Advanced Synthetic Strategy for Teriparatide Impurity F: Ensuring Quality Control in Peptide Manufacturing
The pharmaceutical industry faces continuous pressure to ensure the highest standards of safety and efficacy for biologic drugs, particularly complex peptides like Teriparatide, a vital treatment for osteoporosis. A critical aspect of this quality assurance involves the rigorous identification and control of process-related and degradation impurities. Patent CN112940103A introduces a groundbreaking synthetic method for Teriparatide Impurity F, a specific aspartimide derivative formed through the interaction of residues Asp30 and Val31. This technical breakthrough addresses a significant gap in the availability of high-purity reference standards, which are indispensable for validating analytical methods and ensuring batch-to-batch consistency in commercial manufacturing. By leveraging advanced solid-phase peptide synthesis (SPPS) techniques coupled with a novel cyclization strategy, this invention enables the production of Impurity F with unprecedented purity levels, transforming how manufacturers approach impurity profiling and regulatory compliance for parathyroid hormone analogs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific aspartimide impurities like Impurity F has been plagued by extremely low yields and poor selectivity, creating a bottleneck for quality control laboratories. Traditional approaches often rely on forced degradation conditions, such as treatment with strong bases like DBU or prolonged exposure to acidic environments, to induce the formation of the succinimide ring. However, these harsh conditions lack specificity, leading to a complex mixture of degradation products where the target impurity constitutes less than 1% of the total mass. Furthermore, conventional solution-phase cyclization attempts using standard coupling reagents like HOAt and PyAOP have similarly failed to drive the reaction to completion, typically resulting in crude purities hovering around 1%. This inefficiency necessitates extensive and costly purification efforts, often rendering the production of sufficient quantities of the impurity standard economically unviable for routine testing and long-term stability studies.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing a highly controlled on-resin cyclization strategy that dramatically improves both yield and purity. Instead of relying on random degradation, the process employs orthogonal protecting groups, specifically Allyl (All) or 4-{N-[1-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3-methylbutyl]-amino}benzyl (Dmab), on the side chain of the Aspartic acid residue at position 30. This allows for the selective exposure of the carboxyl group while the peptide chain remains intact on the solid support. The subsequent introduction of diphenyl phosphorazidate (DPPA) facilitates the formation of a reactive acyl azide intermediate, which undergoes efficient intramolecular nucleophilic substitution with the amino group of the adjacent Valine residue. This targeted mechanism bypasses the thermodynamic barriers of previous methods, enabling the crude peptide purity to reach approximately 42%, a massive improvement over prior art, and allowing for final purification to exceed 92% purity with relative ease.
Mechanistic Insights into DPPA-Mediated Aspartimide Formation
The core innovation of this synthesis lies in the precise manipulation of the peptide's chemical environment to favor the formation of the five-membered aspartimide ring. The reaction mechanism initiates with the activation of the free beta-carboxyl group of the Aspartic acid residue at position 30. Upon treatment with diphenyl phosphorazidate in the presence of a base like triethylamine, the carboxyl group is converted into a highly reactive acyl azide species. This activation is crucial because it lowers the energy barrier for the subsequent nucleophilic attack. The proximity of the N-terminal amino group of the Valine residue at position 31, enforced by the rigid conformation of the peptide backbone on the solid support, facilitates an intramolecular attack on the activated carbonyl carbon. This nucleophilic substitution results in the expulsion of nitrogen gas and the formation of the cyclic imide structure characteristic of Impurity F.
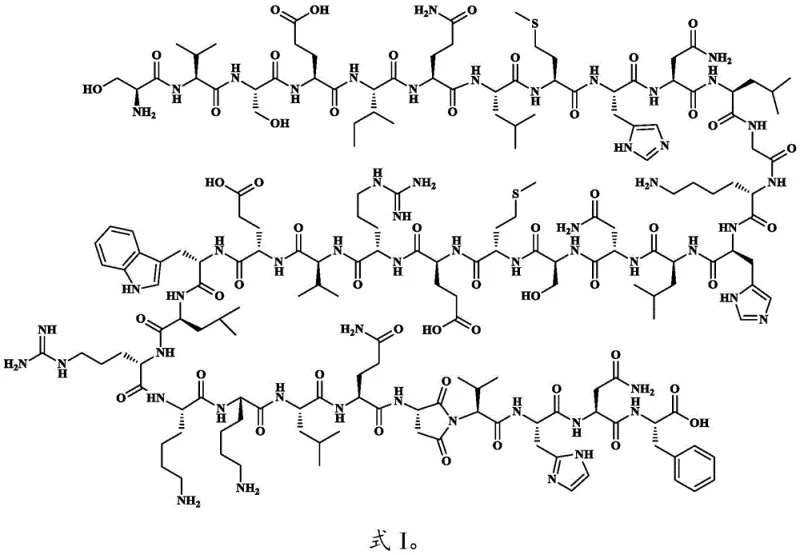
Controlling the stereochemistry and preventing racemization during this cyclization is paramount for generating a valid reference standard. The use of mild deprotection conditions for the orthogonal groups—such as palladium-catalyzed removal for the Allyl group or hydrazine hydrate for the Dmab group—ensures that the rest of the peptide sequence remains untouched and optically pure. This selectivity is a distinct advantage over global deprotection methods that might expose other sensitive residues to reactive conditions. Furthermore, the solid-phase nature of the reaction helps to mitigate intermolecular aggregation, a common issue in peptide synthesis that can lead to deletion sequences or oligomerization. By maintaining the peptide in a pseudo-dilute state on the resin, the intramolecular reaction is kinetically favored over intermolecular side reactions, ensuring that the resulting impurity profile is clean and dominated by the target Asu-Val cyclic structure.
