Advanced Production Method For Pyrrolidinylthio Carbapenem Derivatives And Commercial Scale-Up
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for agents with broader antimicrobial spectra and reduced toxicity profiles. Patent CN1032257C introduces a sophisticated production method for pyrrolidinylthio carbapenem derivatives, representing a significant advancement in the field of antibiotic synthesis. This technology addresses critical challenges in the manufacture of carbapenems by providing a robust route to novel intermediates that enhance both biological efficacy and chemical stability. The disclosed compounds feature a unique pyrrolidine ring linked via a sulfur atom to the carbapenem core, a structural motif that has been shown to improve pharmacokinetic properties. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable carbapenem intermediate supplier capable of meeting stringent quality standards.
The core innovation lies in the precise manipulation of the pyrrolidine side chain. By starting from 4-hydroxyl pyrrolidine-2-carboxylic acid derivatives, the method systematically transforms functional groups to introduce the necessary sulfamoylamino methyl moiety while maintaining strict stereochemical control. This level of precision is essential for ensuring the final API possesses the desired biological activity. The patent details multiple approaches to achieve this transformation, utilizing various protecting groups such as p-nitrobenzyloxycarbonyl (Pnz), tert-butyloxycarbonyl (Boc), and allyloxycarbonyl (Alz). These protecting groups are not merely incidental; they are strategic choices that facilitate purification and prevent unwanted side reactions during the complex multi-step synthesis.
Traditional methods for synthesizing carbapenem derivatives often suffer from low overall yields due to the sensitivity of the beta-lactam ring and the complexity of introducing sulfur-linked side chains. Conventional routes may require harsh conditions that degrade the core structure or result in difficult-to-separate diastereomers. In contrast, the novel approach outlined in the patent mitigates these risks by separating the construction of the side chain from the final coupling step. This modular strategy allows for the optimization of the pyrrolidine intermediate independently before it is introduced to the sensitive carbapenem nucleus. By isolating the synthesis of the thiol-containing pyrrolidine derivative, manufacturers can ensure high purity of the side chain, which directly translates to improved quality of the final antibiotic product.
The novel approach leverages a series of well-defined chemical transformations that are amenable to scale-up. Key steps include the conversion of the 4-position hydroxyl group into a leaving group, such as a mesylate or tosylate, followed by nucleophilic substitution with a sulfur source like potassium thioacetate or triphenylmethanethiol. Subsequent reduction of the 2-position ester to a hydroxymethyl group, followed by conversion to an amine and finally sulfamoylation, creates the complex side chain with high fidelity. This sequence avoids the use of unstable intermediates that plague older methodologies. Furthermore, the final coupling reaction utilizes activated carbapenem derivatives, such as diphenylphosphoryl esters, which react efficiently with the thiol side chain under mild conditions, preserving the integrity of the beta-lactam ring.
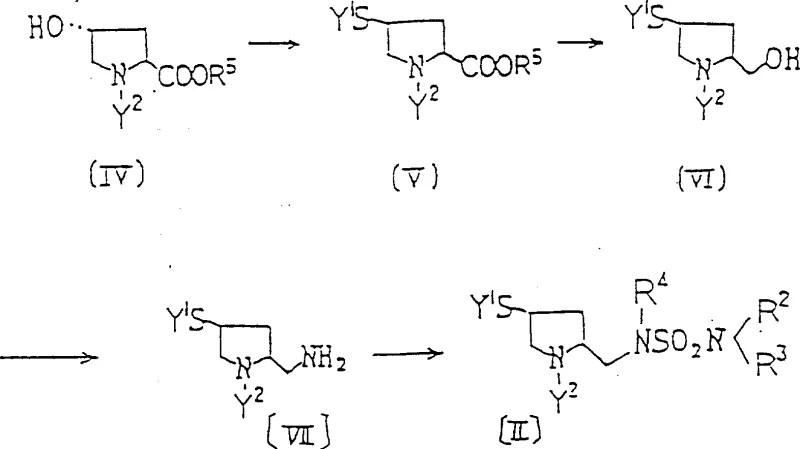
Mechanistically, the synthesis relies on precise stereochemical control to ensure the formation of the biologically active isomers. The starting material, typically trans-4-hydroxy-L-proline or its derivatives, provides the initial chiral framework. Throughout the synthesis, inversion of configuration at the 4-position is carefully managed during the nucleophilic substitution steps to achieve the desired (2S, 4S) or (2S, 4R) stereochemistry required for optimal binding to penicillin-binding proteins. The use of phthalimide or azide intermediates for the introduction of the amino group at the 2-position side chain allows for clean transformations with minimal racemization. The sulfamoylation step, often achieved using chlorosulfonyl isocyanate or similar reagents, is conducted at low temperatures to prevent decomposition. This attention to mechanistic detail ensures that impurity profiles remain within acceptable limits, a critical factor for regulatory approval.
Impurity control is further enhanced by the strategic selection of protecting groups. For instance, the use of the p-nitrobenzyloxycarbonyl (Pnz) group allows for removal under specific hydrogenation conditions that do not affect other sensitive functionalities. Similarly, the allyloxycarbonyl (Alz) group can be removed using palladium catalysts under neutral conditions, offering flexibility in the deprotection sequence. This orthogonality in protecting group chemistry is a hallmark of robust process design, enabling the removal of byproducts and unreacted materials at various stages. The result is a final product with high chemical purity, reducing the burden on downstream purification processes and lowering the overall cost of goods.
