying on plant-source starting materials ensures a continuous and scalable feedstock supply that is not subject to the seasonal or regulatory constraints of animal slaughterhouses. This reliability is crucial for maintaining uninterrupted production schedules for downstream APIs. The synthetic nature of the intermediate also simplifies quality assurance processes, as the absence of biological contaminants reduces the testing burden and accelerates batch release times. This robustness makes the supply chain more resilient to external disruptions, ensuring that customers receive their orders on time without compromise.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and common organic solvents that are easily handled in large-scale facilities. The mild operating conditions enhance safety profiles, reducing the need for specialized high-pressure or cryogenic equipment. From an environmental perspective, the synthetic route offers a cleaner alternative to animal extraction, generating less biological waste and allowing for more effective solvent recovery and recycling. This alignment with green chemistry principles supports corporate sustainability goals and facilitates compliance with increasingly stringent environmental regulations in major pharmaceutical markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cholic Acid Intermediate A5. These answers are derived directly from the experimental data and beneficial effects reported in patent CN115626944A, providing clarity on the practical implementation and advantages of this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this synthetic route over animal extraction?
A: The synthetic route described in CN115626944A utilizes common plant-source compounds as raw materials, completely eliminating the risk of virus infection associated with extracting cholic acid from animal viscera.
Q: How does the use of N-hydroxyphthalimide impact the reaction efficiency?
A: N-hydroxyphthalimide acts as a co-oxidant that facilitates the selective oxidation of the steroid backbone under mild temperatures (20-25°C), reducing side reactions and improving the overall purity profile of intermediate A5.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process employs standard solvents like acetone and acetonitrile and operates at near-ambient temperatures, making it highly scalable and compatible with existing pharmaceutical manufacturing infrastructure without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Intermediate A5 Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of secure and high-quality supply chains for critical pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Cholic Acid Intermediate A5 with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to manage complex steroid syntheses positions us as a trusted partner for global pharmaceutical companies seeking to diversify their sourcing strategies.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can optimize your production costs and mitigate supply risks. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to our plant-based synthetic intermediates. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a collaborative approach to advancing your drug development pipeline.
