Advanced Synthesis of Active Carbonyl Sialic Acid Derivatives for Commercial Scale-up
The pharmaceutical and biotechnology sectors are increasingly recognizing the critical role of sialic acid derivatives in modulating the pharmacokinetics of therapeutic proteins. Patent CN87108160A discloses a groundbreaking methodology for preparing sialic acid derivatives possessing active carbonyl groups, specifically designed to serve as versatile intermediates for creating complexes with various biomolecules. This innovation addresses a long-standing challenge in glycobiology: the efficient chemical modification of the sialic acid scaffold to introduce reactive handles without compromising the integrity of the sugar ring. By enabling the attachment of sialic acid to amino acids, peptides, and proteins, this technology facilitates the development of half-life extension agents and advanced affinity chromatography media. For R&D teams seeking reliable sialic acid derivative suppliers, understanding the mechanistic depth of this patent is essential for evaluating the feasibility of scaling these complex molecules for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the modification of sialic acid has been hindered by the molecule's inherent instability and the presence of multiple hydroxyl groups that require rigorous protection strategies. Conventional approaches often relied on enzymatic pathways which, while specific, suffered from low throughput, high costs associated with biocatalysts, and difficulties in controlling the degree of substitution. Furthermore, direct chemical functionalization at the anomeric center or the C-2 position frequently resulted in elimination reactions or degradation of the pyranose ring under harsh acidic or basic conditions. The lack of a robust method to introduce a distinct, reactive carbonyl functionality—distinct from the native carboxylic acid—limited the ability of chemists to create stable amide or imine linkages with target proteins. These limitations created a bottleneck in the manufacturing of glycoconjugates, leading to inconsistent purity profiles and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
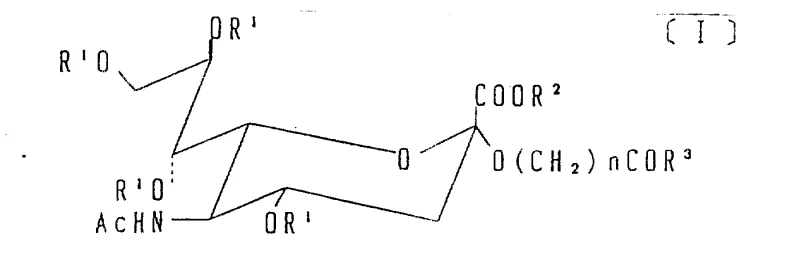
The novel approach detailed in the patent overcomes these hurdles by employing a strategic sequence of protection, substitution, and selective oxidation. Instead of attempting direct oxidation of the sugar ring, the process introduces an unsaturated alkoxy side chain at the C-2 position via nucleophilic substitution of a chloro-intermediate. This unsaturated chain serves as a masked precursor to the active carbonyl group. By utilizing mild oxidative cleavage techniques, such as ozonolysis or permanganate oxidation, the terminal alkene is converted into an aldehyde or carboxylic acid with high regioselectivity. This method preserves the stereochemistry of the sialic acid core while providing a flexible linker arm. The result is a derivative that exhibits high reactivity towards nucleophiles, enabling efficient conjugation under physiological conditions. This chemical logic drastically simplifies the synthesis of complex glycoconjugates, offering a scalable alternative to biological methods.
Mechanistic Insights into Oxidative Cleavage and Activation
The core of this synthetic strategy lies in the precise manipulation of the C-2 substituent. The process begins with the conversion of N-acetylneuraminic acid into a 2-chloro-derivative, where the hydroxyl group is replaced by a chlorine atom, activating the position for nucleophilic attack. An unsaturated alcohol, such as allyl alcohol or oleyl alcohol, then displaces the chlorine atom, installing an alkene tail. The subsequent oxidative cleavage is the critical step that generates the active carbonyl functionality. When ozone is introduced at low temperatures (e.g., -78°C), it reacts specifically with the carbon-carbon double bond to form an ozonide intermediate. Subsequent reductive workup with zinc powder and acetic acid cleaves this intermediate to yield an aldehyde group. Alternatively, oxidative workup or the use of potassium permanganate can drive the reaction further to produce a carboxylic acid. This mechanistic pathway ensures that the reactive group is positioned at the end of a spacer arm, minimizing steric hindrance during subsequent coupling reactions with bulky protein targets.
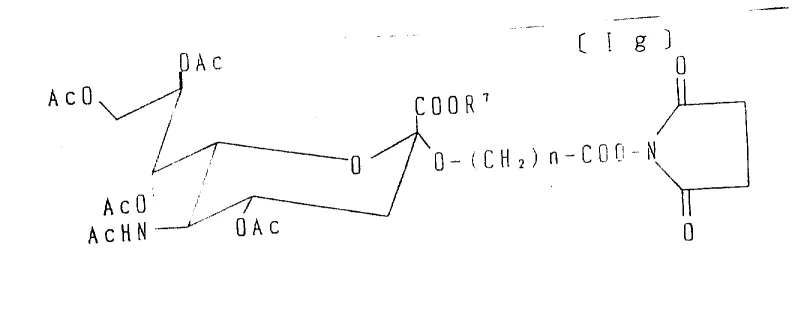
Following the generation of the carboxylic acid derivative, the patent describes a further activation step to create active esters, such as N-hydroxysuccinimide (NHS) esters. This transformation involves reacting the carboxylic acid with N-hydroxysuccinimide in the presence of a condensing agent like DCC or EDC. The resulting NHS ester is highly electrophilic and reacts rapidly with primary amines on lysine residues of proteins or amino acids to form stable amide bonds. This two-stage activation—first creating the acid via oxidation, then converting it to an active ester—provides exceptional control over the conjugation process. It allows manufacturers to produce stable, shelf-ready intermediates that can be stored and shipped before final coupling, thereby enhancing supply chain flexibility. The ability to tune the length of the spacer arm (by varying the unsaturated alcohol) also offers a mechanism to optimize the spatial orientation of the sialic acid on the final conjugate, which is crucial for maintaining biological activity.