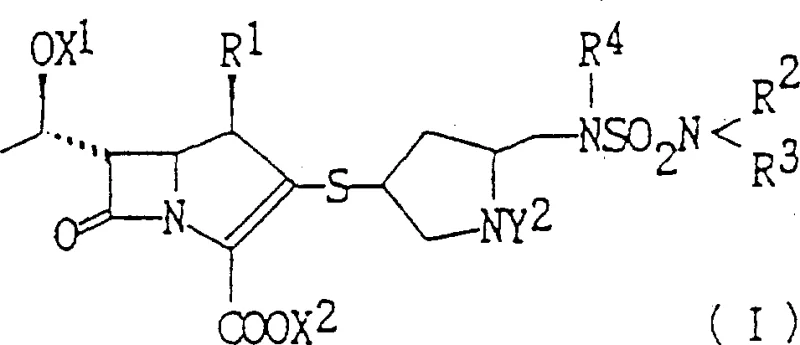
To synthesize pyrrolidinylthio carbapenem derivatives efficiently, the process begins with the protection of the nitrogen atom in 4-hydroxyl pyrrolidine-2-carboxylic acid, followed by activation of the 4-hydroxyl group. The subsequent steps involve nucleophilic displacement to introduce sulfur, reduction of the ester, and functionalization of the resulting alcohol to a sulfamoylamino group. Finally, the deprotected thiol is coupled with an activated carbapenem core. Detailed standardized synthesis steps are provided below to guide process implementation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits in terms of cost efficiency and supply security. The reliance on commodity chemicals such as proline derivatives, methanesulfonyl chloride, and standard protecting group reagents reduces exposure to volatile raw material markets. Unlike processes that depend on exotic catalysts or rare earth metals, this route utilizes widely available organic reagents, ensuring consistent availability and pricing stability. This stability is crucial for long-term supply agreements and helps mitigate the risk of production delays caused by raw material shortages.
Cost reduction in antibiotic manufacturing is achieved through the high yield and selectivity of the reaction steps. The modular nature of the synthesis allows for the recycling of solvents and the recovery of valuable byproducts, such as phthalic acid from phthalimide deprotection. Additionally, the ability to perform reactions at ambient or moderately low temperatures reduces energy consumption compared to high-temperature processes. The streamlined purification protocols, often involving simple crystallization or chromatography, minimize solvent usage and waste generation. These factors collectively contribute to a more sustainable and economically viable manufacturing process, aligning with modern green chemistry principles.
Scalability and environmental compliance are inherent strengths of this methodology. The reaction conditions are designed to be safe for large-scale operation, avoiding the use of highly explosive or toxic reagents where possible. The waste streams generated are primarily organic solvents and salts, which can be treated using standard industrial wastewater treatment facilities. This ease of waste management simplifies the regulatory compliance process for new manufacturing sites. Furthermore, the robustness of the chemistry ensures that the process can be transferred from pilot scale to commercial production with minimal re-optimization, accelerating time-to-market for new generic or branded carbapenem products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of pyrrolidinylthio carbapenem derivatives. These answers are derived from the detailed experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology.
Q: What are the key advantages of the pyrrolidinylthio carbapenem derivatives described in CN1032257C?
A: These derivatives exhibit an extensive antimicrobial spectrum, showing superior activity against Gram-positive bacteria compared to meropenem and better efficacy against Gram-negative bacteria compared to imipenem. They also demonstrate lower renal toxicity and higher water solubility.
Q: How does the new synthesis method improve supply chain reliability for carbapenem intermediates?
A: The method utilizes readily available starting materials like 4-hydroxyl pyrrolidine-2-carboxylic acid and employs robust protecting group strategies (such as Boc, Pnz, and Alz) that allow for scalable purification and high-yield transformations, reducing dependency on scarce reagents.
Q: Is the deprotection process compatible with large-scale manufacturing?
A: Yes, the patent describes deprotection steps using standard reagents like aluminum chloride and nitromethane or catalytic hydrogenation, which are well-established in industrial settings, ensuring the process can be scaled from laboratory to commercial production without significant modification.
- Convert 4-hydroxyl pyrrolidine-2-carboxylic acid derivatives into protected intermediates by transforming the 4-position hydroxyl into a sulfhydryl group and the 2-position carboxyl into a hydroxymethyl group.
- Introduce the sulfamoyl group by converting the hydroxymethyl hydroxyl into an amino or protected amino group, followed by sulfamoylation.
- React the resulting pyrrolidine derivative with a carbapenem derivative containing a leaving group at the 2-position to form the final thio-linked carbapenem structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidinylthio Carbapenem Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for complex pharmaceutical intermediates, leveraging deep expertise in beta-lactam chemistry to deliver high-quality solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications in antibiotic manufacturing and employ rigorous QC labs to verify every batch against the highest industry standards. Our commitment to quality assurance guarantees that the intermediates we supply will perform reliably in your downstream API synthesis.
We invite you to collaborate with us to optimize your supply chain for carbapenem production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Contact us today to request specific COA data and route feasibility assessments for pyrrolidinylthio carbapenem derivatives. Let us partner with you to drive innovation and efficiency in your antibiotic manufacturing operations.
