Advanced PMS Synthesis Route for High-Purity Cosmetic Intermediates Manufacturing
The landscape of bioactive lipid synthesis is undergoing a significant transformation with the introduction of patent CN112341354A, which details a robust and efficient method for synthesizing PMS, a critical Pseudoceramide compound. As the demand for advanced skin barrier repair agents surges in the global cosmetics and dermatological sectors, the ability to produce high-purity pseudoceramide intermediates with consistent quality has become a paramount challenge for manufacturers. This patent addresses these challenges by introducing a novel synthetic pathway that leverages strategic protective group chemistry to overcome the inherent limitations of traditional serine derivatization. By utilizing L-serine methyl ester hydrochloride as a foundational building block, the process ensures a cost-effective entry point while maintaining rigorous control over stereochemistry and functional group reactivity throughout the multi-step sequence. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards more reliable cosmetic intermediate supplier capabilities, offering a pathway to secure supply chains for high-value moisturizing agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ceramide analogues like PMS has been plagued by issues related to regioselectivity and purification complexity. In conventional approaches, attempting to directly esterify the carboxylic acid of serine derivatives with long-chain alcohols often results in competitive reactions at the free hydroxyl group on the serine backbone. This lack of chemical discrimination leads to the formation of unwanted by-products, such as O-acylated species, which are structurally similar to the target molecule and notoriously difficult to separate. These impurities not only drag down the overall yield but also compromise the biological efficacy of the final cosmetic ingredient, necessitating expensive and time-consuming chromatographic purification steps. Furthermore, many existing methods rely on harsh reaction conditions or exotic reagents that are difficult to source at scale, creating bottlenecks for cost reduction in cosmetic ingredient manufacturing and limiting the ability to meet the volumetric demands of the personal care industry.
The Novel Approach
The methodology outlined in CN112341354A elegantly circumvents these pitfalls through a strategic benzyl protection strategy that guarantees reaction specificity. By temporarily masking the hydroxyl group of the serine precursor with a benzyl moiety prior to the critical esterification step, the synthesis effectively shuts down competing side reactions, directing the chemical transformation exclusively towards the formation of the desired ester bond with tetradecanol. This approach not only drastically simplifies the reaction profile but also enhances the overall conversion rate, allowing for the isolation of intermediates with exceptional purity using standard workup procedures like crystallization and washing. The result is a streamlined process that minimizes waste generation and maximizes throughput, providing a clear advantage for companies seeking the commercial scale-up of complex lipid intermediates without the burden of extensive downstream processing.
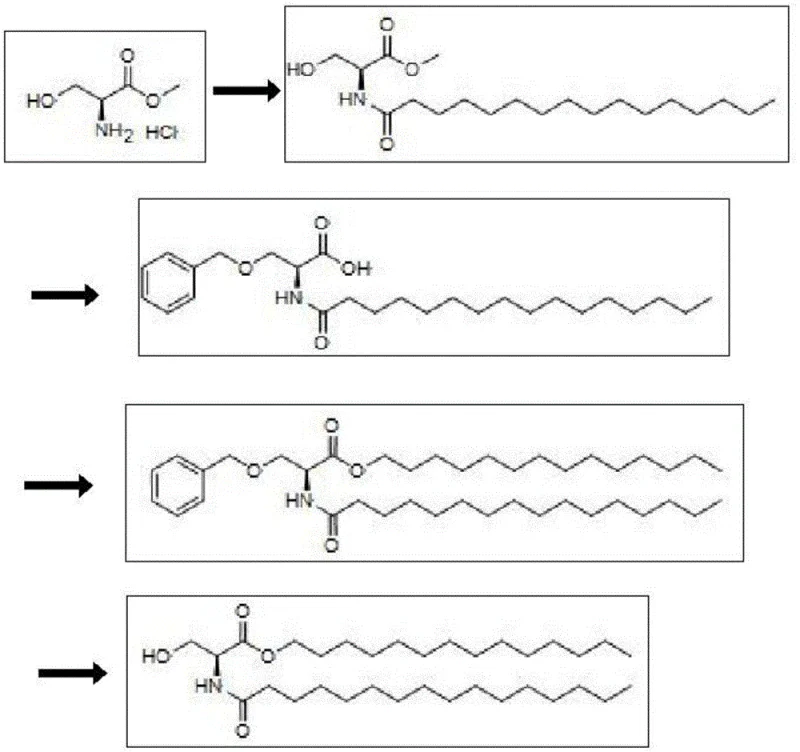
Mechanistic Insights into Benzyl-Protected Serine Derivatization
The core of this synthetic innovation lies in the precise orchestration of acylation and protection-deprotection cycles that preserve the integrity of the chiral center while modifying the peripheral functional groups. The process initiates with the acylation of L-serine methyl ester hydrochloride using palmitoyl chloride, a reaction facilitated by an acid-binding agent such as triethylamine to neutralize the generated HCl and drive the equilibrium forward. Following this, the introduction of the benzyl group via nucleophilic substitution with a benzyl halide in the presence of a strong base like sodium hydride creates a robust ether linkage that withstands the subsequent acidic and basic conditions of the esterification phase. This stability is crucial, as it allows the carboxylic acid to be activated, typically via conversion to an acyl chloride using oxalyl chloride, and subsequently reacted with tetradecanol without fear of degrading the protecting group or racemizing the alpha-carbon.
