Revolutionizing Cartap Intermediate Production: A Safe, High-Yield Thiocyanate Synthesis Strategy
The global agrochemical industry is constantly seeking safer and more efficient pathways for producing critical insecticide intermediates, particularly for cartap hydrochloride. Patent CN101519371B introduces a groundbreaking preparation method for 2-N,N-dimethyl-1,3-dithiocyanopropane, a pivotal precursor in the synthesis of this widely used pesticide. Traditionally, the production of this intermediate has been plagued by significant safety hazards and environmental concerns due to the reliance on highly toxic sodium cyanide. This new technology fundamentally shifts the paradigm by utilizing 1-N,N-dimethyl-2,3-dihalopropylamine and thiocyanate salts as primary raw materials. The innovation lies not just in the substitution of reagents but in a sophisticated separation and recycling protocol that addresses the persistent issue of isomer formation. By achieving a dynamic equilibrium between the normal form and the isomer, the process ensures high yields while maintaining exceptional product purity, making it a highly attractive option for reliable agrochemical intermediate suppliers aiming to modernize their production lines.
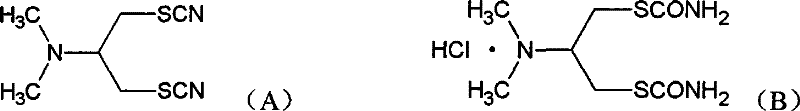
This technological advancement is particularly relevant for manufacturers facing stringent regulatory pressures regarding cyanide usage and waste disposal. The patent details a robust synthetic route that circumvents the need for dangerous cyanation reactions typically associated with converting cartap back to its intermediate form. Instead, it leverages a nucleophilic substitution reaction that is inherently safer and easier to control on an industrial scale. The resulting intermediate, characterized by its high content of the 1,3-dithiocyanopropane structure, serves as a superior feedstock for subsequent hydrolysis steps. For R&D directors and procurement managers, understanding the nuances of this process is essential, as it offers a clear pathway to reducing lead time for high-purity agrochemical intermediates while simultaneously mitigating the operational risks associated with handling剧毒 substances. The ability to produce a normal form content of ≥98% directly impacts the efficacy and quality of the final insecticide product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
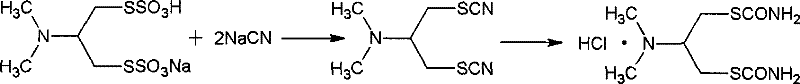
Historically, the synthesis of high-purity cartap has relied heavily on a circuitous and hazardous route involving the reaction of cartap itself with sodium cyanide to regenerate the thiocyanate intermediate. As illustrated in the reaction scheme below, this conventional method necessitates the handling of large quantities of剧毒 sodium cyanide, creating severe occupational health risks and generating substantial volumes of cyanide-containing wastewater that require expensive and complex treatment protocols. Furthermore, alternative non-cyanide routes, such as those described in prior art like CN1830957A, often suffer from poor selectivity, yielding mixtures where the desired normal form constitutes only 50% to 60% of the product. This low selectivity results in a final cartap product contaminated with significant amounts of inactive isomers, thereby diminishing the biological efficacy of the pesticide and complicating the purification process. The economic burden of disposing of toxic waste and the low yield of the active ingredient render these traditional methods increasingly unsustainable in a modern regulatory environment.
The Novel Approach
In stark contrast, the methodology disclosed in CN101519371B employs a direct thiocyanation of 1-N,N-dimethyl-2,3-dihalopropylamine, effectively bypassing the need for free cyanide ions entirely. The core of this innovation is the recognition that while the initial reaction produces a mixture of the desired 1,3-dithiocyanopropane (normal form) and the undesired 1,2-dithiocyanopropane (isomer), these components can be efficiently separated and managed. The process utilizes specific solvent systems, such as alcohols or ketones, to facilitate the selective crystallization of the isomer, which is then physically removed from the reaction matrix. Rather than discarding this byproduct, the novel approach recycles the separated isomer back into the reaction vessel for subsequent batches. This recycling mechanism establishes a dynamic equilibrium that progressively converts the isomer into the normal form over multiple cycles, dramatically improving the overall yield without compromising safety. This strategic loop transforms a potential waste stream into a valuable resource, significantly enhancing the economic viability of the process.
