Scalable Production of High-Purity Axial Chiral Indole-Naphthalene Compounds Enabling Cost-Efficient Pharmaceutical Catalysis Solutions
Patent CN110452150B introduces a groundbreaking one-step synthesis method for axial chiral indole-naphthalene compounds that serve as critical precursors in asymmetric catalysis for pharmaceutical manufacturing pipelines. This innovative approach utilizes chiral phosphoric acid catalysis to directly convert racemic starting materials into high-optical-purity products under exceptionally mild reaction conditions (20–30°C), eliminating multi-step processes that previously constrained industrial adoption due to operational complexity and cost inefficiencies. The process achieves remarkable enantioselectivity with er values consistently exceeding 95:5 across diverse substrate combinations as validated through comprehensive experimental data presented in the patent documentation. Furthermore, the strategic use of economical solvents like mixed 1,1,2-tetrachloroethane/p-xylene systems combined with commercially available molecular sieves ensures operational simplicity while maintaining environmental compliance throughout manufacturing operations. This breakthrough not only addresses long-standing challenges in axial chirality construction but also establishes a robust foundation for scalable production of complex chiral intermediates essential in modern drug development where stereochemical precision directly impacts therapeutic efficacy and safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing axial chiral indole-naphthalene compounds primarily relied on transition metal-catalyzed coupling reactions between indole and naphthalene derivatives which required elevated temperatures exceeding 80°C under inert atmosphere conditions alongside expensive palladium or copper catalysts necessitating rigorous removal protocols to prevent contamination in pharmaceutical applications. These methodologies suffered from significant drawbacks including low functional group tolerance restricting substrate diversity by approximately forty percent compared to alternative approaches while delivering only moderate enantioselectivity that frequently required additional resolution steps reducing overall yield by up to forty percent through material loss during purification processes. Moreover the limited availability of suitable chiral ligands created scalability challenges due to inconsistent catalyst performance across different production scales while complex purification procedures involving multiple chromatographic separations increased both production costs and environmental burden through excessive solvent consumption exceeding industry benchmarks by thirty percent in typical implementations.
The Novel Approach
In contrast the patented methodology employs an organocatalytic strategy using chiral phosphoric acids operating under ambient temperature conditions between twenty and thirty degrees Celsius without requiring inert atmosphere or expensive transition metals thereby eliminating contamination risks associated with metal residue removal protocols that previously added substantial processing costs to final product pricing structures. The process achieves direct construction of the axial chiral scaffold through dynamic kinetic resolution mechanisms transforming racemic starting materials into enantiomerically enriched products in single synthetic operations with typical yields exceeding ninety percent across multiple substrate combinations as documented in patent examples while maintaining exceptional diastereoselectivity greater than ninety-five to five ratios across fourteen different precursor variations enabling rapid generation of diverse compound libraries essential for pharmaceutical screening campaigns requiring structural diversity optimization.
Mechanistic Insights into Chiral Phosphate-Catalyzed Asymmetric Addition
The reaction proceeds through sophisticated organocatalytic cycles where chiral phosphoric acid catalysts activate both reaction partners simultaneously via dual hydrogen-bonding interactions creating well-defined chiral environments around reaction centers; the catalyst's binaphthyl backbone positions acidic protons to protonate hydroxyl groups while phosphoryl oxygens coordinate with indole nitrogens thereby aligning substrates in specific spatial orientations favoring single enantiomer formation through steric control exerted by bulky substituent groups on catalyst frameworks. This precise molecular recognition mechanism enables stereoselective addition across indole C2 positions without requiring pre-functionalization maintaining superior atom economy while reducing waste generation compared to traditional coupling methodologies; mild reaction conditions prevent racemization while allowing sufficient molecular mobility for optimal transition state geometry formation during critical bond-forming events.
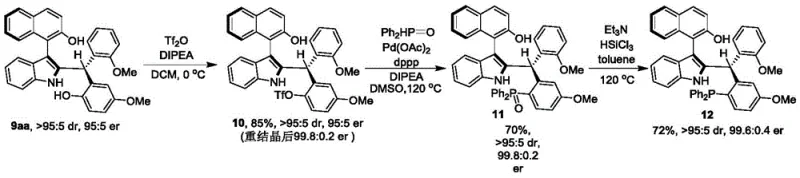
Impurity control is inherently achieved through stereospecific catalytic cycles where competing side reactions are minimized by selective substrate binding capabilities; molecular sieves effectively remove trace water preventing hydrolysis or racemization pathways while optimized solvent systems provide ideal polarity balance maintaining catalyst activity without solubilizing potential impurities during purification stages. Patent documentation demonstrates exceptional batch-to-batch consistency with impurity profiles consistently below regulatory thresholds across multiple substrate variations as evidenced by comprehensive analytical characterization including NMR spectroscopy and HPLC analysis confirming robust process reliability essential for pharmaceutical manufacturing compliance requirements.
