Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously demands more efficient and cost-effective methods for producing chiral intermediates, which are the foundational building blocks for numerous life-saving medications. Patent CN1275077A introduces a groundbreaking process for the preparation of chiral compounds, specifically focusing on the resolution of N-methyl-3(R,S)-hydroxy-3-phenylpropylamine. This technology addresses the critical challenge of separating enantiomers from racemic mixtures, a step that is often the most costly and technically demanding part of synthesizing active pharmaceutical ingredients (APIs). By leveraging diastereomeric salt formation with mandelic acid, this method offers a robust pathway to obtain high-purity single enantiomers essential for drugs like Tomoxetine and Fluoxetine. The strategic implementation of this resolution process not only enhances the optical purity of the final product but also streamlines the manufacturing workflow, making it an attractive option for large-scale commercial production. Understanding the nuances of this patent is vital for R&D directors and procurement managers looking to optimize their supply chains for chiral pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the separation of enantiomers for pharmaceutical intermediates has relied heavily on chiral chromatography or enzymatic resolution, both of which present significant drawbacks in a commercial manufacturing setting. Chiral chromatography, while effective for analytical purposes, is often prohibitively expensive and difficult to scale due to the high cost of chiral stationary phases and the low throughput associated with column operations. Enzymatic methods, although green, can suffer from substrate specificity issues and require stringent control of reaction conditions to maintain enzyme activity, leading to potential batch-to-batch variability. Furthermore, many conventional chemical resolution methods utilize solvents that are environmentally hazardous or require complex downstream processing to remove toxic catalysts. These limitations result in extended lead times, increased production costs, and a larger environmental footprint, which are critical pain points for supply chain heads and procurement managers aiming to reduce overall manufacturing expenses and improve sustainability metrics in the production of complex pharmaceutical intermediates.
The Novel Approach
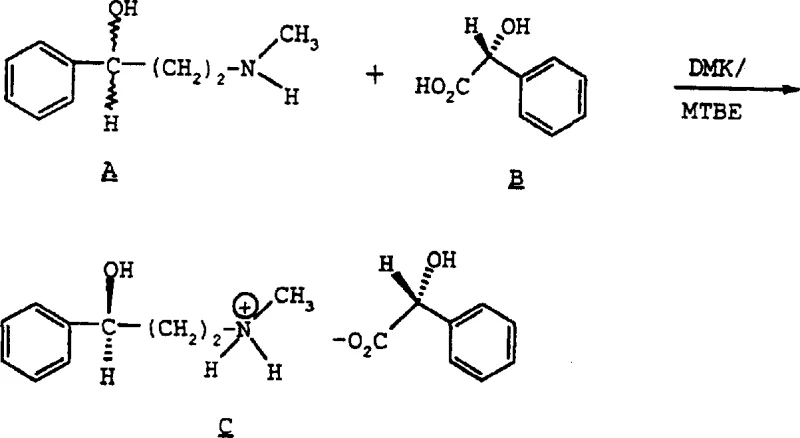
The novel approach described in the patent utilizes a diastereomeric salt crystallization technique that fundamentally shifts the paradigm from complex separation to efficient physical isolation. By reacting the racemic amine with a chiral resolving agent like S-(+)-mandelic acid, the process creates two diastereomeric salts that possess different physical properties, particularly solubility. This difference allows for the selective crystallization of the desired enantiomer simply by adjusting temperature and solvent composition, bypassing the need for expensive chromatographic columns. The method employs common, industrially friendly solvents such as ethyl acetate and methyl tert-butyl ether (MTBE), which are easily recoverable and recycle, significantly reducing solvent waste. This approach not only simplifies the operational workflow but also enhances the overall yield and purity of the target compound, providing a scalable solution that aligns perfectly with the needs of modern commercial scale-up of complex pharmaceutical intermediates. The ability to achieve high enantiomeric excess through simple filtration and washing steps represents a major leap forward in process chemistry efficiency.
Mechanistic Insights into Diastereomeric Salt Crystallization
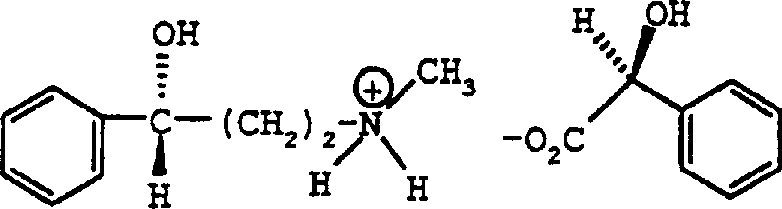
The core mechanism driving this resolution process is the formation of diastereomeric salts through an acid-base reaction between the racemic amine and the chiral mandelic acid. When N-methyl-3(R,S)-hydroxy-3-phenylpropylamine reacts with S-(+)-mandelic acid, it forms two distinct salts: the S-(+)-mandelate of the R-amine and the S-(+)-mandelate of the S-amine. Although these salts are chemically similar, their three-dimensional spatial arrangements differ, leading to distinct crystal lattice energies and solubility profiles in organic solvents. The process exploits the fact that one diastereomer is significantly less soluble than the other under specific conditions, causing it to precipitate out of the solution first while the unwanted enantiomer remains dissolved in the mother liquor. This selective precipitation is kinetically and thermodynamically controlled by the cooling rate and the specific solvent mixture used, ensuring that the crystalline lattice incorporates primarily the desired isomer. Understanding this solubility differential is crucial for R&D teams to optimize the crystallization parameters and maximize the recovery of the high-value chiral intermediate.
