Advanced Synthesis of Racemic Hydroxy Methionine Calcium for Pharmaceutical and Feed Applications
Advanced Synthesis of Racemic Hydroxy Methionine Calcium for Pharmaceutical and Feed Applications
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and high-purity intermediates to ensure the efficacy and safety of final therapeutic products. A significant breakthrough in this domain is detailed in patent CN112645857A, which outlines a novel preparation method for racemic hydroxy methionine calcium. This compound serves as a critical precursor in the synthesis of compound alpha-keto acid tablets, widely utilized for treating chronic renal insufficiency and uremia. The patented technology addresses long-standing challenges in the synthesis of acyclic organic compounds, specifically focusing on improving yield, purity, and process safety. By leveraging a controlled diazotization strategy followed by a sophisticated purification regimen, this method delivers a product with purity exceeding 97.0% and a substantial yield improvement over prior art. For global procurement teams and R&D directors, this represents a viable pathway for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality standards.
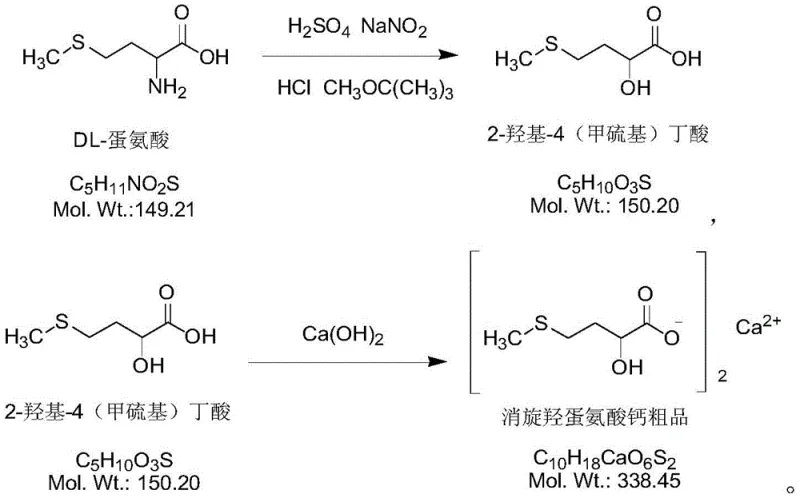
The core innovation lies in the meticulous control of reaction parameters and the implementation of a multi-step purification protocol that effectively removes trace impurities. Unlike traditional methods that often suffer from complex operational controls and safety hazards, this approach utilizes nitrogen protection and precise temperature regulation to stabilize the reaction environment. The process begins with the conversion of DL-methionine into 2-hydroxy-4-methylthio-butyric acid, followed by salt formation with calcium hydroxide. The subsequent refining steps involve acidification, dissociation, and re-salt formation, ensuring that the final crystalline product meets the rigorous specifications required for medical and feed applications. This technological advancement not only enhances product quality but also aligns with modern green chemistry principles by enabling solvent recycling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of racemic calcium hydroxymethionine has been plagued by significant operational inefficiencies and safety concerns. Conventional protocols typically involve reacting DL-methionine with dilute sulfuric acid and sodium nitrite under heating, followed by extraction with methyl tert-butyl ether. These legacy methods are characterized by multiple reaction steps that are difficult to control, often resulting in low product content and high levels of impurities. Furthermore, the diazotization reaction, if not managed with extreme precision, poses inherent safety risks due to the potential for exothermic runaway and the generation of unstable intermediates. The complexity of the post-reaction workup, including repeated acidification and salt formation cycles without effective impurity removal strategies, leads to considerable yield losses. Consequently, these factors contribute to relatively high production costs and make large-scale industrial production challenging and economically unviable for many manufacturers.
The Novel Approach
In stark contrast, the method disclosed in patent CN112645857A introduces a streamlined and safer workflow that fundamentally reshapes the production landscape. By employing concentrated sulfuric acid and controlling the addition of sodium nitrite as a slow drip under a nitrogen atmosphere, the process effectively mitigates the safety risks associated with diazotization. The introduction of specific extraction agents, such as butanone, in the early stages allows for the selective removal of impurities before the final salt formation, drastically improving the quality of the intermediate. Moreover, the ability to recycle organic solvents like butanone and ethyl acetate for multiple batches significantly reduces waste generation and raw material consumption. This novel approach ensures that the reaction conditions remain mild and easy to control, facilitating a smoother transition from laboratory scale to commercial manufacturing while maintaining high purity and yield.
Mechanistic Insights into Diazotization and Salt Formation
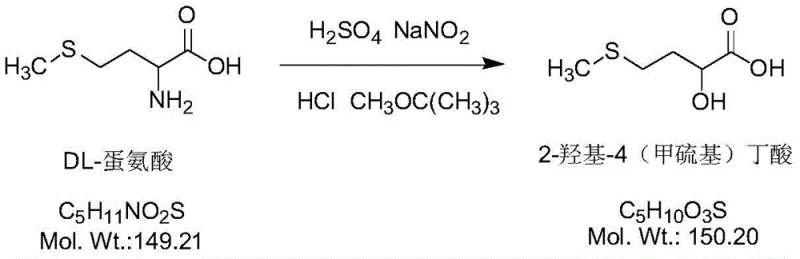
The chemical transformation at the heart of this process is the conversion of the amino group in DL-methionine to a hydroxyl group via a diazonium intermediate. This reaction is initiated by the interaction of DL-methionine with sodium nitrite in an acidic medium provided by concentrated sulfuric acid. The mechanism proceeds through the formation of a diazonium salt, which is inherently unstable and must be handled with care. In this patented method, the reaction is conducted at a controlled temperature range of 78-84°C, which is optimal for the decomposition of the diazonium species into the desired carbocation and subsequent nucleophilic attack by water to form the hydroxyl group. The use of nitrogen protection throughout this stage is critical to prevent oxidation side reactions that could lead to the formation of sulfones or other oxidized sulfur byproducts, thereby preserving the integrity of the thioether moiety.
