Advanced Synthesis of Berberine Platinum II Complex for High-Purity Antitumor Drug Development
Advanced Synthesis of Berberine Platinum II Complex for High-Purity Antitumor Drug Development
The pharmaceutical industry is constantly seeking next-generation metallodrugs that can overcome the severe limitations of classical platinum-based therapies, such as cisplatin resistance and systemic toxicity. Patent CN110105402B introduces a groundbreaking approach by conjugating a bioactive berberine derivative with a platinum (II) center, creating a hybrid molecule designated as Pt1. This novel berberine antitumor platinum (II) complex represents a significant leap forward in medicinal chemistry, merging the DNA-intercalating properties of alkaloids with the cross-linking capability of platinum. For R&D directors and procurement specialists alike, this technology offers a robust pathway to developing high-purity pharmaceutical intermediates with enhanced therapeutic indices. The synthesis described in the patent is not merely a laboratory curiosity but a viable, scalable process that addresses critical supply chain and efficacy challenges in oncology drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional platinum anticancer agents, including cisplatin, carboplatin, and oxaliplatin, have long been the cornerstone of chemotherapy, yet their clinical utility is increasingly hampered by acquired drug resistance and dose-limiting nephrotoxicity. Conventional synthesis methods for new platinum complexes often involve harsh reaction conditions, expensive ligands, or complex purification protocols that drive up manufacturing costs and extend lead times. Furthermore, many existing metal-based drugs lack specific targeting mechanisms, leading to indiscriminate damage to healthy tissues and severe side effects for patients. The synthesis of effective analogues frequently suffers from low yields and poor reproducibility when scaled, creating bottlenecks for reliable pharmaceutical intermediate suppliers trying to meet global demand. Additionally, the inability of many classical complexes to penetrate resistant tumor cell lines, such as the cisplatin-resistant ovarian cancer line SK-OV-3/DDP, necessitates the exploration of entirely new chemical scaffolds that can bypass these biological defense mechanisms.
The Novel Approach
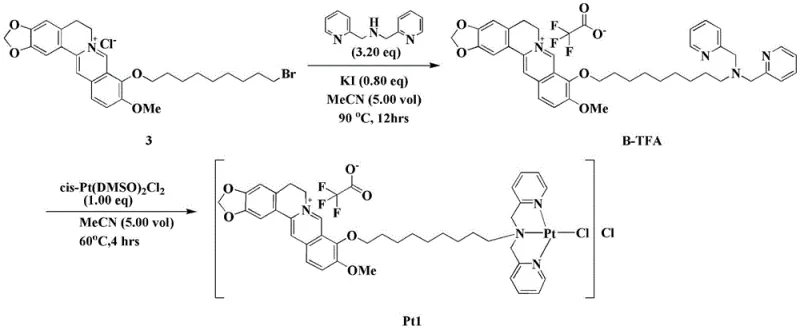
The methodology outlined in patent CN110105402B presents a sophisticated yet operationally simple solution by utilizing a bromo-berberine derivative as a functional ligand. This innovative route involves a two-step process that avoids extreme conditions, operating at moderate temperatures of 90°C and 60°C respectively. By employing readily available starting materials like dimethyl pyridylamine and dichloro-bis(dimethyl sulfoxide) platinum (II), the process significantly simplifies the supply chain requirements. The reaction proceeds in acetonitrile, a common industrial solvent that facilitates easy recovery and recycling, thereby aligning with green chemistry principles. Most notably, this approach yields the target complex Pt1 with exceptional efficiency, achieving yields upwards of 90% in optimized examples, which drastically reduces material waste and cost per kilogram. This streamlined synthesis ensures a consistent supply of high-quality material, essential for maintaining the rigorous standards required in antitumor drug manufacturing.
Mechanistic Insights into Pt(II) Coordination and Ligand Integration
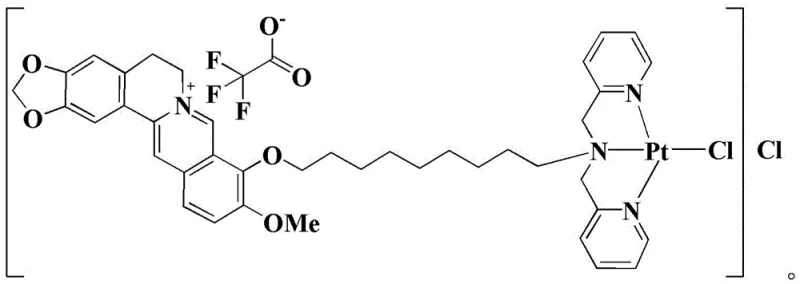
The core of this technology lies in the precise coordination chemistry that stabilizes the platinum center within the bulky berberine framework. The synthesis begins with the nucleophilic substitution of the bromo-berberine derivative by dimethyl pyridylamine, facilitated by potassium iodide, to form the B-TFA ligand. This step is critical as it installs the nitrogen donor atoms necessary for subsequent metal binding. In the second stage, the platinum (II) ion, initially coordinated with dimethyl sulfoxide (DMSO) ligands in the precursor cis-Pt(DMSO)2Cl2, undergoes a ligand exchange reaction. The nitrogen atoms of the pyridyl groups in the B-TFA ligand displace the DMSO molecules, forming stable Pt-N bonds. This coordination geometry is confirmed by detailed spectroscopic analysis, including 1H NMR and 19F NMR, which verify the integrity of the trifluoroacetate counterion and the organic scaffold. The resulting structure creates a cationic complex where the platinum is chelated, enhancing its kinetic stability in physiological conditions while retaining the ability to interact with biological targets.
