Advanced Manufacturing of 4-Substituted Pyridine Derivatives via Novel Phosphorus Chemistry
The chemical industry continuously seeks robust methodologies for constructing heterocyclic scaffolds, particularly substituted pyridines which serve as critical building blocks in modern drug discovery and crop protection. Patent CN1898255B introduces a transformative approach for the preparation of pyridine derivatives, specifically addressing the longstanding challenge of introducing electron-withdrawing substituents at the 4-position of the pyridine ring. Historically, synthetic efforts often favored the formation of 6-substituted isomers due to thermodynamic stability, leaving a gap in the reliable supply of 4-substituted analogs required for high-performance agrochemicals and pharmaceuticals. This invention leverages the versatility of Wittig and Horner-Wadsworth-Emmons (HWE) reactions coupled with specialized cyclization protocols to overcome these regioselectivity barriers. By utilizing alpha-beta unsaturated carbonyl compounds reacting with tailored phosphorus reagents, the process enables the precise installation of trifluoromethyl and sulfur-containing groups. This technological advancement represents a significant leap forward for manufacturers aiming to secure a stable supply chain for complex heterocyclic intermediates without relying on legacy routes that suffer from poor yield or selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for substituted pyridines, such as the Reformatsky route or various condensation reactions described in earlier literature, often struggle with regiocontrol when electron-withdrawing groups are involved. For instance, attempts to synthesize trifluoromethyl-substituted pyridines frequently result in the predominant formation of 6-substituted isomers rather than the desired 4-substituted targets, necessitating costly and inefficient separation processes. Furthermore, many conventional methods require harsh reaction conditions or expensive transition metal catalysts that introduce impurities difficult to remove to pharmaceutical standards. The reliance on specific starting materials that are not readily available on a commercial scale further exacerbates supply chain vulnerabilities for procurement teams. These legacy processes often lack the flexibility to accommodate diverse substituents at the 3-position, such as sulfur-containing groups, which are increasingly demanded in next-generation herbicide and fungicide designs. Consequently, the industry has faced persistent bottlenecks in scaling up the production of high-purity 4-substituted pyridine derivatives efficiently.
The Novel Approach
The methodology disclosed in CN1898255B circumvents these issues by employing a modular strategy based on phosphorus ylide chemistry. This novel approach utilizes phosphonium salts or phosphonates reacting with alpha-beta unsaturated carbonyl compounds that possess either an amino group or a leaving group at the 4-position. 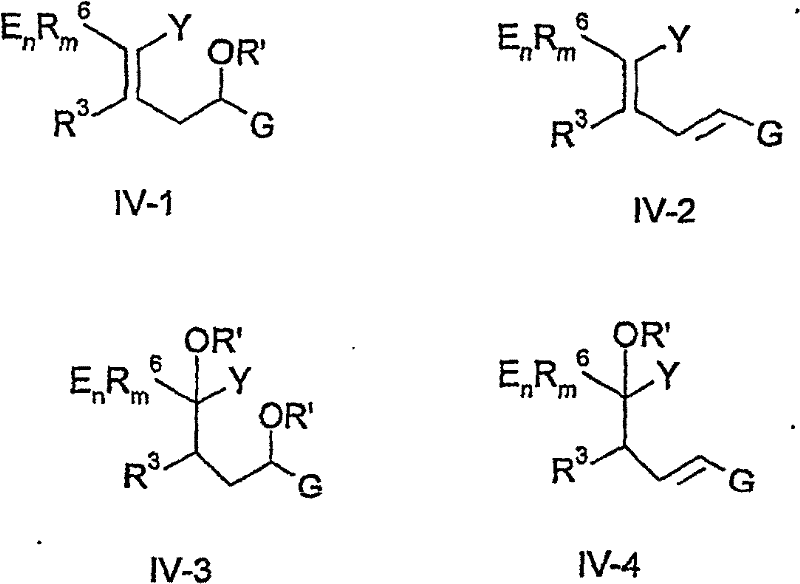 This reaction sequence generates a mixture of intermediates, designated as Formula IV, which can be directly cyclized without the need for rigorous purification, thereby drastically simplifying the operational workflow. The ability to use zeolite catalysts or ammonium salts for the final ring-closing step introduces a level of green chemistry compatibility that is absent in many traditional acid-mediated cyclizations. Moreover, the process demonstrates exceptional tolerance for various electron-withdrawing groups, including perfluorinated alkyl chains and cyano groups, ensuring broad applicability across different chemical portfolios. This flexibility allows manufacturers to adapt the same core platform technology to produce a wide array of derivatives, optimizing asset utilization and reducing the need for dedicated production lines for each specific molecule.
