Revolutionizing Peptide Synthesis: Advanced Polypeptide Guanylating Agents for Commercial Scale
The landscape of peptide drug manufacturing is undergoing a significant transformation driven by the need for more efficient and cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN112358423A, which discloses a novel preparation method for polypeptide guanylating agents. These agents are critical for introducing guanidino functional groups into amino acid sequences, a modification essential for enhancing the biological activity and molecular recognition properties of therapeutic peptides. Unlike traditional methods that rely on hazardous and expensive reagents, this innovation utilizes p-toluenesulfonyl chloride or methanesulfonyl chloride to achieve high-effency guanidinylation under mild conditions. For global procurement leaders and R&D directors, this represents a strategic opportunity to optimize supply chains for pharmaceutical intermediates while maintaining rigorous quality standards.
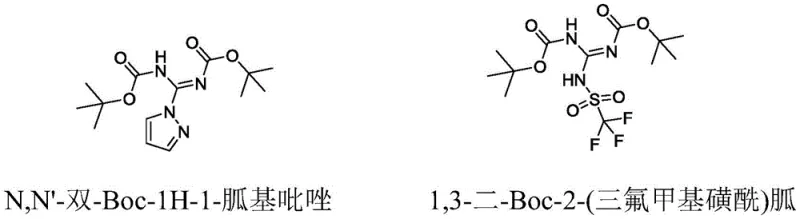
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of guanidino compounds has been plagued by significant technical and economic hurdles that hinder large-scale production efficiency. Traditional guanylating agents, such as N,N'-di-Boc-1H-1-guanidylpyrazole or those derived from trifluoromethanesulfonyl chloride, present severe challenges in an industrial setting. The reliance on triflyl chloride introduces extreme corrosivity, necessitating specialized equipment and stringent safety protocols that drastically inflate capital expenditure. Furthermore, these conventional routes often demand ultra-low temperature reactions, sometimes reaching cryogenic levels, which imposes a heavy energy burden and complicates process control. The high cost of raw materials combined with the operational complexity of handling hazardous byproducts creates a bottleneck for cost reduction in peptide manufacturing, making it difficult for suppliers to offer competitive pricing without compromising margins.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a robust alternative that leverages readily available sulfonyl chlorides to drive the reaction forward with exceptional efficiency. By reacting 1,3-di(tert-butoxycarbonyl) guanidine with p-toluenesulfonyl chloride in the presence of a mild organic base, the process achieves the desired transformation at temperatures ranging from 0°C to 30°C. This shift from cryogenic to near-ambient conditions simplifies the engineering requirements and allows for the use of standard reactor vessels. The resulting intermediates, such as 1,3-di-Boc-2-(p-toluenesulfonyl) guanidine, serve as potent electrophiles that react smoothly with primary amines. This approach not only mitigates safety risks associated with corrosive reagents but also streamlines the workflow, positioning it as a superior choice for a reliable pharmaceutical intermediates supplier aiming to enhance production throughput.

Mechanistic Insights into Sulfonyl-Mediated Guanidinylation
The chemical elegance of this new route lies in the activation of the guanidine nitrogen through sulfonylation, creating a highly reactive species capable of efficient nucleophilic substitution. In the initial step, the organic base deprotonates the guanidine derivative, facilitating the attack on the sulfur atom of the sulfonyl chloride. This results in the formation of a sulfonamide linkage where the sulfonyl group acts as a powerful electron-withdrawing entity. This electronic effect significantly increases the electrophilicity of the central carbon atom within the guanidine core, priming it for subsequent attack by amine nucleophiles. The stability of the Boc protecting groups throughout this process ensures that the integrity of the molecule is maintained, preventing unwanted side reactions that could lead to complex impurity profiles difficult to remove during purification.
Furthermore, the mechanism ensures superior impurity control, a critical factor for R&D directors focused on regulatory compliance. The leaving group ability of the tosylate or mesylate anion is excellent, driving the reaction to completion with minimal residual starting material. Unlike older methods that might generate difficult-to-remove metal salts or fluorinated byproducts, this organic transformation yields byproducts that are easily separated during the aqueous workup phase. The high selectivity of the reaction means that the final high-purity polypeptide guanylating agent exhibits liquid phase content often exceeding 99%, reducing the need for extensive chromatographic purification. This mechanistic robustness translates directly into higher overall yields and a cleaner process mass intensity profile, which is essential for sustainable chemical manufacturing.