How to Synthesize Teriparatide Impurity F Efficiently
The synthesis of this complex peptide impurity requires a meticulous adherence to solid-phase protocols, beginning with the selection of an appropriate resin and the sequential assembly of the amino acid sequence. The process demands precise control over coupling efficiencies and deprotection steps to minimize deletion sequences before the critical cyclization event. The following guide outlines the strategic workflow derived from the patent data, highlighting the critical junctures where process parameters must be tightly controlled to achieve the reported high purity outcomes. For the detailed standardized operating procedures and specific reagent quantities, please refer to the step-by-step synthesis guide below.
- Load Wang resin and sequentially couple Fmoc-protected amino acids from C-terminus to N-terminus, utilizing orthogonal protecting groups (All or Dmab) for the Asp30 side chain.
- Selectively remove the side-chain protecting group on Asp30 using either palladium catalysis (for Allyl) or hydrazine hydrate (for Dmab) without cleaving the peptide from the resin.
- Perform intramolecular cyclization using diphenyl phosphorazidate (DPPA) to form the aspartimide ring, followed by acidic cleavage to release the target impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical superiority. The ability to generate high-purity impurity standards efficiently translates directly into reduced operational costs and enhanced supply chain resilience. By eliminating the need for extensive recycling of low-yield batches, manufacturers can secure a more stable supply of critical reference materials, ensuring that quality control timelines are met without delay. This reliability is crucial for maintaining regulatory compliance and avoiding costly production stoppages due to failed batch releases.
- Cost Reduction in Manufacturing: The dramatic increase in crude purity from less than 1% to over 40% fundamentally alters the cost structure of producing this intermediate. In traditional low-yield processes, the cost of goods sold is heavily inflated by the sheer volume of raw materials and solvents required to isolate a minute amount of product. By improving the reaction efficiency, the consumption of expensive protected amino acids and coupling reagents is drastically reduced per gram of final product. Additionally, the simplified purification profile means less preparative HPLC runtime and solvent usage, leading to significant savings in downstream processing costs and waste disposal fees.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing or synthesizing niche impurity standards. The robustness of this new method, which utilizes commercially available reagents like DPPA and standard Fmoc-amino acids, reduces dependency on custom synthesis vendors who may struggle with low-yield routes. The scalability of the solid-phase approach allows for rapid ramp-up from gram to kilogram scales if demand increases, ensuring that pharmaceutical companies can maintain their inventory of reference standards without facing long lead times or supply shortages.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the move away from harsh degradation conditions (like prolonged heating in strong acid or base) to a controlled enzymatic-like cyclization reduces the generation of hazardous waste. The process operates under milder conditions, lowering energy consumption associated with heating and cooling cycles. Furthermore, the higher selectivity reduces the burden on wastewater treatment facilities by minimizing the load of complex organic byproducts, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Teriparatide Impurity F. These insights are derived directly from the patented technology and are intended to assist R&D and quality assurance teams in understanding the value proposition of this advanced synthesis method. Understanding these details is key to integrating this impurity standard into your broader quality control framework effectively.
Q: Why is the synthesis of Teriparatide Impurity F critical for drug development?
A: Teriparatide is prone to forming aspartimide impurities during production and storage, specifically involving residues Asp30 and Val31. Regulatory bodies require rigorous identification and quantification of these degradation products to ensure patient safety and drug efficacy. Having a reliable reference standard of Impurity F is essential for validating analytical methods and establishing shelf-life specifications.
Q: How does the novel DPPA-mediated cyclization method improve upon traditional synthesis routes?
A: Conventional methods such as DBU catalysis or acid degradation typically yield less than 1% of the target impurity, making isolation difficult and costly. The patented approach utilizing diphenyl phosphorazidate (DPPA) for ring closure significantly enhances reaction efficiency, achieving crude peptide purities around 42% and final purified purities exceeding 92%, thereby streamlining the supply of high-quality reference materials.
Q: What are the key technical challenges in synthesizing aspartimide peptides?
A: The primary challenge lies in controlling the regioselectivity of the cyclization to form the specific five-membered succinimide ring between the Asp side chain and the subsequent amine without causing racemization or unwanted side reactions. The use of orthogonal protecting groups like Allyl or Dmab allows for precise deprotection of the Asp side chain while the peptide remains anchored to the solid support, facilitating the specific intramolecular nucleophilic substitution required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Teriparatide Impurity F Supplier
As the global demand for complex peptide therapeutics continues to rise, the need for high-quality impurity profiles becomes ever more critical for regulatory approval and market access. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in peptide chemistry to deliver solutions that meet the rigorous demands of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in early-stage development or full-scale commercial manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Teriparatide Impurity F meets the highest standards of identity and purity required by global pharmacopeias.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and purity requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control strategies are supported by the most reliable and advanced synthetic technologies available in the market.
