Advanced Enzymatic Synthesis of Argatroban Impurity G for Global Pharmaceutical Quality Control
The pharmaceutical industry relies heavily on the availability of high-quality reference standards to ensure the safety and efficacy of active pharmaceutical ingredients (APIs) throughout their lifecycle. A prime example of this critical need is found in the quality control of Argatroban, a potent direct thrombin inhibitor used clinically as an anticoagulant. To guarantee patient safety, regulatory bodies mandate the rigorous identification and quantification of degradation products, specifically Argatroban Impurity G, which can form during synthesis and storage. The recent technological breakthrough detailed in patent CN112625086A offers a transformative approach to producing this vital reference standard. Unlike previous methods that struggled with selectivity and yield, this new protocol leverages biocatalytic precision to deliver Argatroban Impurity G with exceptional purity levels exceeding 98 percent. For R&D directors and procurement specialists alike, this development signals a shift towards more sustainable, efficient, and reliable supply chains for complex pharmaceutical intermediates. By adopting this enzymatic route, manufacturers can overcome the historical bottlenecks associated with generating high-purity degradation impurities, ensuring that QC laboratories have consistent access to the materials they need for accurate HPLC and MS analysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Argatroban Impurity G has been plagued by significant chemical challenges that hindered both yield and purity. Traditional synthetic routes, such as those disclosed in earlier patents like CN201410254533.2, relied heavily on harsh chemical hydrolysis using strong alkalis such as barium hydroxide, sodium hydroxide, or potassium hydroxide. These reactions typically required elevated temperatures ranging from 60°C to 100°C to drive the conversion. However, the molecular structure of Argatroban contains multiple hydrolyzable functional groups, including amide, guanidyl, and sulfonamide moieties. Under such aggressive alkaline conditions, the reaction lacks specificity, leading to the simultaneous hydrolysis of these unintended groups. This non-selective degradation results in a complex mixture of by-products, including carbamide intermediate states, which makes the subsequent separation and purification processes extremely difficult and inefficient. Consequently, the overall yield of the target impurity was often abysmal, frequently hovering around 10 percent to 14 percent, while the purity rarely exceeded 93 percent, necessitating costly and time-consuming additional purification steps that further eroded process economics.
The Novel Approach
In stark contrast to the brute-force chemistry of the past, the novel approach outlined in patent CN112625086A introduces a sophisticated biocatalytic strategy that fundamentally changes the reaction landscape. This method utilizes immobilized arginase in the presence of manganese ions to catalyze the hydrolysis of Argatroban salts, specifically targeting the guanidino group with high specificity. By operating under mild physiological conditions, typically between 20°C and 40°C, the process avoids the thermal degradation and non-specific bond cleavage associated with strong alkalis. The use of Argatroban hydrochloride as the starting substrate further enhances solubility and reaction kinetics, allowing for a homogeneous reaction environment that maximizes enzyme-substrate interaction. This gentle yet highly effective transformation not only simplifies the downstream processing but also dramatically improves the material balance of the operation. The result is a streamlined workflow that produces Argatroban Impurity G with a purity of greater than 98 percent and yields that are multiples higher than conventional methods, representing a paradigm shift in how complex pharmaceutical impurities are manufactured for analytical purposes.
Mechanistic Insights into Arginase-Catalyzed Hydrolysis
The core of this technological advancement lies in the exquisite selectivity of the arginase enzyme, which acts as a biological scalpel to cleave specific bonds within the Argatroban molecule. Arginase is a binuclear manganese metalloenzyme that naturally catalyzes the hydrolysis of L-arginine to L-ornithine and urea. In this synthetic application, the enzyme recognizes the guanidino functionality of the Argatroban side chain as a substrate analogue. The manganese ions (Mn2+) play a pivotal role as cofactors, coordinating with the enzyme's active site residues to activate a water molecule for nucleophilic attack on the guanidino carbon. This mechanism ensures that only the specific bond connecting the guanidino group is hydrolyzed, converting it into a terminal primary amino group to form the impurity structure, while leaving the sensitive amide and sulfonamide linkages completely intact. This level of chemoselectivity is virtually impossible to achieve with small-molecule chemical catalysts under similar mild conditions, highlighting the power of biocatalysis in fine chemical synthesis.
