Advanced Manufacturing of 2-Substituted-5-Alkylthio Pyridines for Global Agrochemical Supply Chains
The chemical landscape for agrochemical and pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, scalable, and cost-effective synthetic routes. Patent CN101547901A introduces a significant advancement in the preparation of 2-substituted-5-((1-alkylthio)alkyl)pyridines, a structural motif highly valued in the development of next-generation pesticides and bioactive molecules. As referenced in related art such as US Patent Application Publication 2005/0228027, these pyridine derivatives serve as critical building blocks. The disclosed methodology offers a streamlined approach that bypasses the limitations of older, less efficient syntheses, providing a robust pathway for industrial manufacturers. By leveraging a condensation strategy between substituted enones and enamines, followed by selective functionalization, this process ensures high yields and operational simplicity.
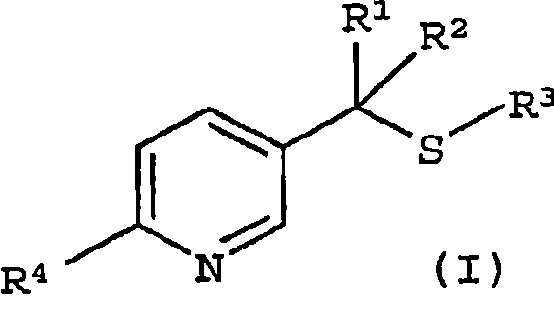
For R&D directors and process chemists, understanding the nuances of this transformation is vital. The ability to introduce specific alkyl and haloalkyl groups at the 2 and 5 positions of the pyridine ring allows for fine-tuning of biological activity and physicochemical properties. This patent details a versatile protocol where R1 and R2 can be hydrogen or alkyl groups, and R4 can be a haloalkyl moiety like trifluoromethyl, expanding the chemical space accessible to medicinal and agricultural chemists. The integration of this technology into existing supply chains represents a strategic opportunity for reliable agrochemical intermediate suppliers to enhance their portfolio with high-value, difficult-to-make scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing substituted pyridines often suffer from poor regioselectivity, requiring complex protection-deprotection sequences that drive up costs and reduce overall throughput. Many legacy processes rely on harsh cyclization conditions or expensive transition metal catalysts that leave behind trace impurities, complicating downstream purification. Furthermore, introducing sulfur functionality at specific benzylic positions on a pyridine ring has historically been challenging due to the susceptibility of the heterocycle to oxidation or undesired side reactions. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher volatility in pricing for key intermediates. For procurement managers, these factors translate into unpredictable availability and increased raw material costs, hindering the economic viability of final active ingredient production.
The Novel Approach
The methodology described in CN101547901A addresses these pain points through a clever three-step sequence that maximizes atom economy and operational ease. The process begins with the condensation of a substituted enone with an enamine, a reaction that proceeds smoothly under mild conditions to form the pyridine core. This is followed by a highly selective radical halogenation step, utilizing reagents like N-bromosuccinimide (NBS) to functionalize the alkyl side chain without damaging the aromatic ring. Finally, a nucleophilic substitution with a thiolate salt installs the desired sulfur group with high fidelity. This route eliminates the need for exotic catalysts and allows for the use of commodity chemicals, significantly simplifying the manufacturing workflow. The result is a process that is not only chemically elegant but also commercially superior, offering substantial cost savings and improved supply chain reliability.
Mechanistic Insights into Enamine-Enone Condensation and Functionalization
The core of this synthetic strategy lies in the initial construction of the pyridine ring via a variation of the Hantzsch pyridine synthesis. The reaction between the alpha,beta-unsaturated ketone (II) and the enamine (III) generates a dihydropyridine intermediate, which subsequently undergoes aromatization in the presence of an ammonia source. This step is critical, as the choice of ammonia generator, such as ammonium acetate or formamide, influences the rate of cyclization and the purity of the resulting 2,5-disubstituted pyridine (IV). The use of polar, high-boiling solvents like acetonitrile or dimethylformamide ensures that all reactants remain in solution, facilitating efficient molecular collisions and heat transfer during the exothermic cyclization phase.
Following ring formation, the process employs a radical mechanism for halogenation. By using N-bromosuccinimide in conjunction with a Lewis acid catalyst like zirconium tetrachloride, the reaction selectively targets the benzylic hydrogen of the alkyl group at the 5-position. This selectivity is paramount for maintaining the integrity of the trifluoromethyl or other sensitive groups at the 2-position. The resulting haloalkylpyridine (V) is then primed for nucleophilic attack. In the final step, a metal thiolate acts as a soft nucleophile, displacing the halide to form the stable carbon-sulfur bond. This sequence demonstrates a deep understanding of physical organic chemistry, leveraging electronic effects to achieve transformations that are otherwise difficult to control.