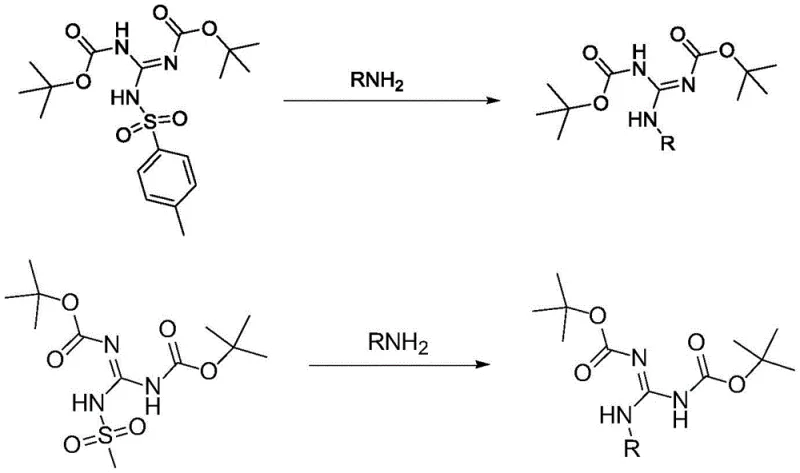
How to Synthesize 1,3-di-Boc-2-(p-toluenesulfonyl) Guanidine Efficiently
Implementing this synthesis requires precise control over stoichiometry and addition rates to maximize the formation of the target sulfonyl guanidine. The process begins by dissolving the bis-Boc guanidine precursor in a solvent such as dichloromethane or tetrahydrofuran, followed by the addition of a tertiary amine base to scavenge the generated acid. The sulfonyl chloride is then introduced dropwise to manage the exotherm, ensuring the temperature remains within the optimal 20-25°C window. Detailed standardized synthetic steps see the guide below.
- Dissolve 1,3-di(tert-butoxycarbonyl) guanidine and an organic base such as triethylamine in a suitable solvent like tetrahydrofuran or dichloromethane.
- Dropwise add p-toluenesulfonyl chloride solution while maintaining the reaction temperature between 0°C and 30°C to ensure controlled sulfonylation.
- Stir the mixture for approximately 2 hours until TLC indicates completion, then concentrate and purify via extraction and filtration to obtain the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits that extend beyond mere chemical novelty. The shift away from specialized, high-cost reagents like triflyl chloride to commodity chemicals like p-toluenesulfonyl chloride fundamentally alters the cost structure of the supply chain. Since the raw materials are widely produced and available from multiple global sources, the risk of supply disruption is significantly minimized. This availability ensures commercial scale-up of complex pharmaceutical intermediates can proceed without the bottlenecks typically associated with niche reagents. Additionally, the elimination of ultra-low temperature requirements reduces energy consumption and infrastructure costs, allowing for more flexible manufacturing scheduling and faster turnaround times.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of expensive fluorinated reagents with inexpensive sulfonyl chlorides. By removing the need for specialized cryogenic cooling systems, facilities can operate with standard thermal control units, leading to substantial savings in both capital investment and ongoing utility expenses. The high yields reported in the patent examples, often surpassing 90%, mean that less raw material is wasted per kilogram of product, further driving down the unit cost. Moreover, the simplified workup procedure reduces solvent usage and labor hours, contributing to a leaner and more cost-effective production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for multinational corporations, and this synthesis route inherently strengthens supply chain resilience. The reliance on common organic solvents and bases means that sourcing is not dependent on single-source suppliers or geopolitically sensitive materials. The robustness of the reaction conditions allows for production in a wider range of facilities, diversifying the manufacturing base and reducing lead time for high-purity pharmaceutical intermediates. This flexibility ensures that even during periods of market volatility, the flow of critical peptide building blocks remains uninterrupted, safeguarding downstream drug development timelines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with modern green chemistry principles. The avoidance of fluorinated waste streams simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The reaction generates solid products that can be isolated via filtration, minimizing the volume of liquid waste requiring disposal. This ease of isolation facilitates seamless scaling from pilot batches to multi-ton production runs without the need for complex process re-engineering. Consequently, manufacturers can meet increasing market demand while adhering to strict environmental regulations, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel guanylating agent technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What are the advantages of this new guanylating agent over traditional triflyl-based reagents?
A: The novel agent avoids the use of highly corrosive trifluoromethanesulfonyl chloride and eliminates the need for ultra-low temperature conditions, significantly reducing operational costs and safety risks.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes common organic solvents and reagents like p-toluenesulfonyl chloride, operates at room temperature, and achieves high yields (up to 94%), making it highly suitable for commercial scale-up.
Q: What is the purity profile of the synthesized intermediate?
A: Experimental data demonstrates liquid phase content exceeding 99%, ensuring high-purity standards required for downstream peptide drug synthesis and minimizing impurity-related failures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polypeptide Guanylating Agent Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced intermediates play in the successful development of next-generation peptide therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications and utilize our rigorous QC labs to verify every batch against the highest international standards. Our capability to implement the efficient sulfonylation techniques described in CN112358423A allows us to offer a competitive advantage in both quality and consistency.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can support your supply chain goals and accelerate your time to market.