Impurity control in this process is inherently managed through the crystallization dynamics and the subsequent washing steps. As the desired diastereomeric salt crystallizes, the crystal lattice tends to exclude impurities and the opposite enantiomer, a phenomenon known as lattice rejection. However, to ensure the highest levels of purity, the patent describes specific washing protocols using solvent mixtures like MTBE and acetone. These washes are designed to dissolve any surface-adhered mother liquor which may contain the unwanted enantiomer or residual starting materials, without dissolving the bulk of the purified crystals. Furthermore, the process allows for recrystallization, where the crude salt is dissolved again and crystallized under refined conditions to further boost the enantiomeric excess to levels exceeding 99%. This multi-stage purification capability ensures that the final free base, obtained after basification, meets the stringent purity specifications required for downstream synthesis of APIs like Tomoxetine, thereby minimizing the risk of chiral impurities in the final drug product.
How to Synthesize N-methyl-3R-hydroxy-3-phenylpropylamine Efficiently
The synthesis of this critical chiral intermediate involves a straightforward yet highly optimized sequence of dissolution, reaction, and crystallization steps that can be readily adapted for industrial reactors. The process begins with the dissolution of the racemic amine in a solvent system, followed by the controlled addition of the chiral resolving agent to initiate salt formation. Careful monitoring of temperature and addition rates is essential to ensure uniform nucleation and crystal growth, which directly impacts the filterability and purity of the product. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and solvent volumes, are outlined in the structured guide below to assist technical teams in replicating this high-efficiency route.
- Dissolve racemic N-methyl-3-hydroxy-3-phenylpropylamine in a suitable organic solvent such as ethyl acetate or MTBE.
- Add stoichiometric amounts of S-(+)-mandelic acid or R-(-)-mandelic acid to the solution under controlled temperature conditions.
- Induce crystallization by cooling the mixture slowly to ambient temperature, then filter and wash the resulting diastereomeric salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this resolution technology offers substantial advantages that directly address the key concerns of procurement managers and supply chain heads regarding cost, reliability, and scalability. The elimination of expensive chiral catalysts and chromatography media significantly reduces the raw material costs associated with production, while the use of common solvents facilitates easier sourcing and recycling. The robustness of the crystallization process ensures consistent batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. Additionally, the simplicity of the unit operations involved—mixing, heating, cooling, and filtering—means that the process can be easily transferred to existing manufacturing facilities without requiring specialized equipment, thereby accelerating the time to market for new chiral intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this manufacturing process is the replacement of high-cost separation technologies with low-cost crystallization unit operations. By avoiding the use of precious metal catalysts or expensive enzymatic systems, the direct material cost is drastically simplified, leading to substantial cost savings over the lifecycle of the product. Furthermore, the solvents used, such as ethyl acetate and MTBE, are commodity chemicals with stable pricing and high recovery rates, which minimizes waste disposal costs and solvent purchase expenses. The high yield and purity achieved through this method also reduce the need for reprocessing or discarding off-spec material, further enhancing the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the use of readily available starting materials and reagents that are not subject to the geopolitical or supply constraints often associated with specialized chiral catalysts. The process relies on mandelic acid and simple amines, which are produced by multiple global suppliers, ensuring a continuous and secure supply of raw materials. The operational simplicity of the process also reduces the dependency on highly specialized technical staff, making it easier to maintain production continuity across different manufacturing sites. This resilience against supply disruptions is critical for maintaining the steady flow of high-purity pharmaceutical intermediates required to support downstream API production schedules.
- Scalability and Environmental Compliance: The scalability of this process is inherent in its design, as crystallization is a unit operation that scales linearly from laboratory to multi-ton reactors with minimal parameter adjustments. The use of environmentally benign solvents and the absence of heavy metal waste streams simplify the environmental compliance burden, reducing the costs and complexities associated with waste treatment and regulatory reporting. This green chemistry profile aligns with modern sustainability goals, making the supply chain more robust against tightening environmental regulations. The ability to scale up complex pharmaceutical intermediates without generating hazardous byproducts ensures long-term operational viability and reduces the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These answers are derived directly from the patent specifications and practical manufacturing considerations, providing clarity on purity, scalability, and process robustness. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: What is the primary advantage of using mandelic acid for chiral resolution?
A: Using mandelic acid allows for the formation of diastereomeric salts with distinct solubility profiles, enabling high-purity separation through simple crystallization rather than expensive chromatography.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like ethyl acetate and standard crystallization techniques, making it highly scalable from kilogram to multi-ton production levels.
Q: What enantiomeric excess can be achieved with this method?
A: Optimization of solvent ratios and crystallization temperatures can achieve enantiomeric excess values exceeding 99%, suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-3R-hydroxy-3-phenylpropylamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral technology implementation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing crystallization parameters to ensure stringent purity specifications are met consistently, leveraging rigorous QC labs to verify enantiomeric excess and chemical purity at every stage. We understand the critical nature of chiral intermediates in the pharmaceutical value chain and are committed to delivering high-quality materials that support your drug development and commercialization goals. Our facility is equipped to handle the specific solvent systems and temperature controls required for this mandelate resolution process, ensuring a seamless transition from pilot scale to full commercial manufacturing.
We invite you to collaborate with us to optimize your supply chain for chiral intermediates. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your production efficiency and reduce overall costs. Let us be your partner in navigating the complexities of chiral synthesis and securing a reliable supply of high-purity intermediates for your critical pharmaceutical projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
