Revolutionizing Nilapanib Intermediate Production: A Dual Catalytic Photoredox Approach for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the synthesis of complex oncology drug intermediates, particularly for PARP inhibitors like nilapanib. Patent CN113461597A introduces a groundbreaking preparation method that fundamentally shifts the synthetic paradigm from traditional precious metal catalysis to a synergistic photoredox and nickel dual-catalytic system. This innovation addresses critical bottlenecks in the supply chain of nilapanib intermediates, specifically the 3-(4-bromophenyl)piperidine scaffold, by leveraging visible light energy to drive decarboxylative cross-coupling reactions. By replacing harsh thermal conditions and expensive noble metals with mild photochemical activation and earth-abundant nickel, this technology offers a compelling value proposition for manufacturers aiming to reduce both environmental impact and production costs while maintaining high purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
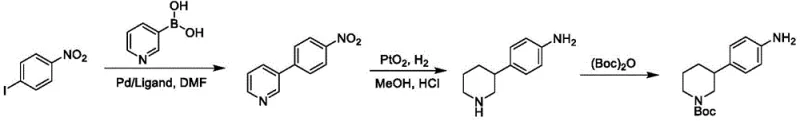
The Limitations of Conventional Methods
Historically, the synthesis of key nilapanib intermediates has relied heavily on Suzuki-Miyaura cross-coupling reactions, which, while robust, present significant economic and logistical challenges for large-scale manufacturing. As illustrated in the prior art routes, the conventional synthesis often necessitates the use of 3-pyridine boronic acid derivatives as starting materials, which are notoriously difficult to prepare, exhibit poor stability during storage, and command high market prices due to complex supply chains. Furthermore, these traditional routes typically require the continuous use of palladium and platinum catalysts across multiple steps, such as the initial coupling followed by hydrogenation using PtO2, leading to exorbitant raw material costs and stringent requirements for heavy metal removal in the final API. The reliance on these precious metals not only inflates the cost of goods sold (COGS) but also introduces supply chain vulnerabilities associated with the geopolitical scarcity of platinum group metals.
The Novel Approach
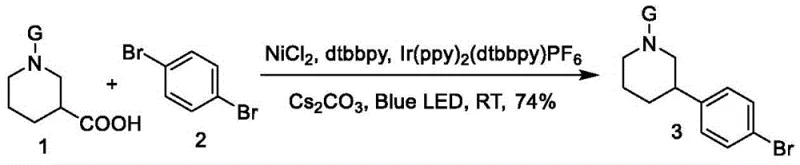
In stark contrast to the legacy methods, the novel approach disclosed in the patent utilizes a decarboxylative cross-coupling strategy that bypasses the need for pre-functionalized boronic acids entirely. Instead, it employs readily available and stable N-protected 3-carboxylic acid piperidine derivatives as the nucleophilic partners, reacting them directly with 1,4-dibromobenzene under the concerted catalysis of a photocatalyst and a nickel catalyst. This methodological shift eliminates the instability issues associated with boronic acids and removes the necessity for expensive palladium ligands and platinum hydrogenation catalysts. The reaction proceeds under mild conditions using visible light irradiation, which not only enhances safety by avoiding high-temperature and high-pressure regimes but also improves atom economy by directly utilizing the carboxylic acid moiety as a leaving group, thereby streamlining the synthetic sequence and reducing the overall waste generation associated with multi-step protection and deprotection strategies.
Mechanistic Insights into Photoredox/Nickel Dual Catalysis
The core of this technological advancement lies in the intricate interplay between the photoredox cycle and the nickel catalytic cycle, which enables the activation of inert carboxylic acids for C(sp3)-C(sp2) bond formation. Upon irradiation with blue LED light, the transition metal photocatalyst, such as Ir(ppy)2(dtbbpy)PF6, absorbs photons to reach an excited state capable of engaging in single-electron transfer (SET) processes. This excitation facilitates the oxidation of the carboxylate substrate, generating a highly reactive alkyl radical intermediate via decarboxylation. Simultaneously, the nickel catalyst, typically NiCl2 or NiBr2 coordinated with a bipyridine ligand like dtbbpy, undergoes oxidative addition with the aryl bromide electrophile. The convergence of these two cycles occurs when the photogenerated alkyl radical is captured by the nickel-aryl species, followed by reductive elimination to forge the desired carbon-carbon bond and regenerate the active nickel catalyst, ensuring a continuous and efficient turnover.
From an impurity control perspective, this radical-mediated mechanism offers distinct advantages over traditional ionic pathways by operating under kinetic control that favors the desired cross-coupling product. The mild reaction temperature range of 0°C to 60°C significantly suppresses thermal degradation pathways and unwanted side reactions, such as homocoupling of the aryl halide or beta-hydride elimination, which are common pitfalls in thermal nickel catalysis. Furthermore, the use of cesium carbonate as a base ensures effective deprotonation of the carboxylic acid without introducing harsh alkaline conditions that could compromise the integrity of the piperidine ring or the protecting group. The result is a cleaner reaction profile that simplifies downstream processing, allowing for the isolation of the target nilapanib intermediate as a high-purity white powder through standard workup procedures like extraction and silica gel chromatography, thus meeting the rigorous quality specifications demanded by pharmaceutical regulators.