This reaction sequence generates a mixture of intermediates, designated as Formula IV, which can be directly cyclized without the need for rigorous purification, thereby drastically simplifying the operational workflow. The ability to use zeolite catalysts or ammonium salts for the final ring-closing step introduces a level of green chemistry compatibility that is absent in many traditional acid-mediated cyclizations. Moreover, the process demonstrates exceptional tolerance for various electron-withdrawing groups, including perfluorinated alkyl chains and cyano groups, ensuring broad applicability across different chemical portfolios. This flexibility allows manufacturers to adapt the same core platform technology to produce a wide array of derivatives, optimizing asset utilization and reducing the need for dedicated production lines for each specific molecule.
Mechanistic Insights into Wittig-HWE Mediated Cyclization
The core of this innovation lies in the strategic design of the phosphorus reagents and their subsequent reaction with activated enones. The process begins with the generation of Wittig or Horner-Wadsworth-Emmons reagents from phosphorus compounds such as triphenylphosphine or trialkyl phosphites reacting with organic halides. 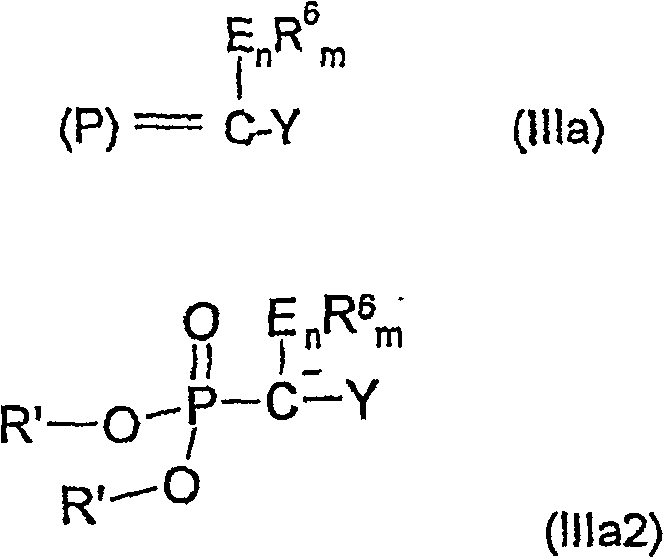 These reagents, characterized by the general Formula IIIa, act as nucleophiles that attack the beta-carbon of the alpha-beta unsaturated carbonyl system. When the carbonyl component contains an amino group at the 4-position, the reaction can proceed directly to the pyridine derivative through an intramolecular condensation mechanism. Alternatively, when a leaving group is present, the reaction forms an open-chain intermediate that requires a subsequent cyclization step. This mechanistic divergence provides chemists with the ability to tune the reaction pathway based on the desired substitution pattern and the stability of the intermediates involved. The use of bases such as alkali metal alcoholates or organolithium compounds ensures the efficient generation of the active ylide species under controlled temperature ranges typically between -15°C and 120°C.
These reagents, characterized by the general Formula IIIa, act as nucleophiles that attack the beta-carbon of the alpha-beta unsaturated carbonyl system. When the carbonyl component contains an amino group at the 4-position, the reaction can proceed directly to the pyridine derivative through an intramolecular condensation mechanism. Alternatively, when a leaving group is present, the reaction forms an open-chain intermediate that requires a subsequent cyclization step. This mechanistic divergence provides chemists with the ability to tune the reaction pathway based on the desired substitution pattern and the stability of the intermediates involved. The use of bases such as alkali metal alcoholates or organolithium compounds ensures the efficient generation of the active ylide species under controlled temperature ranges typically between -15°C and 120°C.
Impurity control is inherently managed through the choice of cyclization catalysts and the nature of the intermediate mixture. In pathways utilizing zeolite catalysts, the heterogeneous nature of the catalyst minimizes the formation of side products associated with homogeneous acid catalysis, such as polymerization or over-alkylation. The intermediate mixture (Formula IV) exists as stereoisomers, but the cyclization conditions are robust enough to drive the equilibrium toward the aromatic pyridine system regardless of the initial stereochemistry. This eliminates the need for expensive chiral resolution steps at the intermediate stage, significantly enhancing the overall mass balance of the process. Furthermore, the final hydrolysis and extraction steps are designed to remove phosphorus byproducts effectively, ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies. The mechanistic understanding of these pathways allows for precise optimization of reaction parameters to minimize waste and maximize throughput.
