Scalable Synthesis of R-(+)-2-p-hydroxyphenoxypropionic Acid via Optimized Mitsunobu Inversion
The global demand for high-efficiency Aryloxy Phenoxy Propionate (APP) herbicides, such as quizalofop-p-ethyl and haloxyfop-R-methyl, continues to drive innovation in the synthesis of their key chiral intermediates. A pivotal advancement in this domain is detailed in patent CN108727187B, which discloses a robust preparation method for (R)-(+)-2-p-hydroxyphenoxypropionic acid (CAS: 94050-90-5). This compound serves as the critical chiral building block for a wide array of gramineous weed control agents. The disclosed technology leverages a strategic three-step sequence starting from the abundant chiral pool material, (S)-(-)-lactic acid. By optimizing reaction conditions and reagent selection, this methodology addresses long-standing challenges in optical purity and yield that have historically plagued the commercial production of this agrochemical intermediate.
For procurement managers and supply chain directors seeking a reliable agrochemical intermediate supplier, understanding the underlying chemistry is essential for assessing long-term viability. The process outlined in the patent not only shortens the synthetic route but also significantly enhances the R/S enantiomeric ratio, ensuring that downstream herbicide formulations meet stringent regulatory standards for biological activity. As the agricultural sector increasingly demands more potent and environmentally compatible crop protection solutions, the ability to source high-purity chiral acids becomes a decisive competitive advantage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-(+)-2-p-hydroxyphenoxypropionic acid has relied on nucleophilic substitution reactions involving halogenated propionic acid derivatives. As illustrated in the conventional pathway below, these methods typically employ alkali metal bases such as sodium ethoxide or potassium hydroxide in industrial ethanol at elevated temperatures around 60°C.
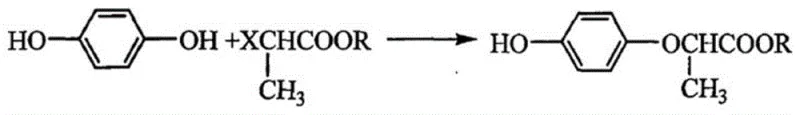
While conceptually straightforward, this traditional approach suffers from significant technical drawbacks that impact both cost and quality. The primary issue is the formation of disubstituted by-products, where the phenolic oxygen reacts twice, creating diether impurities that are chemically similar to the target molecule and notoriously difficult to separate via standard crystallization or distillation. Furthermore, the harsh alkaline conditions required can lead to racemization, degrading the optical purity of the final product. For large-scale manufacturers, these separation challenges translate into lower overall yields and increased waste generation, complicating the cost reduction in herbicide manufacturing initiatives.
The Novel Approach
In stark contrast, the methodology presented in CN108727187B utilizes a Mitsunobu reaction strategy to achieve stereochemical inversion with exceptional precision. Instead of relying on halogen leaving groups, this novel route employs a phosphine/azodicarboxylate system (Ph3P/DEAD) to couple (S)-(-)-lactic acid esters directly with protected hydroquinone derivatives. This approach fundamentally alters the reaction landscape by operating under mild, neutral conditions that preserve the integrity of the chiral center while forcing a complete Walden inversion.
The result is a streamlined three-step process: esterification of the lactic acid, nucleophilic substitution via the Mitsunobu coupling, and final hydrolysis. This sequence eliminates the formation of the problematic diether by-products associated with the older halogen methods. By shifting the paradigm from simple displacement to a concerted inversion mechanism, the process achieves superior control over the impurity profile, making it an ideal candidate for commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Ph3P/DEAD Catalyzed Stereoinversion
The core of this technological breakthrough lies in the meticulous optimization of the Mitsunobu reaction conditions. The mechanism involves the activation of the phenolic hydroxyl group through the formation of an alkoxyphosphonium salt intermediate, which is then attacked by the carboxylate nucleophile in an SN2 fashion. This bimolecular nucleophilic substitution ensures that the stereochemistry at the alpha-carbon is inverted, converting the starting (S)-configuration of the lactic acid derivative into the desired (R)-configuration of the final phenoxypropionic acid.
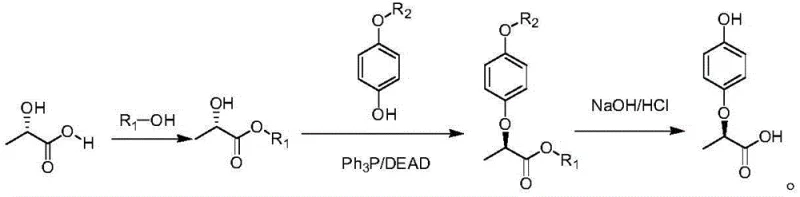
Crucially, the patent data reveals that the steric environment of the ester group plays a pivotal role in determining the success of this inversion. Experimental results indicate that when the ester group (R1) is a bulky moiety such as tert-butyl or benzyl, the R/S value reaches an impressive 99/1. This phenomenon is attributed to the increased steric hindrance provided by these larger groups, which likely stabilizes the transition state favoring inversion and suppresses competing side reactions. In comparison, smaller ester groups like methyl or ethyl yield lower optical purities (92/8 and 93/7 respectively), demonstrating that molecular architecture is a critical lever for quality control.
