Scalable Production of 3-Isopropyl-5-Cresol and Carvacrol via Catalytic Oxidation and Isomerization
Introduction to Advanced Phenolic Synthesis via Patent CN109232193B
The global demand for high-value phenolic compounds such as 3-isopropyl-5-cresol and carvacrol has surged due to their critical applications in agrochemicals, food preservatives, and pharmaceutical intermediates. Traditional sourcing methods often struggle with supply consistency and purity standards, creating a bottleneck for large-scale manufacturing. A groundbreaking technical solution is presented in Chinese Patent CN109232193B, which discloses a highly efficient, two-step catalytic method for converting 3-carene, an abundant monoterpene derived from turpentine, into these valuable isomers. This process leverages a unique combination of allylic oxidation and solid-acid catalyzed isomerization to achieve unprecedented selectivity and yield. By utilizing 3-carene as a renewable feedstock, this technology not only addresses the scarcity of natural sources but also establishes a robust synthetic pathway that bypasses the complex separation issues associated with conventional cresol alkylation. For industry leaders seeking a reliable agrochemical intermediate supplier, this patent represents a pivotal shift towards more sustainable and economically viable production methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of isopropyl-substituted cresols like thymol and carvacrol has relied heavily on the Friedel-Crafts alkylation of cresols with propylene or isopropanol. While widely practiced, this legacy approach suffers from significant thermodynamic and kinetic limitations that hinder optimal commercial performance. The reaction typically generates a complex mixture of ortho-, meta-, and para-substituted isomers, necessitating energy-intensive and costly separation processes to isolate the desired meta- or ortho-isomers. Furthermore, the use of liquid strong acids or corrosive Lewis acids as catalysts introduces severe environmental and safety hazards, complicating waste disposal and equipment maintenance. In the context of 3-isopropyl-5-cresol specifically, conventional methods often result in it being a minor by-product with low selectivity, making its dedicated production economically unfeasible. Additionally, extraction from natural sources like oregano oil is constrained by seasonal variability, geographical limitations, and fluctuating agricultural yields, rendering the supply chain vulnerable to external shocks and price volatility.
The Novel Approach
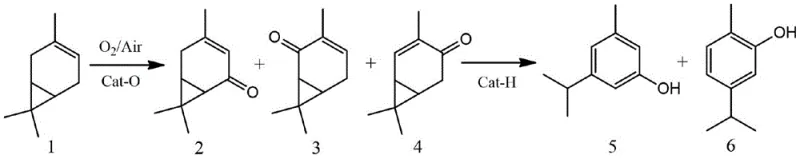
In stark contrast to these traditional constraints, the methodology outlined in CN109232193B introduces a streamlined cascade reaction system that transforms 3-carene directly into the target phenols with remarkable efficiency. The process initiates with a mild allylic oxidation using molecular oxygen or air over a supported chromium oxide catalyst, generating a specific mixture of carenone ketones without the need for harsh oxidants. Crucially, this oxidation step allows for the recovery and recycling of unreacted 3-carene, maximizing atom economy and reducing raw material costs. The subsequent isomerization step utilizes a shape-selective 13X molecular sieve catalyst at elevated temperatures to rearrange the ketone intermediates into the desired phenolic structures. This solid-acid catalysis eliminates the need for corrosive liquid acids and simplifies the downstream workup significantly. By integrating these two steps, the novel approach achieves a total phenol selectivity of up to 98% and allows for the tunable production of 3-isopropyl-5-cresol and carvacrol, offering a distinct competitive advantage in cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into CrO3-Al2O3 Catalyzed Oxidation and Isomerization
The core of this technological breakthrough lies in the precise control of reaction mechanisms across two distinct catalytic environments. In the first stage, the supported CrO3-Al2O3 catalyst facilitates a radical-mediated allylic oxidation of the 3-carene double bond. The chromium species activate molecular oxygen to abstract an allylic hydrogen, forming a hydroperoxide intermediate that decomposes to yield the conjugated enones: 5-carenone, 2-carenone, and 4-carenone. The support material, alumina, plays a critical role in dispersing the active chromium sites and stabilizing the transition states, which results in a highly favorable selectivity ratio of approximately 10.8:3.5:1.0 favoring the 5-carenone isomer. This specific distribution is vital because the subsequent isomerization efficiency depends heavily on the structure of these ketone precursors. The ability to conduct this reaction at room temperature (15-35°C) further underscores the mildness of the catalytic system, preventing thermal degradation of the sensitive terpene skeleton and minimizing the formation of polymeric by-products that often plague high-temperature oxidations.
Following the oxidation, the reaction mixture undergoes a profound structural rearrangement driven by the Brønsted and Lewis acid sites within the pores of the 13X molecular sieve. At temperatures around 230°C, the carenone ketones adsorb onto the acidic sites of the zeolite framework, initiating a protonation of the carbonyl oxygen. This triggers a series of hydride shifts and ring-expansion or contraction events that ultimately aromatize the six-membered ring, yielding the stable phenolic products. The pore size of the 13X sieve is instrumental in this process, as it imposes steric constraints that favor the formation of the specific 3-isopropyl-5-cresol and carvacrol isomers over other potential rearrangement products. This shape-selective catalysis ensures a total phenol selectivity reaching 98%, with a controllable ratio of roughly 2.2:1.0 between the meta-cresol and carvacrol. Understanding this mechanistic pathway is essential for R&D teams aiming to optimize reactor conditions for commercial scale-up of complex fine chemicals, as it highlights the importance of catalyst acidity and pore architecture in directing product distribution.