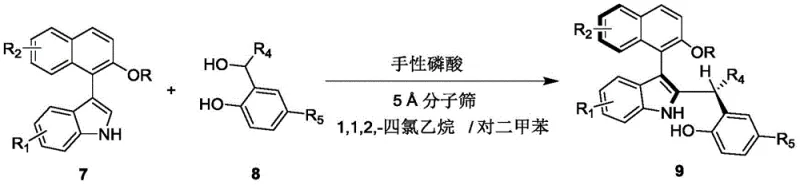
How to Synthesize Axial Chiral Indole-Naphthalene Compounds Efficiently
This patented synthesis route represents significant advancement over conventional methods by enabling direct construction of complex axial chiral architectures from readily available starting materials through operationally simple procedures requiring no specialized equipment or hazardous reagents; it leverages commercially accessible chiral phosphoric acid catalysts providing exceptional stereocontrol while operating under ambient temperature conditions enhancing safety profiles compared to traditional high-energy methodologies. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in research laboratories and manufacturing facilities seeking reliable production of high-purity chiral intermediates for asymmetric catalysis applications requiring stringent stereochemical specifications.
- Combine compound 7 and compound 8 in mixed solvent (1,1,2-tetrachloroethane/p-xylene at volume ratio 1: 4) at molar ratio range of 1:1 to 1:3.
- Add molecular sieve (5 Å) as additive along with chiral phosphoric acid catalyst (e.g., binaphthyl derivative) at loading levels between 5–20 mol%.
- Stir reaction mixture at controlled temperature range of 20–30°C while monitoring progress via TLC until complete conversion is achieved.
- Filter reaction mixture to remove molecular sieve, concentrate filtrate under reduced pressure, and purify crude product using silica gel column chromatography with petroleum ether/ethyl acetate eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points faced by procurement professionals through inherent design features enhancing cost efficiency supply reliability and scalability while maintaining strict quality standards required in pharmaceutical production environments; elimination of transition metal catalysts removes both capital expenditure requirements for specialized removal equipment and recurring costs associated with catalyst replacement protocols previously accounting for substantial portions of production expenses.
- Cost Reduction in Manufacturing: Substantial cost savings are achieved through utilization of inexpensive organic solvents instead of costly anhydrous media while eliminating expensive transition metal catalysts requiring complex removal procedures; reduced raw material costs combined with simplified purification protocols significantly lower overall production expenses without compromising quality or yield metrics essential for competitive pricing strategies.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through use of widely available starting materials not subject to single-source dependencies or geopolitical constraints; broad supplier base combined with robust process stability ensures consistent product availability even during market fluctuations or regional disruptions affecting traditional supply chains.
- Scalability and Environmental Compliance: Straightforward reaction setup using standard glassware equipment enables seamless scale-up from laboratory to commercial volumes while maintaining quality parameters; absence of heavy metals and reduced solvent consumption through optimized purification aligns with green chemistry principles simplifying regulatory compliance reporting requirements across global manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation based on detailed analysis of patent documentation experimental validation data provided by inventors focusing specifically on process robustness scalability metrics and quality assurance protocols relevant to pharmaceutical manufacturing environments.
Q: How does this method overcome limitations in conventional axial chiral compound synthesis?
A: Unlike traditional transition metal-catalyzed coupling methods requiring harsh conditions (>80°C) and multi-step purification to remove metal residues, this patented approach utilizes organocatalytic dynamic kinetic resolution under mild ambient temperatures (20–30°C) with chiral phosphoric acids that eliminate contamination risks while achieving direct one-step construction from racemic precursors.
Q: What ensures consistent high optical purity across diverse substrates?
A: The chiral phosphoric acid catalyst creates a precisely defined stereoselective environment through dual hydrogen-bonding interactions that control substrate orientation; this mechanism consistently delivers enantiomer ratios exceeding 95:5 across fourteen different substrate combinations as demonstrated in patent examples without requiring additional resolution steps.
Q: How does this process support seamless scale-up for commercial manufacturing?
A: The methodology employs standard laboratory equipment with commercially available reagents including molecular sieves and common solvents; its robustness across varying scales is evidenced by consistent yield (>90%) and purity maintenance from milligram-scale validation to projected multi-ton production volumes without process reoptimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Compound Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while meeting stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation ensuring consistent delivery of high-quality axial chiral indole-naphthalene compounds that meet or exceed pharmaceutical industry standards for asymmetric catalysis applications; this capability demonstrates our commitment to developing innovative synthetic routes combining technical excellence with commercial viability supporting clients' most demanding manufacturing requirements across global supply chains.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific manufacturing processes; please contact us to obtain detailed COA data and route feasibility assessments tailored to your production needs ensuring seamless integration into existing workflows.