How to Synthesize 4-Trifluoromethyl Pyridine Efficiently
The synthesis of high-value 4-trifluoromethyl pyridine derivatives follows a streamlined protocol that integrates reagent preparation, coupling, and cyclization into a cohesive manufacturing sequence. The process begins with the in situ or separate preparation of the phosphorus ylide, followed by its addition to the fluorinated enone substrate under inert atmosphere conditions. Detailed standard operating procedures for stoichiometry, temperature control, and workup are critical to ensuring consistent batch-to-batch quality and safety.
- Preparation of Wittig or Horner-Wadsworth-Emmons reagents from phosphorus compounds and suitable organic halides.
- Reaction of the phosphorus reagent with an alpha-beta unsaturated carbonyl compound containing a leaving group or amino group at the 4-position.
- Cyclization of the resulting intermediate mixture using Bronsted acid, zeolite catalysts, or ammonium salts to form the final pyridine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible benefits regarding cost structure and supply reliability. The elimination of transition metal catalysts in the primary bond-forming steps removes the necessity for expensive metal scavenging resins and complex validation protocols associated with heavy metal residues. This simplification of the purification train translates directly into reduced manufacturing costs and shorter cycle times, allowing for more competitive pricing in the global market. Additionally, the robustness of the intermediate mixture means that slight variations in upstream synthesis do not necessarily compromise the final product quality, enhancing the resilience of the supply chain against raw material fluctuations. The ability to utilize continuous flow reactors with zeolite catalysts further opens the door for intensified manufacturing, reducing the physical footprint required for production and lowering utility consumption.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by avoiding the use of precious metal catalysts and minimizing the number of isolation steps required for intermediates. By enabling the direct cyclization of crude intermediate mixtures, the technology reduces solvent consumption and waste disposal costs associated with multiple purification stages. The use of commercially available phosphorus reagents and simple inorganic bases further drives down the raw material cost profile compared to specialized organometallic reagents. This economic efficiency is compounded by the high atom economy of the Wittig-HWE sequence when optimized for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable phosphorus reagents and common organic solvents mitigates the risk of supply disruptions often seen with exotic or highly reactive chemicals. The flexibility to produce a range of derivatives from a common intermediate platform allows manufacturers to respond quickly to changing market demands without retooling entire production facilities. Furthermore, the scalability of the zeolite-catalyzed cyclization step ensures that production volumes can be increased seamlessly from pilot scale to multi-ton commercial campaigns. This reliability is crucial for long-term contracts with agrochemical and pharmaceutical companies that require guaranteed continuity of supply.
- Scalability and Environmental Compliance: The integration of heterogeneous catalysis and the potential for solvent recycling aligns the process with modern environmental, health, and safety (EHS) standards. The reduction in hazardous waste generation, particularly phosphorus-containing byproducts that are managed through standard aqueous workups, simplifies regulatory compliance and permitting. The ability to operate at elevated temperatures with solid catalysts also improves energy efficiency compared to cryogenic conditions required by some alternative lithiation methods. These factors collectively position the technology as a sustainable choice for the long-term manufacturing of complex heterocyclic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis route over traditional Reformatsky methods?
A: This process specifically targets the 4-position of the pyridine ring with electron-withdrawing groups like trifluoromethyl, which is historically difficult to achieve compared to 6-substitution. It utilizes versatile Wittig or Horner-Wadsworth-Emmons chemistry that allows for better control over regioselectivity and avoids the harsh conditions often associated with older cyclization methods.
Q: Can the intermediates in this process be isolated before cyclization?
A: Yes, the process generates intermediate mixtures (Formula IV) that can be isolated via distillation or chromatography if high purity is required for specific downstream applications. However, the patent highlights that separation is not strictly necessary, as the mixture can undergo direct cyclization, which significantly streamlines the manufacturing workflow and reduces processing time.
Q: What types of catalysts are suitable for the final ring-closing step?
A: The technology supports multiple cyclization strategies including Bronsted acid catalysis, ammonium salt promotion, and heterogeneous zeolite catalysis. The use of boron-doped zeolite catalysts at elevated temperatures is particularly notable for enabling continuous flow processing and simplifying product purification through heterogeneous catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethyl Pyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the one described in CN1898255B to deliver high-quality intermediates for the global life sciences industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets your exact requirements. Our commitment to technical excellence allows us to tackle complex molecular architectures, including those with sensitive fluorinated motifs and sulfur functionalities, with confidence and precision.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your trusted partner in supply chain optimization and product development.
