Scalable Synthesis of Alkyl Ether Substituted Pyridine-2,3-Dicarboxylic Acid Derivatives for Imidazolinone Herbicides
The landscape of agrochemical intermediate manufacturing is undergoing a significant transformation driven by the need for more efficient, cost-effective synthetic routes for high-value herbicides. A pivotal advancement in this sector is detailed in patent CN107759516B, which discloses a novel preparation method for alkyl ether substituted pyridine-2,3-dicarboxylic acid derivatives. These compounds serve as critical precursors for the synthesis of imidazolinone herbicides, a class of agricultural chemicals renowned for their high efficacy and environmental safety profile. The technical breakthrough lies in shifting from traditional, cumbersome post-alkoxylation strategies to a streamlined pre-alkoxylation approach. By reacting beta-diketone derivatives with acrolein derivatives in the presence of an ammonia source, this methodology bypasses the selectivity issues and low yields that have historically plagued the production of methoxymethyl-substituted pyridine rings. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages and scalability of this route is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl ether substituted pyridine-2,3-dicarboxylic acid derivatives has been fraught with technical challenges, primarily relying on post-alkoxylation methods. In these conventional pathways, the alkoxy group is introduced after the pyridine ring has already been formed, typically involving harsh halogenation steps such as bromination or chlorination followed by nucleophilic substitution. The patent background highlights a severe bottleneck in this approach: the substituent effect on yield is drastic. For instance, while ethyl substituents might afford yields greater than 90%, the introduction of a methoxymethyl group (-CH2OCH3) via traditional routes often results in yields of less than 1%. This catastrophic drop in efficiency is attributed to poor halogenation selectivity and the instability of intermediates under the required reaction conditions. Furthermore, the raw materials for these older methods, such as diethyl aminomaleate or dichloro diethyl maleate, are often expensive and require long reaction times, rendering the process economically unviable for large-scale cost reduction in herbicide manufacturing. The reliance on palladium catalysts in carbonylation routes or Weissmier reagents further exacerbates cost and complexity, creating a supply chain vulnerable to raw material volatility.
The Novel Approach
In stark contrast, the methodology outlined in CN107759516B employs a pre-alkoxylation strategy that fundamentally alters the synthetic logic. Instead of struggling to functionalize a formed pyridine ring, this route constructs the ring around the pre-functionalized carbon skeleton. The process utilizes beta-diketone derivatives (General Formula II) and acrolein derivatives (General Formula III) as key building blocks. This approach allows the alkoxy group to be present on the acrolein starting material, thereby avoiding the difficult late-stage functionalization entirely. The reaction proceeds through a condensation followed by cyclization with an ammonia source, achieving product yields that consistently exceed 80%, with optimized examples reaching up to 86.8%. This represents a monumental improvement over the sub-1% yields of comparable traditional methods. By simplifying the molecular construction to a one-pot or two-step sequence using inexpensive, commercially available solvents like toluene or ethanol, this novel approach offers a robust pathway for the commercial scale-up of complex agrochemical intermediates.
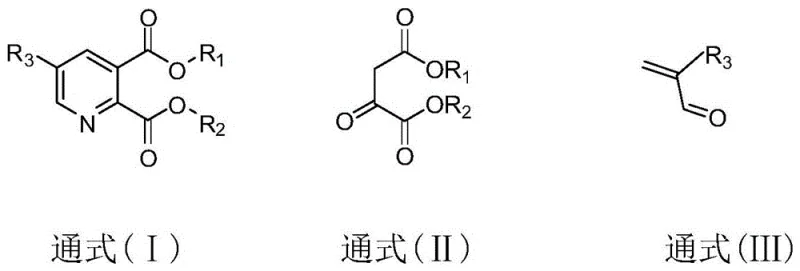
Mechanistic Insights into Pre-Alkoxylation Condensation Cyclization
The core of this synthetic innovation lies in the precise orchestration of a condensation-cyclization cascade. The reaction initiates with the interaction between the active methylene group of the beta-diketone derivative and the electron-deficient double bond of the acrolein derivative. Under the influence of a catalyst—ranging from simple alkali metal hydroxides like NaOH and KOH to hydrides like NaH—or even under catalyst-free conditions, a Michael-type addition or aldol-like condensation occurs. This forms a crucial transition product that contains the necessary carbon framework for the pyridine ring. The presence of the alkoxy-substituted alkyl group (R3) on the acrolein moiety is preserved throughout this step, ensuring the final product possesses the desired substitution pattern without requiring additional protection-deprotection sequences. The subsequent addition of an ammonia source, such as ammonium sulfamate or ammonium acetate, triggers the ring-closure event. The nitrogen atom from the ammonia source attacks the carbonyl centers, facilitating dehydration and aromatization to form the stable pyridine nucleus. This mechanism is highly tolerant of various solvents, including aromatic hydrocarbons, alcohols, and esters, providing flexibility for process optimization.

From an impurity control perspective, this mechanism offers distinct advantages over halogenation-based routes. Traditional methods often generate halogenated by-products and isomeric impurities that are difficult to separate, complicating the purification of high-purity pyridine derivatives. In the pre-alkoxylation route described here, the primary by-products are water and unreacted starting materials, which are easily removed via standard workup procedures like extraction and distillation. The use of mild reaction temperatures (typically -20 to 160°C, with preferred ranges of 10-60°C for condensation and reflux for cyclization) minimizes thermal decomposition and polymerization side reactions. The patent data indicates that the reaction time is inversely proportional to temperature, allowing manufacturers to balance throughput with selectivity. For example, maintaining the condensation step at 20°C for 1 hour followed by reflux for 3 hours provides an optimal balance, ensuring complete conversion while suppressing the formation of tarry residues. This clean reaction profile significantly reduces the burden on downstream purification units, directly translating to higher overall process efficiency.