How to Synthesize 3-Isopropyl-5-Cresol Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to ensure reproducibility and high yield. The process begins with the impregnation of neutral alumina with a chromium oxide solution to create the oxidation catalyst, followed by a controlled oxidation of 3-carene with air or oxygen. Once the ketone mixture is obtained and unreacted starting material is distilled off, the residue is passed through a fixed-bed reactor containing the molecular sieve catalyst. The operational simplicity of this continuous flow isomerization step makes it particularly attractive for industrial adoption. For detailed procedural specifics regarding catalyst loading, space velocity, and temperature gradients, please refer to the standardized synthesis protocol below.
- Perform allylic oxidation of 3-carene using oxygen or air over a supported CrO3-Al2O3 catalyst at room temperature to generate a mixture of carenone isomers.
- Recover unreacted 3-carene via distillation and subject the remaining ketone residue to isomerization over a 13X molecular sieve catalyst at 230°C.
- Purify the resulting phenolic mixture via reduced pressure distillation or column chromatography to isolate high-purity 3-isopropyl-5-cresol and carvacrol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this 3-carene-based pathway offers transformative benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the decoupling of production from agricultural harvest cycles, as 3-carene is a consistent by-product of the turpentine industry, available in substantial quantities year-round. This stability mitigates the risks associated with crop failures or seasonal shortages that plague natural extraction methods, ensuring a continuous and reliable supply of critical intermediates for downstream formulation. Furthermore, the use of heterogeneous catalysts in both reaction steps significantly simplifies the purification workflow, eliminating the need for extensive aqueous washes to remove homogeneous acid catalysts. This reduction in unit operations translates directly into lower utility consumption and reduced wastewater treatment burdens, aligning with modern sustainability mandates and lowering the overall cost of goods sold.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the high atom economy and the recyclability of key reagents. The supported chromium catalyst can be recovered and reused multiple times without significant loss of activity, drastically reducing the recurring cost of catalytic materials compared to single-use homogeneous systems. Additionally, the ability to recover and recycle over 80% of unreacted 3-carene means that raw material utilization is maximized, minimizing waste and purchase requirements. The elimination of expensive separation columns for isomer mixtures, which is a hallmark of traditional Friedel-Crafts processes, further drives down capital and operational expenditures. These factors combine to create a leaner manufacturing profile that offers substantial cost savings in flavor and fragrance manufacturing without compromising on product quality.
- Enhanced Supply Chain Reliability: Sourcing 3-carene from the established turpentine supply chain provides a robust foundation for long-term production planning. Unlike botanical extracts which are subject to geopolitical and climatic variables, turpentine derivatives are produced through established industrial forestry operations with predictable output volumes. The synthetic nature of this process also allows for rapid scaling of production capacity to meet sudden spikes in market demand, a flexibility that natural extraction simply cannot match. By securing a supply line based on this technology, companies can insulate themselves from the volatility of natural commodity markets and guarantee consistent delivery schedules to their own customers. This reliability is paramount for maintaining the integrity of global supply chains for essential agrochemicals and food additives.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a cleaner alternative to legacy chemistries by avoiding the generation of large volumes of acidic wastewater and heavy metal sludge. The use of molecular oxygen as the oxidant produces water as the only by-product in the first step, adhering to green chemistry principles. The solid-state nature of the isomerization catalyst facilitates easy handling and disposal, reducing the regulatory burden associated with hazardous waste management. Moreover, the process operates under relatively mild pressures and temperatures that are easily manageable in standard stainless steel reactors, facilitating straightforward scale-up from pilot plant to multi-ton commercial production. This ease of scalability ensures that the technology can grow alongside market demand, providing a future-proof solution for high-purity phenolic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing clarity on catalyst performance, product purity, and process robustness. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production facilities.
Q: What is the selectivity ratio of the ketone intermediates in the oxidation step?
A: The oxidation of 3-carene over CrO3-Al2O3 yields a ketone mixture primarily composed of 5-carenone, 2-carenone, and 4-carenone with a selectivity ratio of approximately 10.8:3.5:1.0, allowing for direct downstream processing without separation.
Q: Can the oxidation catalyst be recycled in this process?
A: Yes, the supported CrO3-Al2O3 catalyst demonstrates excellent stability and can be recovered via filtration or centrifugation and reused for at least three cycles without significant loss in catalytic activity or selectivity.
Q: What is the purity of the final phenolic products obtained?
A: Following reduced pressure distillation, the mixed phenol product achieves a purity greater than 96%. Further separation via column chromatography yields individual 3-isopropyl-5-cresol and carvacrol with purities exceeding 97% and 98% respectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Isopropyl-5-Cresol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing high-quality intermediates for the agrochemical and flavor industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our capability to handle complex catalytic systems, such as the chromium-alumina and molecular sieve technologies described in CN109232193B, positions us as a preferred partner for companies seeking to optimize their supply chains.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your product portfolio and operational efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and help you secure a competitive edge in the global market.