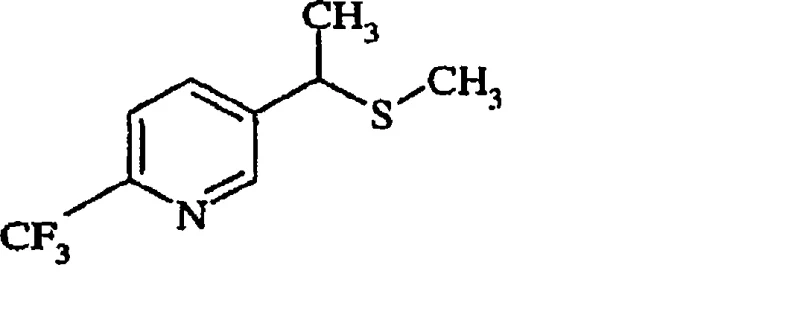
How to Synthesize 2-Substituted-5-Alkylthio Pyridines Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control and stoichiometry. The patent outlines a clear procedure starting from simple aldehydes and amines to generate the necessary enamine in situ or as a pre-formed reagent. The subsequent cyclization is typically conducted at temperatures ranging from 0°C to 80°C, allowing for flexibility depending on the specific substituents involved. For the halogenation step, maintaining anhydrous conditions is essential to prevent hydrolysis of the succinimide byproduct and ensure high conversion. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results at scale.
- Preparation of Enamine: React substituted propionaldehyde with a secondary amine (e.g., pyrrolidine) in the presence of a desiccant like potassium carbonate at 0°C to 20°C.
- Pyridine Ring Formation: Condense the prepared enamine with an alpha,beta-unsaturated ketone (enone) in a polar solvent like acetonitrile, followed by cyclization using an ammonia source such as ammonium acetate.
- Halogenation and Thiolation: Subject the resulting 2,5-disubstituted pyridine to radical halogenation using N-bromosuccinimide (NBS), followed by nucleophilic substitution with a metal thiolate to install the alkylthio group.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers distinct advantages that align with the goals of cost reduction and supply security. By utilizing widely available starting materials such as pyrrolidine, butyraldehyde, and commodity solvents, the dependency on specialized or scarce reagents is minimized. This accessibility translates directly into lower raw material costs and reduced risk of supply disruptions. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, streamlining the purification process and reducing waste disposal costs. These factors combine to create a manufacturing profile that is both economically attractive and environmentally sustainable.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by avoiding the use of precious metal catalysts and complex protecting group strategies. The high yields reported in the examples, such as the 93% yield in the final thiolation step, indicate minimal material loss, which directly improves the cost of goods sold. Additionally, the ability to isolate intermediates via simple distillation or crystallization reduces the reliance on costly chromatographic purification methods typically required for complex heterocycles.
- Enhanced Supply Chain Reliability: Because the synthesis relies on bulk chemicals that are produced globally in large volumes, the supply chain is inherently more resilient. There is no single point of failure associated with a proprietary catalyst or a sole-source reagent. This diversification of supply sources ensures consistent availability of the intermediate, allowing downstream manufacturers to plan production schedules with greater confidence and reduced lead time variability.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate within standard temperature ranges suitable for large-scale reactors. The use of common solvents like ethanol and ethyl acetate simplifies solvent recovery and recycling programs, contributing to a lower environmental footprint. The process generates manageable waste streams, primarily consisting of succinimide and inorganic salts, which are easier to treat than heavy metal-contaminated waste, ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific product portfolios.
Q: What are the key advantages of this pyridine synthesis method over conventional routes?
A: This process utilizes readily available enamines and enones to construct the pyridine core efficiently. It avoids harsh conditions often associated with traditional heterocycle synthesis, offering high yields (up to 93% in final steps) and excellent selectivity during the halogenation phase.
Q: Can this process be scaled for industrial production of agrochemical intermediates?
A: Yes, the reaction conditions are robust, operating between -20°C and 150°C using common solvents like acetonitrile and ethanol. The use of standard reagents like N-bromosuccinimide and sodium methyl mercaptide facilitates straightforward scale-up from laboratory to commercial tonnage.
Q: How is purity controlled in the final alkylthio-pyridine product?
A: Purity is managed through precise control of the radical halogenation step using Lewis acid catalysts like zirconium tetrachloride, which minimizes over-halogenation. Final purification is achieved via standard techniques such as vacuum distillation or silica gel chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted-5-Alkylthio Pyridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-substituted-5-alkylthio pyridines meets the exacting standards required by the global agrochemical and pharmaceutical industries. Our commitment to quality ensures that your downstream processes run smoothly without unexpected interruptions due to impurity profiles.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and value in your supply chain.