Mechanistic Insights into Thiocyanate Substitution and Isomer Equilibrium
The chemical foundation of this process rests on a nucleophilic substitution reaction where the thiocyanate anion attacks the halogenated carbon centers of the 1-N,N-dimethyl-2,3-dihalopropylamine substrate. As depicted in the structural formulas, the starting material contains two reactive halogen sites, which can lead to the formation of either the linear 1,3-substituted product or the branched 1,2-substituted isomer depending on the reaction kinetics and steric factors. The patent specifies that the reaction is typically conducted at temperatures ranging from 45°C to 85°C, with a preferred range of 60°C to 75°C, to optimize the reaction rate while minimizing side reactions. The choice of solvent plays a critical role in this mechanism; polar aprotic solvents or alcohols like n-butanol and acetone are preferred because they effectively dissolve the thiocyanate salt while allowing for the differential solubility required for the subsequent separation step. The molar ratio of thiocyanate to the halogenated amine is carefully controlled, typically between 2.0:1 and 2.5:1, to ensure complete conversion of the starting material without excessive reagent waste.
Following the reaction, the separation mechanism relies on the distinct physical properties of the normal form and the isomer within the chosen solvent system. Upon cooling the reaction mixture, the 1,2-isomer exhibits a higher tendency to crystallize out of the solution compared to the 1,3-normal form. This fractional crystallization allows for the isolation of the isomer with high purity (often >90% isomer content in the solid phase), leaving the mother liquor enriched with the desired normal form. The patent highlights that this separation is not merely a purification step but a crucial part of the yield enhancement strategy. By returning the crystallized isomer to the reactor, it undergoes further reaction conditions that favor its conversion or simply dilutes the formation of new isomers in the next batch, effectively driving the system towards the thermodynamic preference for the normal form. This intricate balance between reaction kinetics and physical separation ensures that the final filtrate contains ≥98% of the 2-N,N-dimethyl-1,3-dithiocyanopropane, meeting the rigorous standards required for high-performance agrochemical manufacturing.
How to Synthesize 2-N,N-dimethyl-1,3-dithiocyanopropane Efficiently
The synthesis of this critical intermediate requires precise control over reaction parameters and separation techniques to maximize the yield of the normal form. The patent outlines a procedure where the halogenated amine and thiocyanate salt are heated in a solvent like n-butanol or acetone, followed by a cooling crystallization step to remove impurities. The efficiency of this process is heavily dependent on the recycling of the separated isomer, which acts as a buffer to maintain high conversion rates in continuous production cycles. For technical teams looking to implement this, the key lies in optimizing the solvent-to-feed ratio and the cooling profile to ensure maximum recovery of the isomer for recycling. The detailed standardized synthesis steps, including specific temperatures, stirring rates, and filtration protocols necessary for commercial scale-up of complex agrochemical intermediates, are outlined in the guide below.
- React 1-N,N-dimethyl-2,3-dihalopropylamine with sodium thiocyanate in a suitable solvent like n-butanol or acetone at 45-85°C.
- Separate the resulting mixture by cooling to crystallize out the 3-N,N-dimethyl-1,2-dithiocyanopropane isomer impurity.