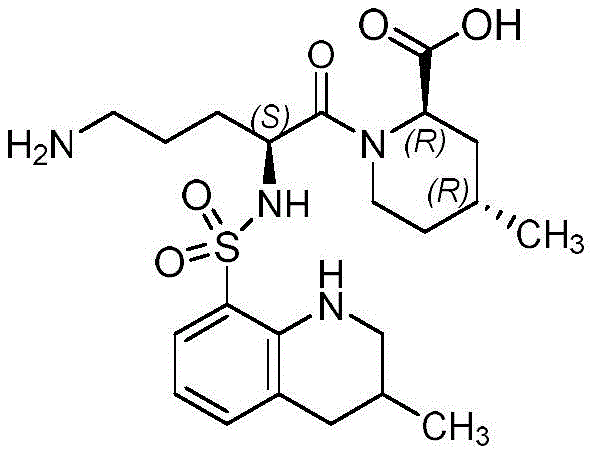
Furthermore, the control of impurities in this enzymatic process is inherently superior due to the absence of side reactions. In the traditional alkaline hydrolysis, the high pH and temperature create a chaotic environment where thermodynamic control is lost, leading to a broad spectrum of degradation products that co-elute during chromatography. Conversely, the enzymatic pathway is kinetically controlled and highly specific, meaning that the formation of by-products is minimized at the source. The patent data indicates that by optimizing the mass ratio of Argatroban to arginase (preferably 1:0.1 to 0.25) and maintaining the manganese ion concentration within a narrow window (1:0.01 to 0.02), the reaction proceeds cleanly to completion. The use of thin-layer chromatography (TLC) with a dichloromethane and methanol system allows for precise monitoring of the reaction endpoint, ensuring that the reaction is stopped exactly when the starting material is consumed, preventing any potential over-hydrolysis or enzyme-mediated degradation of the product itself. This precise control translates directly to the high purity (>98.65%) observed in the experimental examples, significantly reducing the burden on analytical teams to characterize and separate complex impurity profiles.
How to Synthesize Argatroban Impurity G Efficiently
Implementing this enzymatic synthesis route requires careful attention to reaction parameters to maximize the efficiency of the biocatalyst and ensure reproducible results. The process begins with the preparation of the Argatroban hydrochloride salt, which is crucial for solubility, followed by the addition of the immobilized enzyme and manganese cofactor under strictly controlled temperature conditions. The reaction is allowed to proceed with moderate stirring to ensure mass transfer without denaturing the enzyme, typically monitored via TLC until the starting material spot disappears. Following the reaction, the workup involves a clever desalting strategy using ammonium carbonate to liberate the free base, followed by extraction and crystallization. For a comprehensive, step-by-step technical guide including exact reagent quantities and safety precautions, please refer to the standardized synthesis protocol provided below.
- Prepare argatroban hydrochloride by adjusting the pH of an argatroban solution to 7-8 using hydrochloric acid, ensuring optimal solubility for the enzymatic reaction.
- Conduct the hydrolysis reaction by adding immobilized arginase and manganese chloride tetrahydrate to the solution, maintaining a temperature between 34-36°C for 12 hours.
