Advanced Manufacturing of Cis-Rose Oxide: A Technical Breakthrough in Fragrance Intermediates
Advanced Manufacturing of Cis-Rose Oxide: A Technical Breakthrough in Fragrance Intermediates
The global demand for high-quality fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for valued terpenes like cis-rose oxide. Patent CN101903366B introduces a sophisticated and commercially viable method for the preparation of cis-2-(2-methylprop-1-enyl)-4-methyltetrahydropyran, commonly known as cis-rose oxide. This patent outlines a dual-stage catalytic process that leverages heterogeneous ruthenium catalysts for hydrogenation followed by solid-acid isomerization. This approach represents a significant departure from traditional homogeneous acid catalysis, offering a pathway to superior purity profiles and streamlined downstream processing. For R&D directors and procurement specialists in the flavors and fragrances sector, understanding this technology is crucial for securing a reliable supply of this high-value intermediate.
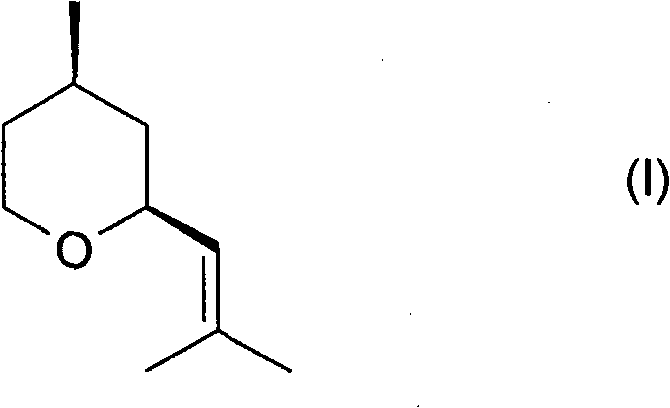
The core innovation lies in the specific sequence of reactions that maximizes the yield of the cis-diastereomer while minimizing waste. Unlike older methods that struggle with difficult separations of diastereomers, this process integrates an isomerization step that actively converts the unwanted trans-isomer back into the desired cis-form. This capability is transformative for industrial manufacturing, as it reduces the reliance on energy-intensive distillation columns to separate closely boiling isomers. By adopting this methodology, manufacturers can achieve a final product composition comprising at least 90% of the valuable cis-isomer, meeting the stringent quality standards required by top-tier perfume houses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of rose oxide has relied heavily on homogeneous acid catalysts such as tin tetrachloride (SnCl4) or boron trifluoride (BF3) to catalyze the cyclization and isomerization steps. While these reagents are effective in laboratory settings, they present severe drawbacks when translated to large-scale industrial production. The primary issue is the difficulty in separating the catalyst from the reaction mixture, which often necessitates extensive aqueous workups, neutralization steps, and multiple washing cycles. This not only generates substantial volumes of hazardous wastewater containing heavy metals but also leads to product loss during extraction. Furthermore, homogeneous catalysts often lack selectivity, resulting in complex mixtures of cis and trans isomers that are notoriously difficult to separate due to their similar boiling points, ultimately capping the overall yield and purity of the final fragrance ingredient.
The Novel Approach
The method disclosed in CN101903366B overcomes these legacy challenges by employing a heterogeneous catalytic system. The process begins with the catalytic hydrogenation of 2-(2-methylprop-1-enyl)-4-methylenetetrahydropyran (dehydrogenation rose oxide) using a ruthenium catalyst supported on carbon. This solid catalyst can be easily removed via simple filtration, eliminating the need for quenching and neutralization. Following hydrogenation, the crude mixture is subjected to a second catalytic step using a strongly acidic cation exchanger resin. This solid acid facilitates the isomerization of the trans-diastereomer into the cis-diastereomer. This two-step heterogeneous approach simplifies the workflow, drastically reduces solvent consumption, and enables the potential for continuous flow processing, marking a substantial upgrade in process efficiency and environmental compliance compared to traditional batch processes using liquid acids.
Mechanistic Insights into Ru-Catalyzed Hydrogenation and Solid-Acid Isomerization
The mechanistic elegance of this process begins with the selective hydrogenation of the exocyclic methylene group. As illustrated by the precursor structure, the starting material contains both an endocyclic double bond within the pyran ring and an exocyclic methylene group. The specialized ruthenium catalyst, often doped with iron on a carbon support, exhibits high chemoselectivity. It preferentially reduces the exocyclic double bond to a methyl group while preserving the endocyclic ether linkage and the side-chain alkene geometry initially. This step is critical because over-reduction or isomerization at this stage could lead to saturated byproducts or incorrect stereoisomers. The use of a supported metal catalyst ensures that the active sites are accessible for hydrogen activation while maintaining stability under the elevated temperatures and pressures required for efficient conversion.