Furthermore, the final deprotection step demonstrates remarkable chemoselectivity, utilizing palladium on carbon catalysis to cleave the benzyl ether under mild hydrogenolytic conditions or transfer hydrogenation with ammonium formate. This specific choice of deprotection chemistry is vital for maintaining the stability of the newly formed ester bond and the amide linkage, both of which could be susceptible to hydrolysis under more vigorous acidic or basic deprotection conditions. By operating at moderate temperatures ranging from 10°C to 60°C, the process minimizes thermal degradation risks, ensuring that the final PMS product retains its structural fidelity and bioactivity. For technical teams, understanding this mechanistic nuance is key to troubleshooting and optimizing the process, as it highlights the importance of catalyst loading and hydrogen source selection in achieving the high yields reported in the patent examples.
How to Synthesize PMS Efficiently
The synthesis of PMS described in this patent offers a reproducible framework for laboratory and pilot-scale production, relying on standard organic synthesis techniques that are easily transferable to larger reactors. The protocol emphasizes the importance of stoichiometric control, particularly in the acylation and protection steps, where slight excesses of reagents like palmitoyl chloride and benzyl bromide are used to ensure complete consumption of the valuable serine starting material. Detailed operational parameters, including solvent choices like tetrahydrofuran and dichloromethane, are optimized to balance solubility and reaction kinetics, ensuring that the viscous long-chain intermediates remain in solution for efficient mixing and heat transfer. While the specific experimental details provide a solid foundation, operators must adhere to strict safety protocols when handling reagents like sodium hydride and oxalyl chloride, and the following guide outlines the standardized workflow for implementation.
- Acylation of L-serine methyl ester hydrochloride with palmitoyl chloride in the presence of an acid-binding agent to form N-palmitoyl-L-serine methyl ester.
- Protection of the hydroxyl group using a benzyl halide followed by hydrolysis of the methyl ester to yield N-palmitoyl-O-benzyl-L-serine.
- Esterification of the protected serine derivative with tetradecanol using an acylating reagent to form the tetradecyl ester intermediate.
- Catalytic deprotection of the benzyl group using palladium on carbon and a hydrogen source to obtain the final PMS product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemicals sector. The reliance on commodity chemicals such as L-serine methyl ester hydrochloride and palmitoyl chloride means that raw material costs are inherently lower and supply security is higher compared to routes requiring specialized, low-volume precursors. Additionally, the high selectivity of the benzyl protection strategy translates into substantial cost savings by eliminating the need for complex purification technologies like preparative HPLC, which are often the primary cost drivers in the manufacture of high-purity pseudoceramide. The ability to isolate products via simple crystallization or extraction significantly reduces solvent consumption and waste disposal costs, aligning with modern sustainability goals while improving the overall margin profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the early stages and the use of readily available acid-binding agents contribute to a leaner cost structure. By avoiding the formation of difficult-to-separate isomers, the process reduces the loss of valuable intermediates during purification, effectively increasing the mass balance and lowering the cost per kilogram of the final active ingredient. This efficiency is critical for maintaining competitiveness in the price-sensitive cosmetic ingredients market, where margin compression is a constant threat.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which often proceed at room temperature, reduces the energy footprint and the risk of batch failures due to thermal runaway or equipment malfunction. This reliability ensures reducing lead time for high-purity cosmetic actives, as production schedules can be maintained with greater predictability. Furthermore, the use of common solvents and reagents mitigates the risk of supply disruptions caused by the scarcity of niche chemicals, providing a more resilient supply chain for long-term production contracts.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in multipurpose chemical plants, such as stirred tank reactors and filtration units. The mild deprotection conditions and the potential to recycle solvents like dichloromethane and methanol support environmental compliance by minimizing volatile organic compound (VOC) emissions. This ease of scale-up allows manufacturers to respond rapidly to market demand fluctuations, moving from kilogram-scale development to multi-ton commercial production without the need for significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this PMS synthesis technology, drawing directly from the patent specifications and practical application scenarios. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this route for their own production lines or sourcing strategies. The answers provided reflect the specific advantages of the benzyl protection methodology and its impact on product quality and process economics.
Q: Why is benzyl protection critical in the synthesis of PMS?
A: Benzyl protection is essential to prevent side reactions at the hydroxyl group of the serine backbone during the esterification step with tetradecanol. By masking the hydroxyl group, the reaction becomes highly regioselective for the carboxylic acid, significantly improving conversion rates and simplifying downstream purification.
Q: What are the key raw materials required for this PMS synthesis route?
A: The process utilizes commercially available and cost-effective starting materials, primarily L-serine methyl ester hydrochloride, palmitoyl chloride, benzyl halides (such as benzyl bromide), and tetradecanol (myristyl alcohol), making it highly suitable for industrial sourcing.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method employs mild reaction conditions, often at room temperature, and avoids extreme pressures or temperatures. The high yields reported in the examples (exceeding 90% in key steps) and the use of standard reagents indicate strong potential for commercial scale-up of complex lipid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PMS Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN112341354A for the production of high-performance skincare actives. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of PMS meets the exacting standards required for topical pharmaceutical and premium cosmetic applications. We are committed to bridging the gap between innovative patent chemistry and industrial reality, delivering solutions that enhance product performance while optimizing manufacturing costs.
We invite you to collaborate with us to leverage this advanced synthetic route for your next product launch. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient process can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the dynamic market for bioactive lipid ingredients.
