Advanced Palladium-Catalyzed Synthesis of High-Purity Cis-Styryl Boron Derivatives for Pharmaceutical Applications
The landscape of organic synthesis is continually evolving with the introduction of more efficient and selective methodologies for constructing valuable building blocks. A significant advancement in this field is detailed in Chinese patent CN115710286A, which discloses a robust preparation method for cis-styryl boron derivatives. These compounds serve as critical intermediates in the synthesis of complex pharmaceutical agents and functional materials due to their unique electronic properties and versatile reactivity profiles. The patented process addresses longstanding challenges in stereoselectivity and operational simplicity, offering a pathway that combines high regioselectivity with mild reaction conditions. By leveraging a specialized palladium-catalyzed system, this technology enables the transformation of readily accessible halogenated aromatic compounds into high-value styrylboranes with exceptional precision. For research and development teams seeking reliable pharma intermediate supplier solutions, understanding the nuances of this synthetic route provides a strategic advantage in designing next-generation drug candidates and optimizing supply chains for complex organic molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing vinylboron species often suffer from significant drawbacks that hinder their widespread adoption in large-scale manufacturing environments. Many conventional protocols rely on harsh reaction conditions, such as extremely low temperatures or the use of highly reactive and hazardous reagents like organolithium or Grignard species, which necessitate stringent safety measures and specialized equipment. Furthermore, achieving high stereocontrol, particularly for the cis-isomer, has historically been a formidable challenge, often resulting in mixtures of geometric isomers that require difficult and yield-reducing separation processes. The lack of functional group tolerance in older methodologies also limits the scope of substrates that can be utilized, forcing chemists to employ extensive protecting group strategies that increase both the step count and the overall cost of production. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher costs for high-purity intermediates, which is a critical pain point for procurement managers aiming to reduce expenses in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach outlined in the patent utilizes a sophisticated palladium-catalyzed cross-coupling strategy that fundamentally reshapes the efficiency of styrylborane production. This method employs 7-oxabicyclo[2.2.1]hepta-2,5-diene-2,3-dicarboxylate as a key reagent alongside 1-pinacol-2-(1,8)naphthalene diamine diboronate to achieve remarkable regioselectivity and stereospecificity. The reaction proceeds under relatively mild thermal conditions, typically between 80°C and 120°C, eliminating the need for cryogenic setups and reducing energy consumption significantly. As illustrated in the specific example of synthesizing thiophene-substituted derivatives, the process demonstrates exceptional versatility and yield, proving its capability to handle diverse heteroaromatic systems effectively.
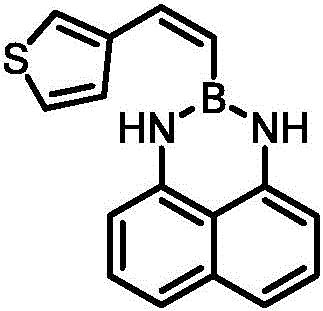
The use of common organic solvents like tetrahydrofuran and stable inorganic bases further simplifies the operational complexity, making this route highly attractive for commercial scale-up of complex polymer additives and pharmaceutical precursors. The streamlined workup procedure, involving simple extraction and chromatography, ensures that the final product meets stringent purity specifications without requiring elaborate purification steps, thereby enhancing the overall economic viability of the process for industrial applications.
Mechanistic Insights into Palladium-Catalyzed Cis-Selective Borylation
The core of this innovative synthesis lies in the intricate mechanistic pathway facilitated by the palladium catalyst and the specific choice of ligands and additives. The reaction initiates with the oxidative addition of the halogenated aromatic compound to the active palladium(0) species, forming an aryl-palladium(II) intermediate. This step is crucial as it sets the stage for the subsequent transmetallation and insertion events that define the stereochemical outcome of the product. The presence of water as an additive plays a pivotal role in modulating the reactivity of the boron species and facilitating the hydrolysis of the oxabicyclo intermediate, which is essential for generating the vinyl moiety with the desired cis-geometry. The phosphine ligand, such as triphenylphosphine, stabilizes the palladium center throughout the catalytic cycle, preventing aggregation and deactivation of the catalyst, which is a common issue in transition metal-catalyzed reactions.
Furthermore, the mechanism ensures excellent impurity control by favoring the formation of the cis-isomer through a concerted insertion-elimination sequence that minimizes the thermodynamic drive towards the more stable trans-isomer. This kinetic control is vital for maintaining high product quality and reducing the burden on downstream purification processes. The compatibility of the system with a wide range of substituents, including electron-withdrawing and electron-donating groups on the aromatic ring, underscores the robustness of the catalytic cycle. For R&D directors focused on purity and impurity profiles, this mechanistic understanding highlights the reliability of the process in delivering consistent batches of high-purity OLED material or API intermediates, free from problematic geometric isomers that could compromise biological activity or material performance in final applications.