How to Synthesize 5-Methoxymethyl Pyridine-2,3-Dicarboxylic Acid Ethyl Ester Efficiently
The practical implementation of this synthesis is designed for seamless integration into existing fine chemical manufacturing facilities. The process typically begins by charging a reactor with a solvent such as toluene or ethanol and the beta-diketone precursor, often diethyl butanedioate derivatives. A catalytic amount of base, such as a 30% NaOH solution, is added to initiate the reaction. The acrolein derivative, specifically 2-methoxymethylacrolein in the case of the target herbicide intermediate, is then added dropwise at controlled temperatures to manage exotherms. Once the transition product is formed, the ammonia source is introduced, and the mixture is heated to reflux to drive the cyclization to completion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching protocols are critical for reproducibility.
- React beta-diketone derivatives (Formula II) with acrolein derivatives (Formula III) at -20 to 80°C in the presence of a solvent and optional catalyst to form a transition product.
- Add an ammonia source compound (such as ammonium sulfamate or acetate) to the reaction mixture.
- Heat the mixture to reflux temperature (20-160°C) to effect cyclization, yielding the target alkyl ether substituted pyridine-2,3-dicarboxylic acid derivative (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN107759516B presents a compelling value proposition centered on cost stability and operational reliability. The shift away from expensive, specialized raw materials like diethyl aminomaleate towards commodity chemicals like beta-diketones and acrolein derivatives drastically simplifies the sourcing landscape. This diversification of raw material inputs mitigates the risk of supply disruptions caused by the monopoly of specific niche chemical suppliers. Furthermore, the elimination of multi-step halogenation and subsequent substitution processes reduces the total number of unit operations required. Fewer processing steps inherently mean lower energy consumption, reduced solvent usage, and diminished waste generation, all of which contribute to a leaner manufacturing cost structure without compromising on quality.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived principally from the high atom economy and the avoidance of precious metal catalysts. Unlike palladium-catalyzed carbonylation methods which require expensive metal recovery systems to meet regulatory standards, this organic synthesis route utilizes inexpensive inorganic bases or operates catalyst-free. The high yields reported (consistently above 80% compared to <1% for traditional methoxymethyl routes) mean that less raw material is wasted per kilogram of finished product. This efficiency gain allows for a significant reduction in the cost of goods sold (COGS), enabling more competitive pricing for the final herbicide active ingredient. Additionally, the simplified purification process reduces the load on chromatography or recrystallization units, lowering utility and labor costs associated with downstream processing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain continuity by shortening the overall production cycle time. The patent examples demonstrate that the entire reaction sequence can be completed within approximately 5 hours, excluding workup time. This rapid turnover allows for increased batch frequency and better responsiveness to fluctuating market demands. Moreover, the tolerance of the reaction to various solvents (toluene, ethanol, ethyl acetate) provides operational flexibility; if a specific solvent faces logistical constraints or price spikes, the process can be adapted to use alternative green solvents without re-validating the entire chemistry. This adaptability is crucial for maintaining reducing lead time for high-purity agrochemical intermediates in a volatile global market.
- Scalability and Environmental Compliance: The technology has been proven effective at the pilot and industrial scale, as evidenced by Example 31 in the patent, which details a successful run in a 1000L enamel kettle producing 40kg scale batches with consistent yields. The absence of hazardous halogenating agents (like bromine or thionyl chloride) and heavy metals simplifies waste treatment and aligns with increasingly stringent environmental regulations. The primary waste streams are aqueous salts and organic solvents which are easier to treat or recycle compared to halogenated waste. This environmental compatibility not only reduces disposal costs but also future-proofs the supply chain against tightening regulatory frameworks regarding chemical manufacturing emissions and effluent quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pre-alkoxylation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility of adopting this route for large-scale production. Understanding these nuances is vital for technical teams evaluating the transition from legacy synthesis methods to this modern, efficient protocol.
Q: Why is the pre-alkoxylation method superior to post-alkoxylation for this intermediate?
A: Traditional post-alkoxylation methods involve difficult halogenation steps with poor selectivity and extremely low yields (often <1%) for methoxymethyl substituents. The pre-alkoxylation route described in CN107759516B introduces the alkoxy group before ring closure, utilizing readily available acrolein derivatives to achieve yields exceeding 85%.
Q: What are the optimal reaction conditions for industrial scale-up?
A: The patent demonstrates successful scale-up in a 1000L reactor using ethanol or toluene as solvents. The condensation step is best performed at mild temperatures (20-60°C), followed by reflux cyclization with an ammonia source like ammonium sulfamate, completing the process within approximately 5 hours.
Q: Can this intermediate be directly converted into imidazolinone herbicides?
A: Yes, the resulting diester (Formula I) can be hydrolyzed to the diacid or reacted directly with aminocarboxamide compounds in the presence of a strong base and inert solvent to produce the final imidazolinone herbicide structure (Formula IV).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-2,3-Dicarboxylic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in fine chemical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped to handle the specific requirements of the pre-alkoxylation process described in CN107759516B, ensuring that the delicate balance of condensation and cyclization is maintained with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkyl ether substituted pyridine derivatives meets the exacting standards required for downstream herbicide synthesis. Our commitment to quality ensures that your supply of critical intermediates remains uninterrupted and consistent.
We invite you to collaborate with us to leverage this cost-effective technology for your agrochemical portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to optimizing your supply chain and driving down manufacturing costs through innovative chemistry.