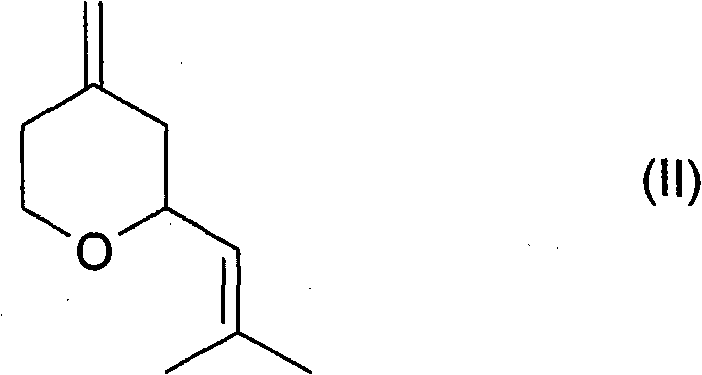
Following the hydrogenation, the reaction mixture typically contains a ratio of cis and trans diastereomers. The patent describes a subsequent equilibration step driven by thermodynamics but kinetically controlled by the acidic sites of the ion exchange resin. The strongly acidic cation exchanger acts as a proton source, facilitating the reversible opening of the pyran ring or the formation of carbocation intermediates. This allows the stereochemical configuration at the chiral centers to equilibrate. Since the cis-isomer is thermodynamically favored or can be enriched through specific resin interactions, the equilibrium shifts towards the formation of cis-rose oxide. This mechanism effectively recycles the 'waste' trans-isomer into the valuable cis-product, pushing the overall yield well beyond the theoretical limits of a single-pass kinetic reaction. The ability to tune this isomerization by selecting specific resin types (e.g., sulfonic acid resins) provides a powerful tool for process optimization.
How to Synthesize Cis-Rose Oxide Efficiently
The synthesis protocol detailed in the patent offers a robust framework for producing high-purity cis-rose oxide suitable for fine fragrance applications. The process is designed to be operationally simple, relying on standard unit operations found in most fine chemical plants, such as hydrogenation autoclaves and fixed-bed reactors. The initial step involves the preparation of the dehydrogenation rose oxide precursor, followed by the key hydrogenation and isomerization sequence. The following guide summarizes the critical operational parameters derived from the patent examples to ensure optimal conversion and selectivity.
- Perform catalytic hydrogenation of 2-(2-methylprop-1-enyl)-4-methylenetetrahydropyran using a heterogeneous ruthenium catalyst on a carbon support under hydrogen pressure.
- Separate the resulting reaction mixture containing cis- and trans-diastereomers, optionally removing the catalyst and solvent.
- Contact the mixture with a strongly acidic cation exchanger to isomerize the trans-isomer into the desired cis-rose oxide, enriching the final product purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the technology described in CN101903366B offers compelling economic and logistical benefits. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of manufacturing cis-rose oxide. By eliminating the need for expensive and hazardous liquid Lewis acids, the process reduces raw material costs and significantly lowers the expense associated with waste disposal and environmental compliance. The simplicity of catalyst removal via filtration translates directly into shorter batch cycles and higher equipment utilization rates, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The implementation of a heterogeneous ruthenium catalyst and solid acid resins removes the necessity for complex aqueous workups and neutralization steps. This reduction in processing stages leads to substantial savings in utility consumption, including water and energy for heating and cooling. Furthermore, the ability to recover and potentially regenerate the solid catalysts contributes to a lower cost of goods sold (COGS) over the long term. The elimination of heavy metal contaminants also reduces the burden on quality control laboratories, streamlining the release of finished goods.
- Enhanced Supply Chain Reliability: The robustness of the heterogeneous catalytic system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with major fragrance houses. The process is less sensitive to minor variations in reaction conditions compared to sensitive homogeneous systems, reducing the risk of batch failures. Additionally, the precursors required for this synthesis, such as isoprenol and prenal, are commodity chemicals with stable supply chains, mitigating the risk of raw material shortages that could disrupt production schedules.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, moving seamlessly from pilot plant to full commercial production. The use of fixed-bed reactors for the isomerization step allows for continuous operation, which is ideal for large-volume manufacturing. From an environmental perspective, the drastic reduction in hazardous waste generation aligns with increasingly strict global regulations on chemical manufacturing. This 'greener' profile not only minimizes regulatory risk but also enhances the brand value of the final product for eco-conscious consumers and clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of cis-rose oxide using this advanced catalytic method. These insights are derived directly from the experimental data and process descriptions within the patent, providing clarity on the feasibility and advantages of this route for potential partners and stakeholders.
Q: What is the primary advantage of using a heterogeneous ruthenium catalyst over homogeneous acids in rose oxide production?
A: The use of a heterogeneous ruthenium catalyst on a carbon support allows for easy filtration and recovery of the catalyst, eliminating the need for complex neutralization and washing steps associated with homogeneous Lewis acids like SnCl4 or BF3, thereby reducing waste and processing time.
Q: How does the patented process improve the cis-to-trans isomer ratio?
A: The process employs a subsequent isomerization step using a strongly acidic cation exchanger. This solid acid catalyst selectively converts the less desirable trans-diastereomer into the valuable cis-isomer, significantly enriching the final mixture to contain over 90% cis-rose oxide without requiring difficult fractional distillation.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method is specifically designed for technical scale production. The use of fixed-bed reactors for the isomerization step and robust heterogeneous hydrogenation catalysts facilitates continuous processing, ensuring consistent quality and high throughput suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Rose Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the creation of world-class fragrances. Our technical team has extensively analyzed the pathways described in CN101903366B and possesses the expertise to implement this sophisticated ruthenium-catalyzed process. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of cis-rose oxide meets the exacting standards of the global flavor and fragrance industry.
We invite you to collaborate with us to optimize your supply chain for this valuable ingredient. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