From a quality control perspective, the mechanism ensures a clean impurity profile. The use of trifluoroacetic acid (TFA) salts aids in the solubility and crystallization of the intermediate, allowing for effective purification through simple washing steps. The patent data indicates that the final complex exhibits distinct spectral signatures, such as specific fluorine shifts, which serve as reliable markers for batch-to-batch consistency. This level of structural definition is crucial for regulatory filings, as it demonstrates a well-characterized active pharmaceutical ingredient (API) intermediate. The synergistic effect observed in biological assays suggests that the berberine moiety does not merely act as a passive carrier but actively contributes to the antitumor activity, potentially through mitochondrial disruption, while the platinum center executes DNA crosslinking. This dual-action mechanism is key to overcoming the resistance profiles that plague monotherapy agents.
How to Synthesize Berberine Platinum II Complex Efficiently
The synthesis of this high-value antitumor intermediate is designed for operational simplicity without compromising on purity or yield. The process leverages standard organic synthesis techniques that are easily transferable from bench scale to pilot plant operations. By adhering to the specific molar ratios and thermal profiles defined in the patent, manufacturers can avoid common pitfalls such as incomplete reactions or ligand degradation. The following guide outlines the critical phases of the production workflow, ensuring that the final product meets the stringent specifications required for preclinical and clinical evaluation. Detailed standard operating procedures regarding exact stoichiometry and safety protocols are essential for maintaining compliance with Good Manufacturing Practice (GMP) standards.
- Synthesize the B-TFA ligand by reacting bromo-berberine derivatives with potassium halide and dimethyl pyridylamine in acetonitrile at 90°C for 12 hours.
- Perform the coordination reaction by dissolving the B-TFA ligand and dichloro-bis(dimethyl sulfoxide) platinum (II) in acetonitrile at 60°C for 4 to 6 hours.
- Isolate the final yellow target product Pt1 by washing with acetonitrile and drying under vacuum at 45°C to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals and standard solvents means that raw material sourcing is robust and less susceptible to geopolitical disruptions or price volatility associated with exotic reagents. The elimination of complex chromatographic purification steps in favor of precipitation and washing significantly reduces processing time and solvent consumption. This efficiency translates directly into lower operational expenditures and a reduced environmental footprint, aligning with corporate sustainability goals. Furthermore, the high yield and reproducibility of the reaction minimize batch failures, ensuring a steady flow of inventory that supports continuous manufacturing schedules and reduces the risk of stockouts for downstream drug formulation.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing a high-yielding two-step sequence that avoids the need for expensive transition metal catalysts or specialized equipment. The use of acetonitrile as a single solvent system for both reaction and purification simplifies solvent management and recovery infrastructure. By eliminating the need for cryogenic cooling or high-pressure reactors, the energy consumption per kilogram of product is drastically lowered. These factors combine to create a highly economical manufacturing profile, making the cost reduction in pharmaceutical intermediate manufacturing a reality for large-scale producers looking to optimize their margins.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on chemically stable precursors that are commercially available from multiple global vendors, mitigating the risk of single-source dependency. The mild reaction conditions (60°C to 90°C) allow the process to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities. This compatibility with existing infrastructure accelerates technology transfer and reduces the capital expenditure required for scale-up. Consequently, reducing lead time for high-purity pharmaceutical intermediates becomes achievable, enabling faster response to market demands and more agile supply chain management.
- Scalability and Environmental Compliance: The protocol is inherently scalable, as demonstrated by the linear relationship between reagent quantities and product yield in the patent examples. The waste stream is primarily composed of acetonitrile and inorganic salts, which are straightforward to treat and dispose of according to standard environmental regulations. The absence of heavy metal contaminants in the final product, other than the intended platinum payload, simplifies the purification burden and ensures compliance with strict residual solvent and metal guidelines. This ease of scale-up and environmental manageability supports the commercial scale-up of complex polymer additives and fine chemicals, ensuring long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this berberine-platinum complex. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their pipelines. Understanding these nuances is vital for making informed decisions about licensing, manufacturing partnerships, and R&D investment strategies.
Q: How does this berberine-platinum complex overcome cisplatin resistance?
A: The complex utilizes a berberine derivative as an active ligand which provides a dual mechanism of action. While the platinum center facilitates DNA crosslinking similar to cisplatin, the berberine moiety enhances mitochondrial targeting and DNA intercalation, showing superior inhibition against cisplatin-resistant cell lines like SK-OV-3/DDP.
Q: What are the critical reaction conditions for maximizing yield?
A: Maintaining the reaction temperature at precisely 90°C for the ligand synthesis and 60°C for the coordination step is critical. Temperatures above 90°C can cause carbon chain breakage, while lower temperatures significantly reduce reaction kinetics. Using a molar ratio of 1:0.8:3.2 for the bromo-derivative, potassium iodide, and amine ensures complete conversion.
Q: Is the synthesis scalable for commercial production?
A: Yes, the process uses common solvents like acetonitrile and operates at moderate temperatures (60-90°C) without requiring cryogenic conditions or exotic catalysts. The simple workup procedure involving washing and vacuum drying makes it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Berberine Platinum Complex Supplier
As the demand for novel antitumor agents grows, partnering with an experienced CDMO is essential for navigating the complexities of commercializing new chemical entities. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market supply. Our state-of-the-art facilities are equipped to handle the specific requirements of platinum chemistry, including dedicated containment systems and rigorous QC labs capable of verifying stringent purity specifications. We understand that consistency is key in oncology drug development, and our quality management systems are designed to deliver batch-after-batch reliability that meets global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation cancer therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive advantage in the global pharmaceutical market through efficient, high-quality manufacturing solutions.