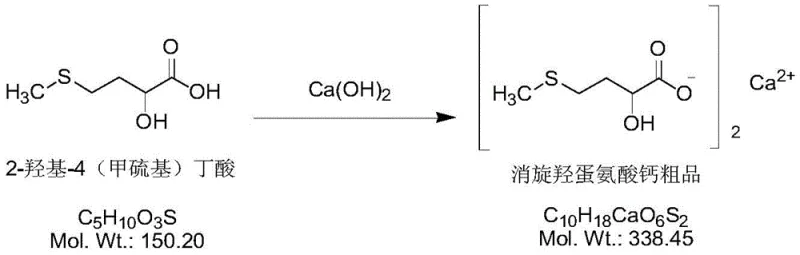
Following the formation of 2-hydroxy-4-methylthio-butyric acid, the process moves to the salt formation stage, which is equally critical for defining the physical properties of the final product. The crude acid is reacted with calcium hydroxide emulsion at a moderate temperature of 40-60°C. The pH is meticulously adjusted to a range of 6.5-7.5 to ensure complete neutralization without causing hydrolysis of the ester or other sensitive functional groups. The addition of methanol during the reflux stage serves to decrease the solubility of the calcium salt, promoting crystallization and facilitating the exclusion of soluble impurities from the crystal lattice. This precise control over the crystallization environment is what enables the production of a high-purity solid with consistent particle size distribution, a key parameter for downstream formulation in pharmaceutical tablets.
How to Synthesize Racemic Hydroxy Methionine Calcium Efficiently
The synthesis of this high-value intermediate requires a disciplined adherence to the optimized parameters outlined in the patent to ensure reproducibility and quality. The process is divided into three distinct phases: the preparation of the hydroxy acid, the formation of the crude calcium salt, and the final refining step. Each phase incorporates specific extraction and purification techniques designed to maximize yield while minimizing impurity carryover. The use of butanone for initial impurity removal and ethyl acetate for final extraction creates a orthogonal purification strategy that effectively cleans the product stream. For R&D teams looking to implement this commercial scale-up of complex pharmaceutical intermediates, understanding the nuances of pH control and solvent selection is paramount to success.
- Prepare racemic hydroxymethionine by reacting DL-methionine with sodium nitrite and sulfuric acid under nitrogen protection at 78-84°C, followed by pH adjustment and solvent extraction.
- Form the crude calcium salt by reacting the hydroxymethionine with calcium hydroxide emulsion at 40-60°C, followed by methanol reflux and crystallization.
- Refine the crude product by acidification, extraction with methyl tert-butyl ether, decolorization, and re-salt formation with calcium hydroxide to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the significant reduction of manufacturing costs driven by the efficient recycling of organic solvents. By recovering and reusing solvents like butanone and ethyl acetate, the process drastically lowers the volume of hazardous waste requiring disposal and reduces the recurring expense of purchasing fresh solvents. This circular approach to solvent management not only aligns with environmental sustainability goals but also insulates the production cost from volatility in raw material markets. Furthermore, the simplified refining steps reduce the overall processing time and labor intensity, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of complex and hazardous reaction conditions translates directly into lower operational expenditures. By avoiding the need for specialized high-pressure equipment or extreme cryogenic conditions often required in alternative syntheses, capital investment and maintenance costs are substantially reduced. The high selectivity of the reaction minimizes the formation of byproducts, which in turn reduces the load on purification units and lowers the consumption of auxiliary chemicals. This efficiency gain allows for a more competitive pricing structure for the final high-purity pharmaceutical intermediate, providing a strategic advantage in cost-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of the process ensures a consistent and reliable supply of material, which is critical for maintaining uninterrupted production schedules for downstream drug manufacturers. The use of readily available raw materials such as DL-methionine and common mineral acids reduces the risk of supply bottlenecks associated with exotic reagents. Additionally, the mild reaction conditions and straightforward workup procedures facilitate easier technology transfer and scale-up, ensuring that production capacity can be rapidly expanded to meet surging demand without compromising quality or safety standards.
- Scalability and Environmental Compliance: From an environmental perspective, the process is designed to minimize the generation of wastewater and organic waste, simplifying compliance with increasingly stringent environmental regulations. The ability to recycle solvents and the reduction in side reactions mean that the effluent load is significantly lower compared to conventional methods. This eco-friendly profile not only reduces the cost of waste treatment but also enhances the corporate social responsibility profile of the supply chain, making it an attractive partner for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of racemic hydroxy methionine calcium. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains.
Q: What is the purity level of racemic hydroxy methionine calcium produced by this method?
A: The patented process consistently achieves a product purity exceeding 97.0%, with total impurities strictly controlled below 0.7% through multi-stage extraction and recrystallization.
Q: How does this method improve safety compared to conventional diazotization?
A: Safety is enhanced by slowly dripping sodium nitrite in solution form under nitrogen protection, which mitigates the exothermic risks and potential runaway reactions associated with traditional batch diazotization.
Q: Can the organic solvents used in extraction be recycled?
A: Yes, the process utilizes solvents like butanone and ethyl acetate which can be recovered and recycled for multiple batches, significantly reducing waste generation and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Hydroxy Methionine Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications and advanced nutritional products. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our dedication to process optimization allows us to offer a reliable supply of complex intermediates that adhere to the highest international standards.
We invite you to collaborate with us to explore how our advanced manufacturing capabilities can support your project goals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in cost reduction in pharmaceutical intermediate manufacturing can add value to your supply chain.