- Isolate the final product by filtering the enzyme, concentrating the filtrate, desalting with ammonium carbonate, and extracting with ethyl acetate to achieve over 98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis method offers profound strategic advantages that extend far beyond simple technical metrics. The shift from harsh chemical hydrolysis to a biocatalytic process fundamentally alters the cost structure and risk profile of manufacturing Argatroban Impurity G. By eliminating the need for corrosive strong alkalis and high-temperature reflux conditions, the process significantly reduces energy consumption and the associated costs of heating and cooling large-scale reactors. Moreover, the simplicity of the workup procedure, which avoids complex chromatographic separations required by the messy traditional routes, translates into shorter batch cycle times and higher throughput. This efficiency gain allows suppliers to respond more rapidly to market demand fluctuations, ensuring a steady flow of high-quality reference standards to QC laboratories globally without the risk of production bottlenecks that often plague complex impurity synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this novel route are driven primarily by the drastic improvement in yield and the recyclability of the catalyst. Traditional methods suffered from yields as low as 10 percent, meaning that nearly 90 percent of the expensive starting material was wasted as unusable by-products. In contrast, the enzymatic method achieves yields exceeding 70 percent, effectively multiplying the output per kilogram of raw material input and substantially lowering the cost of goods sold. Additionally, the use of immobilized arginase allows the catalyst to be recovered via simple filtration and potentially reused in subsequent batches, further amortizing the cost of the biocatalyst over multiple production runs. The elimination of expensive and hazardous waste disposal costs associated with heavy metal catalysts or large volumes of strong alkali waste also contributes to a leaner, more cost-effective manufacturing model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and simplicity of the enzymatic process. The mild reaction conditions (34-36°C) reduce the stress on equipment, minimizing maintenance downtime and the risk of reactor failure compared to high-temperature alkaline processes. Furthermore, the high selectivity of the reaction ensures consistent product quality batch after batch, reducing the likelihood of failed QC releases that can disrupt supply schedules. The ability to produce high-purity material (>98%) directly from the reaction mixture with minimal purification steps means that lead times for order fulfillment can be drastically shortened. This reliability is critical for pharmaceutical companies that require just-in-time delivery of reference standards to support their own regulatory filings and routine quality testing, ensuring that their production lines never halt due to a lack of analytical controls.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this biocatalytic route represents a best-in-class solution for modern chemical manufacturing. The process operates in aqueous media at near-neutral pH, generating significantly less hazardous waste compared to the acidic and alkaline effluents of traditional methods. This simplifies wastewater treatment and helps manufacturers meet increasingly stringent environmental regulations without incurring excessive compliance costs. The use of immobilized enzymes is inherently scalable, as the technology is well-established in the industrial biotechnology sector for ton-scale productions. The straightforward workup involving crystallization and extraction is easily adaptable from liter-scale lab reactors to multi-hundred-liter pilot and production vessels, facilitating a smooth technology transfer. This scalability ensures that as the demand for Argatroban and its related impurities grows, the supply of Impurity G can be expanded seamlessly to meet global regulatory requirements.
Frequently Asked Questions (FAQ)
To assist our technical partners in evaluating this synthesis method for their specific applications, we have compiled a set of frequently asked questions based on the detailed patent data and our internal expertise. These questions address the critical technical differentiators of the enzymatic route, focusing on yield improvements, catalyst stability, and purity specifications. Understanding these nuances is essential for R&D teams planning to integrate this impurity standard into their analytical methods or for procurement teams assessing the long-term viability of this supply source. The answers provided below reflect the specific advantageous effects documented in the patent literature, offering a transparent view of the technology's capabilities.
Q: Why is the enzymatic method superior to traditional alkaline hydrolysis for Argatroban Impurity G?
A: Traditional methods use strong alkalis at high temperatures (60-100°C), which non-selectively hydrolyze multiple functional groups (amide, sulfonamide), leading to complex impurity profiles and low yields (approx. 10-14%). The enzymatic method utilizes arginase specificity to target only the guanidino group under mild conditions (35°C), resulting in significantly higher purity (>98%) and yields (over 70%).
Q: What represents the critical role of manganese ions in this synthesis?
A: Manganese ions (Mn2+) act as essential cofactors for the arginase enzyme. They stabilize the enzyme's active site and facilitate the coordination of the substrate, thereby maximizing catalytic efficiency. Without the precise addition of manganese ions, the hydrolysis rate would be insufficient for practical production timelines.
Q: How does this process impact the scalability of reference standard production?
A: The use of immobilized arginase allows for the catalyst to be recovered and reused via simple filtration, drastically reducing catalyst costs for large-scale batches. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature reactors, making the transition from lab-scale to commercial production seamless and safer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Argatroban Impurity G Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of pharmaceutical quality control depends on the availability of ultra-high purity reference standards. We have closely analyzed the advancements in enzymatic synthesis described in patent CN112625086A and are well-positioned to leverage this technology for our clients. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for initial method validation or routine annual testing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of Argatroban Impurity G we supply meets the >98% purity benchmark required for accurate HPLC and MS quantification.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to our enzymatically produced impurity standards can reduce your overall analytical costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing options tailored to your project needs. Let us be your partner in ensuring the highest standards of pharmaceutical quality and safety.