How to Synthesize Tert-Butyl 3-(4-Bromophenyl)piperidine-1-Carboxylate Efficiently
The practical implementation of this synthesis involves a straightforward four-step protocol that is highly amenable to standard laboratory and pilot plant equipment. The process begins with the dissolution of the N-protected piperidine carboxylic acid and the photocatalyst in tetrahydrofuran, followed by the sequential addition of the nickel catalyst, base, and aryl bromide electrophile to form a homogeneous reaction mixture. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Dissolve the N-protected 3-carboxylic acid piperidine derivative (Compound 1) and the transition metal photocatalyst (e.g., Ir(ppy)2(dtbbpy)PF6) in tetrahydrofuran solvent.
- Add the Nickel catalyst (NiCl2 or NiBr2), cesium carbonate base, and 1,4-dibromobenzene (Compound 2) to the solution and mix until clear.
- Irradiate the mixture with blue LED light (450nm-480nm) at 40°C while stirring until HPLC confirms complete consumption of the starting material, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photoredox-mediated synthesis represents a strategic opportunity to optimize the cost structure and reliability of nilapanib intermediate sourcing. By transitioning away from precious metal-dependent chemistries, manufacturers can achieve substantial cost savings through the elimination of expensive palladium and platinum reagents, which are subject to volatile market pricing and supply constraints. The substitution of these noble metals with earth-abundant nickel and recyclable photocatalysts drastically reduces the raw material expenditure per kilogram of product, while simultaneously lowering the costs associated with heavy metal scavenging and waste disposal, thereby enhancing the overall economic viability of the manufacturing process without compromising on quality or yield.
- Cost Reduction in Manufacturing: The economic benefits of this new route are driven primarily by the replacement of high-cost inputs with commoditized alternatives. The elimination of 3-pyridine boronic acid, a specialized and unstable reagent, in favor of stable carboxylic acid derivatives significantly lowers the bill of materials. Additionally, the removal of platinum oxide (PtO2) from the hydrogenation step eradicates a major cost center, as platinum is one of the most expensive elements in the periodic table. This shift allows for a leaner cost model where the primary expenses are limited to basic solvents and inexpensive nickel salts, resulting in a more competitive pricing structure for the final intermediate that can be passed down the supply chain to API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the use of robust and shelf-stable starting materials. Unlike boronic acids which require careful storage and handling to prevent degradation, the carboxylic acid precursors used in this process are chemically stable and widely available from bulk chemical suppliers, reducing the risk of production delays due to raw material spoilage. Furthermore, the reaction conditions are mild and do not require specialized high-pressure reactors or cryogenic cooling, meaning that production can be easily distributed across multiple manufacturing sites with standard glass-lined or stainless steel equipment, ensuring continuity of supply even in the face of regional disruptions or capacity constraints at single-source facilities.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and community acceptance for manufacturing sites. The use of visible light as the energy source is inherently safer and more sustainable than thermal heating, reducing the carbon footprint of the process. Moreover, the high atom economy of the decarboxylative coupling minimizes the generation of stoichiometric salt waste, and the absence of toxic heavy metals simplifies effluent treatment. This eco-friendly approach not only reduces compliance costs related to waste management but also positions the supply chain favorably for increasingly stringent environmental regulations, ensuring long-term operational sustainability and reducing the risk of shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic methodology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on the feasibility and advantages of the process for potential partners and licensees.
Q: What are the primary cost drivers eliminated in this new nilapanib intermediate synthesis?
A: The new process eliminates the reliance on expensive palladium and platinum catalysts used in traditional Suzuki couplings, replacing them with earth-abundant nickel and reusable photocatalysts. Furthermore, it utilizes stable carboxylic acid starting materials instead of unstable and costly pyridine boronic acids.
Q: How does the photoredox mechanism improve impurity profiles compared to thermal methods?
A: The single-electron transfer (SET) mechanism driven by visible light allows for mild reaction conditions (0-60°C), significantly reducing thermal degradation byproducts and side reactions common in high-temperature cross-coupling, thereby simplifying downstream purification.
Q: Is this synthetic route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates at ambient pressure and moderate temperatures using standard LED light sources, making it highly amenable to flow chemistry and large-scale batch processing without the safety risks associated with high-pressure hydrogenation or pyrophoric reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nilapanib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photoredox catalytic technology in reshaping the landscape of oncology intermediate manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nilapanib intermediate we produce adheres to the highest global quality standards required for clinical and commercial drug development.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and sustainable synthesis route for their nilapanib programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, as well as obtain specific COA data and route feasibility assessments to validate the commercial viability of this innovative approach for your supply chain needs.
