Advanced Manufacturing of Indane Derivatives for Thromboxane A2 Antagonists
Introduction to Novel Indane Derivative Synthesis
The pharmaceutical landscape continuously demands more efficient pathways to access high-purity active ingredients, particularly for cardiovascular applications. Patent CN1025563C discloses a groundbreaking process for preparing indane derivatives of Formula (I), which serve as potent Thromboxane A2 (TxA2) antagonists. Unlike previous generations of antagonists that suffered from undesirable vasoconstrictive or platelet-aggregating side effects, these novel compounds offer superior therapeutic profiles with excellent antagonistic activity. The core innovation lies in the versatile synthetic route that allows for the precise installation of alkoxy carbonyl or carboxyl-lower alkyl amino groups onto the indane skeleton. This structural modification is critical for enhancing biological efficacy while maintaining metabolic stability. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemistry is essential for securing a robust supply chain for next-generation antithrombotic agents.
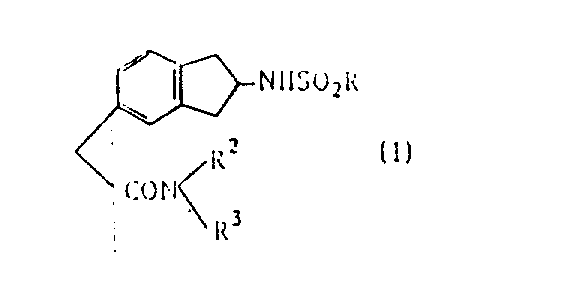
The structural versatility of Formula (I) allows for significant customization, where R represents a substituted or unsubstituted phenyl, naphthyl, or sulfur heterocyclic group. This flexibility enables medicinal chemists to fine-tune the pharmacokinetic properties of the final drug candidate. The process described in the patent not only ensures high chemical purity but also preserves the stereochemical integrity of the asymmetric carbon atoms on the indane ring, which is vital for maintaining specific biological activity. By leveraging this patented methodology, manufacturers can produce complex intermediates that were previously difficult to synthesize with consistent quality, thereby addressing a critical gap in the market for high-performance cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indane-based TxA2 antagonists relied on rigid pathways that often struggled with low overall yields and poor control over stereocenters. Conventional methods frequently involved harsh reaction conditions that could lead to racemization, resulting in a mixture of optically active isomers that complicated downstream purification and reduced the potency of the final API. Furthermore, traditional routes often utilized expensive or hazardous reagents that posed significant challenges for waste management and operator safety in a commercial setting. The inability to easily modify the side chain at the 5-position of the indane ring limited the scope of structure-activity relationship (SAR) studies, slowing down the drug discovery process. These inefficiencies translated into higher production costs and longer lead times, creating bottlenecks for procurement teams aiming to secure cost-effective raw materials for clinical and commercial batches.
The Novel Approach
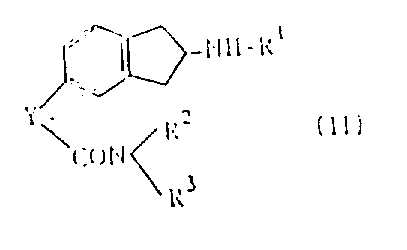
The novel approach detailed in CN1025563C overcomes these hurdles by introducing a modular strategy centered on the reduction of Compound (II) followed by strategic condensation. This method allows for the use of diverse starting materials where the Y group can be a carbonyl, sulfydryl methylene, or halogenated methylene, providing chemists with multiple entry points into the synthesis. By selecting appropriate reduction conditions—such as catalytic hydrogenation for carbonyl groups or acid treatment with zinc for sulfydryl variants—the process achieves high conversion rates without compromising the sensitive indane core. This adaptability significantly simplifies the manufacturing workflow, reducing the number of isolation steps and minimizing solvent consumption. Consequently, this represents a substantial advancement in cost reduction in pharmaceutical intermediate manufacturing, offering a streamlined path from simple precursors to complex, biologically active molecules.
Mechanistic Insights into Catalytic Reduction and Condensation
The heart of this synthetic strategy lies in the selective reduction of the precursor Compound (II), where the nature of the Y group dictates the optimal catalytic system. When Y is a carbonyl group, the patent specifies the use of catalytic hydrogenation in the presence of catalysts such as Raney nickel, cobalt, or palladium, often enhanced by binding substances like oxalic acid. This specific combination facilitates the smooth reduction of the carbonyl functionality to a methylene or hydroxymethylene group under mild temperatures ranging from 10°C to 100°C. Alternatively, when Y represents a phenyl sulfydryl methylene or halogenated methylene group, the process offers the flexibility of using heavy metals like zinc or iron in organic acids such as acetic acid. This dual-pathway capability ensures that regardless of the starting material's electronic properties, the reduction proceeds efficiently, preventing the formation of unwanted by-products that could contaminate the final product.