- Recycle the separated isomer back into the reaction vessel for the next batch to maximize overall yield of the normal form.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers profound strategic advantages that extend beyond simple chemical yield. The most immediate benefit is the elimination of sodium cyanide from the supply chain, which drastically simplifies logistics, storage, and regulatory compliance. Handling剧毒 materials requires specialized infrastructure, extensive safety training, and costly insurance, all of which are rendered unnecessary by this cyanide-free route. Furthermore, the reduction in hazardous waste generation translates directly into lower operational expenditures related to waste treatment and environmental remediation. By removing the need for complex cyanide destruction processes, manufacturers can achieve significant cost reduction in agrochemical intermediate manufacturing, allowing for more competitive pricing in the global market. The robustness of the process also ensures a more stable supply, as it is less susceptible to disruptions caused by strict regulations on cyanide transport and usage.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the internal recycling of the isomer byproduct. In traditional linear processes, isomers are often treated as waste, representing a loss of raw material value. Here, the isomer is reintroduced into the system, effectively increasing the atom economy and reducing the consumption of fresh 1-N,N-dimethyl-2,3-dihalopropylamine per unit of final product. This closed-loop system minimizes raw material costs and reduces the volume of waste requiring disposal. Additionally, the use of common organic solvents like alcohols and ketones, which can often be recovered and reused, further drives down the variable costs of production. The cumulative effect of these efficiencies is a substantially lower cost of goods sold, providing a strong margin advantage for producers.
- Enhanced Supply Chain Reliability: Reliance on剧毒 sodium cyanide creates a fragile supply chain vulnerable to regulatory crackdowns and transportation bans. By shifting to a thiocyanate salt-based process, manufacturers secure a more resilient supply line, as thiocyanates are generally easier to procure, store, and transport with fewer restrictions. This stability is crucial for long-term contracts with major agrochemical companies that demand uninterrupted supply. Moreover, the simplified process flow, which avoids the complex neutralization and washing steps associated with cyanide residues, shortens the overall production cycle time. This agility allows suppliers to respond more quickly to market demand fluctuations, ensuring that high-purity intermediates are available exactly when needed without the delays associated with hazardous material handling protocols.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology represents a significant leap forward. The absence of cyanide wastewater eliminates the risk of catastrophic environmental contamination and removes the burden of maintaining expensive effluent treatment plants dedicated to cyanide destruction. This makes the process highly scalable, as expanding production capacity does not proportionally increase the environmental liability or the complexity of waste management. The process generates primarily solid salt waste (e.g., sodium chloride) and organic solvent waste, which are far easier to manage and treat than cyanide-laden streams. This alignment with green chemistry principles not only future-proofs the manufacturing facility against tightening environmental regulations but also enhances the corporate sustainability profile, a key factor for modern B2B partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiocyanate synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN101519371B, providing a factual basis for decision-making. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own production facilities. The answers cover aspects ranging from raw material specifications to the handling of byproducts, ensuring a comprehensive overview of the process capabilities.
Q: How does this patent avoid the use of toxic sodium cyanide?
A: Unlike traditional methods that convert cartap to the intermediate using hazardous NaCN, this process starts from 1-N,N-dimethyl-2,3-dihalopropylamine and reacts it directly with safer thiocyanate salts.
Q: What is the purity level of the thiocyanate intermediate achieved?
A: The patented separation and recycling technique allows for the production of the normal form thiocyanate with a content greater than 98%, ensuring high-quality downstream cartap synthesis.
Q: How are the unwanted isomers handled in this process?
A: The process utilizes a crystallization step to separate the 1,2-isomer from the desired 1,3-normal form. Crucially, the separated isomer is not discarded but recycled back into the reactor, establishing a dynamic equilibrium that boosts total yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-N,N-dimethyl-1,3-dithiocyanopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, safe, and efficient synthetic routes for key agrochemical intermediates like 2-N,N-dimethyl-1,3-dithiocyanopropane. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101519371B are fully realized in practical, large-scale operations. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to guarantee that every batch meets the ≥98% normal form content required for high-efficacy insecticide production. Our state-of-the-art facilities are designed to handle complex chemistries safely, providing a secure and reliable source for your supply chain needs.
We invite you to collaborate with us to optimize your sourcing strategy for cartap intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our cyanide-free manufacturing process can reduce your total landed costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to build a safer, more sustainable, and cost-effective supply chain for your agrochemical products.