Furthermore, the protection of the hydroquinone ring is equally vital. Using unprotected hydroquinone (where R2 = H) leads to lower yields due to the presence of two reactive nucleophilic sites. However, masking one site with an acetyl or p-toluenesulfonyl group directs the reaction exclusively to the desired position. The data shows that the p-toluenesulfonyl group offers the optimal balance, delivering the highest yields while maintaining the 99/1 enantiomeric ratio. This level of mechanistic understanding allows process chemists to fine-tune the synthesis for maximum efficiency and minimal waste.
How to Synthesize (R)-(+)-2-p-hydroxyphenoxypropionic acid Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the esterification of (S)-(-)-lactic acid in benzene with anhydrous alcohol and concentrated sulfuric acid at 65°C, followed by the critical Mitsunobu coupling in anhydrous THF at 0°C to room temperature. The final step involves basic hydrolysis using 10% NaOH followed by acidification to isolate the pure acid. This standardized approach ensures reproducibility and high quality.
- Esterify (S)-(-)-lactic acid with anhydrous alcohol (methanol, ethanol, isopropanol, tert-butanol, or benzyl alcohol) using concentrated sulfuric acid catalyst at 65°C.
- Perform nucleophilic substitution using Ph3P/DEAD catalyst system with protected hydroquinone derivatives in anhydrous THF at 0°C to room temperature.
- Hydrolyze the intermediate ester using 10% NaOH aqueous solution followed by acidification with HCl to precipitate the final chiral acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this Mitsunobu-based process offers compelling strategic benefits beyond mere chemical elegance. By addressing the root causes of low yield and poor purity found in legacy methods, this technology unlocks significant operational efficiencies that resonate throughout the supply chain.
- Cost Reduction in Manufacturing: The elimination of difficult-to-separate disubstituted by-products drastically simplifies the downstream purification process. In traditional methods, removing these impurities often requires multiple recrystallizations or expensive chromatography, which drives up processing costs and solvent consumption. By preventing the formation of these by-products at the source, the new route reduces the number of unit operations required, leading to substantial savings in energy, labor, and solvent recovery. Additionally, the high optical purity minimizes the risk of batch rejection, ensuring that every kilogram produced meets specification.
- Enhanced Supply Chain Reliability: The process relies on (S)-(-)-lactic acid, a commodity chemical readily available from fermentation processes, ensuring a stable and secure raw material base. Unlike routes that depend on specialized halogenated intermediates which may face supply volatility, lactic acid is produced globally at massive scales. Furthermore, the robustness of the reaction conditions—operating at mild temperatures and avoiding hazardous reagents—reduces the risk of production delays caused by safety incidents or equipment corrosion, thereby reducing lead time for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: The use of common organic solvents like tetrahydrofuran (THF) and ethyl acetate, which are easily recovered and recycled, aligns well with modern green chemistry principles. The avoidance of heavy metal catalysts or extremely corrosive reagents simplifies waste treatment protocols, lowering the environmental compliance burden. This makes the process highly scalable from kilogram to multi-ton production without requiring exotic infrastructure, facilitating a smoother transition from R&D to commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: Why is the Mitsunobu reaction preferred over traditional halogen substitution for this intermediate?
A: Traditional halogen substitution often leads to disubstituted by-products that are difficult to separate and requires harsh alkaline conditions. The Mitsunobu approach described in CN108727187B ensures complete stereochemical inversion with higher R/S values (up to 99/1) and avoids complex purification steps associated with diether byproducts.
Q: Which ester protecting group yields the highest optical purity?
A: According to the patent data, utilizing bulky ester groups such as tert-butyl or benzyl significantly improves the R/S ratio to 99/1 due to increased steric hindrance which favors the SN2 inversion mechanism, compared to methyl or ethyl esters which yield lower optical purity.
Q: What is the role of the R2 protecting group on the hydroquinone ring?
A: Protecting one hydroxyl group of the hydroquinone (using acetyl or p-toluenesulfonyl groups) prevents the formation of diether by-products. The patent indicates that using 4-toluenesulfonyl provides the highest yield while maintaining excellent optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-(+)-2-p-hydroxyphenoxypropionic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process engineering. Our team specializes in translating complex chiral synthesis routes, such as the Mitsunobu inversion described in CN108727187B, into reliable manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify enantiomeric excess and impurity profiles, guaranteeing that every batch of (R)-(+)-2-p-hydroxyphenoxypropionic acid performs flawlessly in your downstream herbicide synthesis.
We invite you to collaborate with us to optimize your supply chain for APP herbicides. Whether you require custom synthesis development or bulk procurement, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth in the global agrochemical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