Following the reduction, the resulting amine intermediate undergoes a condensation reaction with a sulfoacid compound (III) or its reactive derivative, such as a sulfonyl halide. This step is crucial for installing the sulfonamide moiety, which is a key pharmacophore for TxA2 antagonism. The reaction is typically conducted in the presence of an acid acceptor like triethylamine, pyridine, or alkaline carbonates to neutralize the generated acid and drive the equilibrium towards product formation. The patent highlights that this condensation can be performed across a wide temperature range of 0°C to 200°C, allowing for optimization based on the specific reactivity of the sulfonyl chloride used. Importantly, the entire sequence is designed to proceed without racemization, ensuring that if an optically active starting material is used, the final Compound (I) retains its optical purity, a critical quality attribute for regulatory approval.
How to Synthesize Indane Derivatives Efficiently
To implement this synthesis effectively, operators must carefully control the stoichiometry of the reduction and condensation steps to maximize yield and minimize impurities. The process begins with the preparation of the indane precursor, followed by the specific reduction protocol tailored to the Y substituent. Once the reduced amine is isolated, it is immediately subjected to the sulfonylation reaction under controlled pH conditions. Detailed standard operating procedures regarding solvent selection, temperature ramps, and workup protocols are essential for reproducibility. For a comprehensive guide on the exact molar ratios, specific solvent volumes, and purification techniques like silica gel chromatography or recrystallization described in the patent examples, please refer to the standardized synthesis steps provided below.
- Prepare the indane precursor (Compound II) containing a reducible Y group such as carbonyl or sulfydryl methylene.
- Perform reduction using catalytic hydrogenation (Pd/C, Raney Ni) or acid treatment (Zn/Acetic Acid) depending on the Y group functionality.
- Condense the reduced amine intermediate with a sulfonyl chloride or sulfoacid derivative in the presence of an acid acceptor to form the final sulfonamide bond.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers significant strategic advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies. The ability to utilize common, commercially available reagents such as zinc powder, acetic acid, and standard sulfonyl chlorides drastically reduces the dependency on specialized or exotic chemicals that often suffer from supply volatility. This reliance on commodity chemicals ensures a stable supply chain, mitigating the risk of production delays caused by raw material shortages. Furthermore, the high yields reported in the patent examples, such as the 95% yield achieved in the large-scale Example 7, indicate a highly efficient process that minimizes waste and maximizes the output per batch. This efficiency directly translates to lower unit costs, making the final API more competitive in the global marketplace without sacrificing quality.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts in certain variations, replacing them with cost-effective alternatives like zinc and acetic acid. By avoiding complex purification steps associated with heavy metal removal, manufacturers can significantly reduce processing time and consumable costs. The high atom economy of the condensation reaction further contributes to substantial cost savings, as less raw material is lost to side reactions. Additionally, the ability to perform reactions in common solvents like ethyl acetate and methanol simplifies solvent recovery and recycling systems, lowering the overall environmental compliance costs associated with waste disposal.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including 2-aminoindane and various substituted benzenesulfonyl chlorides, are widely produced by multiple chemical suppliers globally. This diversity in the supplier base reduces the risk of single-source dependency, ensuring continuous availability even during market fluctuations. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, means that production is less susceptible to minor variations in raw material specifications. This resilience is crucial for maintaining consistent delivery schedules to downstream pharmaceutical customers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of the process, with examples scaling from milligram laboratory trials to over 100 grams in pilot runs without loss of efficiency. The use of aqueous workups and standard crystallization techniques facilitates easy scale-up to multi-kilogram or ton-scale production in existing manufacturing facilities. Moreover, the process avoids the generation of persistent organic pollutants or highly toxic by-products, aligning with modern green chemistry principles. This environmental compatibility simplifies the permitting process for new production lines and ensures long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indane derivatives. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation. Understanding these details helps stakeholders make informed decisions regarding the integration of this technology into their existing product portfolios. For further technical specifications or custom synthesis requests, our team is available to provide detailed route feasibility assessments.
Q: What is the primary therapeutic application of these indane derivatives?
A: These compounds exhibit potent Thromboxane A2 (TxA2) antagonistic activity, making them valuable for treating thrombosis, embolism, and myocardial ischemia without the vasoconstrictive side effects of earlier antagonists.
Q: How does the new process improve upon conventional synthesis methods?
A: The novel approach allows for flexible reduction conditions (catalytic or chemical) and high-yield condensation steps, avoiding racemization and ensuring high optical purity of the final active pharmaceutical ingredient.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent demonstrates scalability with examples ranging from gram to over 100-gram scales, utilizing standard reagents like zinc powder, acetic acid, and common solvents which are readily available for industrial procurement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular medications. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of handling complex synthetic pathways like the one described in CN1025563C. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to stringent purity specifications guarantees that every batch of indane derivatives meets the highest industry standards, facilitating smoother regulatory filings for your final drug products.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability. Let us be your trusted partner in bringing next-generation TxA2 antagonists to the market.