How to Synthesize Active Carbonyl Sialic Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires strict adherence to anhydrous conditions and temperature control to prevent side reactions such as elimination or hydrolysis of the acetyl protecting groups. The process generally involves dissolving the protected chloro-sialic acid ester in an aprotic solvent like tetrahydrofuran (THF) and reacting it with the unsaturated alcohol in the presence of a silver salt promoter to facilitate the substitution. Following purification, the oxidative step must be carefully monitored; for ozonolysis, the endpoint is typically determined by the persistence of a blue color in the solution, indicating excess ozone. For procurement teams evaluating cost reduction in pharmaceutical intermediate manufacturing, it is important to note that while ozone generators require capital investment, they eliminate the need for stoichiometric amounts of expensive heavy metal oxidants, reducing waste disposal costs. The detailed standardized synthesis steps see the guide below.
- Esterify the C-2 carboxyl group of N-acetylneuraminic acid and protect hydroxyl groups via acetylation.
- Substitute the C-2 chloro group with an unsaturated alcohol (e.g., allyl or oleyl alcohol) to introduce a reactive alkene side chain.
- Perform oxidative cleavage using ozone or potassium permanganate to convert the terminal alkene into an active aldehyde or carboxylic acid group.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this chemical synthesis route offers substantial strategic advantages for supply chain managers and procurement officers looking to secure reliable sources of complex glycochemicals. Unlike fermentation-based processes that are susceptible to biological contamination and scale-up delays, this fully synthetic pathway utilizes standard unit operations found in most fine chemical facilities. The raw materials, including neuraminic acid derivatives and commodity unsaturated alcohols, are commercially available from multiple global vendors, mitigating the risk of single-source dependency. Furthermore, the modular nature of the synthesis allows for the production of a library of derivatives with different spacer lengths and terminal functional groups from a common intermediate, maximizing asset utilization and reducing inventory complexity.
- Cost Reduction in Manufacturing: The elimination of enzymatic steps and the use of scalable chemical oxidants significantly lowers the cost of goods sold (COGS). By avoiding the purification challenges associated with removing proteins and cellular debris from fermentation broths, the downstream processing becomes more straightforward and less resource-intensive. The high yields reported in the patent examples for the oxidation and activation steps suggest that material throughput can be optimized to minimize waste, directly contributing to improved margin structures for the final API or reagent.
- Enhanced Supply Chain Reliability: Chemical synthesis provides a predictable production timeline that is not subject to the biological variability of cell cultures. The robustness of the acetyl protection strategy ensures that the intermediates are stable enough for storage and transport, allowing for the establishment of safety stocks. This stability is critical for maintaining continuity of supply for long-term projects, such as the development of half-life extended therapeutics, where consistent quality of the sialic acid modifier is paramount for regulatory approval.
- Scalability and Environmental Compliance: The process is amenable to kilogram and ton-scale production using standard stainless steel reactors. While ozonolysis requires specific safety protocols, modern flow chemistry techniques can further enhance the safety and efficiency of this step. Additionally, the byproduct profile is relatively clean, primarily consisting of acetic acid and simple organic fragments, which simplifies wastewater treatment compared to processes generating heavy metal waste. This aligns with increasing global pressure for greener manufacturing practices in the specialty chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sialic acid derivatives. The answers are derived from the specific embodiments and claims within the patent documentation, ensuring accuracy for technical due diligence.
Q: What is the primary advantage of introducing an active carbonyl group to sialic acid?
A: The introduction of an active carbonyl group, such as an aldehyde or activated ester, significantly enhances the reactivity of the sialic acid derivative, allowing it to easily conjugate with amino-containing compounds like proteins and amino acids for half-life extension applications.
Q: How does this synthetic route improve supply chain reliability compared to extraction?
A: This chemical synthesis route relies on standard organic transformations like esterification and oxidation, which utilize widely available raw materials and avoid the batch-to-batch variability and seasonal limitations often associated with biological extraction methods.
Q: Can these derivatives be used for affinity chromatography?
A: Yes, the derivatives containing active esters (such as N-hydroxysuccinimide esters) can be covalently bonded to gel support matrices containing amino groups, creating highly specific affinity chromatography coupling agents for purifying antigens or antibodies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sialic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of glycoconjugate therapeutics depends on the quality and consistency of the starting materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry described in CN87108160A can be translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and functional activity of every batch of sialic acid derivative we produce. Our commitment to quality ensures that your downstream conjugation reactions proceed with maximum efficiency and minimal impurity formation.
We invite you to discuss your specific project requirements with our technical procurement team. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogues, we are ready to support your R&D and commercial goals. Contact us today to explore how our advanced synthesis capabilities can accelerate your time to market for next-generation biopharmaceuticals.