How to Synthesize Cis-Styryl Boron Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and selectivity while ensuring safety and reproducibility. The general procedure involves charging a reaction vessel with the halogenated aromatic substrate, the oxabicyclo diester, and the diboron reagent in a suitable organic solvent under an inert nitrogen atmosphere. The addition of the palladium catalyst, phosphine ligand, base, and water must be performed in precise molar ratios to maintain the balance of the catalytic cycle. Heating the mixture to the optimal temperature range allows the transformation to proceed to completion within a practical timeframe. Following the reaction, standard workup techniques are employed to isolate the product, ensuring that the final material is ready for subsequent coupling reactions or direct application.
- Combine halogenated aromatic compounds, 7-oxabicyclo[2.2.1]hepta-2,5-diene-2,3-dicarboxylate, and 1-pinacol-2-(1,8)naphthalene diamine diboronate with a palladium catalyst and phosphine ligand in an organic solvent under nitrogen.
- Heat the reaction mixture to between 80°C and 120°C for 12 to 24 hours to facilitate the cross-coupling transformation.
- Perform workup by diluting with ethyl acetate, washing with water, drying the organic phase, and purifying via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the core concerns of procurement managers and supply chain heads regarding cost, reliability, and scalability. The reliance on commercially available and inexpensive starting materials, such as simple haloarenes and common boron reagents, drastically reduces the raw material costs associated with producing these valuable intermediates. The elimination of exotic or hazardous reagents not only lowers purchasing costs but also simplifies the regulatory compliance and safety protocols required for handling chemicals, leading to significant operational savings. Moreover, the mild reaction conditions and the use of standard solvents mean that existing manufacturing infrastructure can often be utilized without the need for costly capital investments in specialized cryogenic or high-pressure equipment.
- Cost Reduction in Manufacturing: The process achieves cost optimization through the use of earth-abundant palladium catalysts in low loadings and easily sourced ligands, which minimizes the expense of precious metals. The high atom economy and selectivity of the reaction reduce waste generation and the need for extensive purification, lowering the overall cost of goods sold. By avoiding multi-step protection and deprotection sequences often required in alternative routes, the synthesis becomes more direct and economically efficient, providing a clear pathway for cost reduction in electronic chemical manufacturing and other high-value sectors.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply chains for downstream customers. The broad substrate scope means that a single manufacturing platform can produce a diverse library of derivatives, reducing the risk of supply disruptions caused by the unavailability of specific niche precursors. This flexibility allows suppliers to respond rapidly to changing market demands and custom requests, enhancing the reliability of the supply chain for complex pharmaceutical intermediates and ensuring that production schedules are met without delay.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic extraction and filtration, facilitates easy scale-up from laboratory to pilot and commercial production scales. The reduced use of hazardous reagents and the generation of less toxic byproducts align with green chemistry principles, simplifying waste treatment and disposal processes. This environmental compatibility not only reduces the ecological footprint of the manufacturing process but also ensures compliance with increasingly stringent global environmental regulations, making it a sustainable choice for long-term production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this cis-styryl boron synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on reaction scope, conditions, and potential applications for industry stakeholders.
Q: What are the key advantages of this cis-styryl boron synthesis method?
A: This method offers excellent regioselectivity for constructing cis-styryl boron derivatives, utilizes readily available raw materials, and operates under mild reaction conditions with a short reaction time and simple post-treatment procedures.
Q: What catalyst system is employed in this preparation?
A: The process utilizes a palladium catalyst, specifically palladium acetate, in conjunction with a phosphine ligand such as triphenylphosphine, ensuring efficient catalytic turnover and high product yields.
Q: Is this method scalable for industrial production?
A: Yes, the use of common solvents like tetrahydrofuran, stable bases like potassium phosphate, and moderate temperatures makes this protocol highly suitable for commercial scale-up and industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Styryl Boron Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in driving innovation within the pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patents like CN115710286A can be successfully translated into reliable industrial supply. We are committed to meeting stringent purity specifications through our rigorous QC labs and advanced analytical capabilities, guaranteeing that every batch of cis-styryl boron derivatives we deliver meets the exacting standards required for drug discovery and material science applications. Our dedication to technical excellence allows us to navigate the complexities of organometallic chemistry with precision and consistency.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this methodology can optimize your budget and timeline. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability. Let us be your trusted partner in bringing high-performance chemical solutions to market efficiently and sustainably.
