Advanced One-Pot Synthesis of Delta-Dodecalactone for Industrial Flavor Applications
The global demand for high-quality lactones in the flavor and fragrance industry continues to surge, driven by the need for natural-identical butter and fruity notes in food applications. A pivotal advancement in this sector is detailed in patent CN103044374A, which discloses a highly efficient synthesis method for delta-dodecalactone (CAS 713-95-1). This technology represents a significant departure from traditional multi-step protocols by integrating aldol condensation and catalytic hydrogenation into a seamless one-pot operation. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: a streamlined pathway that not only enhances product yield by 3-4% but also drastically reduces energy consumption through simplified purification. As a leading manufacturer, we recognize that adopting such robust synthetic strategies is essential for maintaining competitiveness in the volatile fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of delta-dodecalactone has been plagued by inefficient synthetic routes that hinder scalability and economic viability. Traditional methods often rely on the reaction of cyclohexanedione with n-hexyl bromide, a process burdened by繁琐 oxidation, reduction, and cyclization steps that result in low overall yields and complex waste streams. Alternatively, the Dieckmann condensation of diethyl adipate followed by alkylation and decarboxylation presents another bottleneck; this route involves excessively long reaction sequences that are difficult to control on a commercial scale, leading to inconsistent batch quality. Even the more common approach using cyclopentanone and n-heptanal typically separates the aldol condensation and hydrogenation into distinct unit operations, necessitating intermediate isolation that incurs significant material loss and solvent waste. These legacy processes fail to meet the modern standards of green chemistry and cost-efficiency required by today's supply chains.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent introduces a transformative one-pot tandem catalysis strategy that fundamentally restructures the synthesis workflow. By simultaneously conducting aldol condensation and catalytic hydrogenation in a single reactor under controlled pressure and temperature, the process eliminates the need for intermediate isolation, thereby preserving material integrity and maximizing throughput. This integrated approach allows for the direct conversion of readily available raw materials, cyclopentanone and n-heptanal, into the key 2-heptylcyclopentanone intermediate with superior efficiency. Following this, a straightforward oxidative ring expansion completes the synthesis, requiring only two rectification steps to achieve high purity. This reduction in unit operations translates directly into lower capital expenditure (CAPEX) for equipment and reduced operational expenditure (OPEX) for energy and labor.
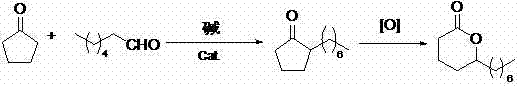
Mechanistic Insights into One-Pot Tandem Aldol-Hydrogenation
The core of this technological breakthrough lies in the precise orchestration of base-catalyzed aldol condensation followed immediately by heterogeneous hydrogenation within the same reaction vessel. In this system, an alkaline solution, such as sodium hydroxide or potassium hydroxide, facilitates the nucleophilic attack of the enolate derived from cyclopentanone onto the carbonyl carbon of n-heptanal. Crucially, the presence of a hydrogenation catalyst, typically comprising active components like Palladium, Platinum, or Nickel supported on carbon or silica, ensures that the resulting unsaturated aldol adduct is rapidly reduced in situ. This immediate hydrogenation prevents the accumulation of reactive intermediates that could otherwise undergo polymerization or side reactions, thus enhancing the selectivity towards the desired saturated ketone. The reaction is maintained at temperatures between 50-100°C and hydrogen pressures of 2.0-5.0 MPa, conditions that are optimized to drive the equilibrium forward while ensuring catalyst stability and activity throughout the extended addition period.
Impurity control is rigorously managed through the specific sequencing of the oxidative ring expansion step, which employs a Baeyer-Villiger oxidation mechanism to insert an oxygen atom adjacent to the carbonyl group. By utilizing peroxide oxidants like peracetic acid or hydrogen peroxide in solvents such as toluene or xylene, the process achieves high regioselectivity for the formation of the seven-membered lactone ring. The protocol mandates careful temperature control between -5°C and 60°C during the oxidant addition to mitigate exothermic risks and prevent over-oxidation byproducts. Post-reaction workup involves sequential washing with sodium sulfite and sodium sulfate solutions to neutralize residual acids and remove inorganic salts, ensuring that the final distillation yields a product with GC analysis content exceeding 99%. This meticulous attention to reaction parameters guarantees a clean impurity profile, which is critical for applications in sensitive food and fragrance formulations.
How to Synthesize Delta-Dodecalactone Efficiently
Implementing this synthesis route requires strict adherence to the patented parameters to ensure safety and reproducibility at scale. The process begins with the preparation of the alkaline catalyst slurry, followed by the controlled dropwise addition of the ketone-aldehyde mixture under a hydrogen atmosphere. Operators must monitor pressure and temperature closely to maintain the tandem reaction kinetics, ensuring that the hydrogenation rate matches the condensation rate to avoid intermediate buildup. After the reaction reaches completion, the catalyst is recovered via filtration for reuse, and the crude product undergoes a specialized distillation sequence to separate the intermediate before the final oxidation step. For a comprehensive breakdown of the specific molar ratios, addition rates, and purification cuts, please refer to the standardized guide below.
- Perform a one-pot tandem reaction by mixing cyclopentanone and n-heptanal in an alkaline solution containing a hydrogenation catalyst (Pd/C, Pt, or Ni) under 2.0-5.0 MPa hydrogen pressure at 50-100°C to form 2-heptylcyclopentanone.
- Filter the reaction mixture to recover the catalyst, separate the layers, and distill the oil layer to recover unreacted cyclopentanone, followed by rectification to isolate the 2-heptylcyclopentanone intermediate.
- Conduct oxidative ring expansion by reacting the intermediate with a peroxide oxidant (such as peracetic acid) in a solvent like toluene at -5 to 60°C, followed by washing and vacuum distillation to collect the final delta-dodecalactone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis technology offers substantial strategic benefits that extend beyond mere technical elegance. The consolidation of two major reaction steps into a single vessel significantly reduces the total cycle time per batch, allowing for higher asset utilization and faster turnaround times to meet fluctuating market demands. Furthermore, the ability to recycle the precious metal catalyst directly from the reaction mixture minimizes the consumption of expensive raw materials, providing a direct lever for cost reduction in flavor and fragrance manufacturing without compromising on quality. The simplified downstream processing, characterized by only two rectification steps, drastically lowers energy consumption and solvent usage, aligning production with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for additional drying, storage, and handling equipment, which significantly lowers both fixed and variable production costs. By avoiding the losses associated with transferring materials between multiple reactors, the overall mass balance is improved, leading to a higher effective yield of the final API or intermediate. Additionally, the recovery and reuse of the hydrogenation catalyst reduce the dependency on volatile precious metal markets, stabilizing the cost structure over the long term. This lean manufacturing approach ensures that the final product remains price-competitive even when raw material costs fluctuate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as cyclopentanone and n-heptanal ensures a robust and resilient supply chain, as these feedstocks are widely available from multiple global suppliers. The robustness of the reaction conditions, which tolerate standard industrial pressure and temperature ranges, reduces the risk of batch failures due to equipment limitations or minor process deviations. This reliability translates into consistent lead times and the ability to secure long-term supply agreements with key customers in the food and beverage sector. Moreover, the simplified process flow reduces the complexity of logistics and inventory management, further strengthening supply chain continuity.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations that can be easily expanded from pilot plant to multi-ton production capacities. The reduction in solvent volume and the minimization of waste streams through efficient catalyst recovery contribute to a lower environmental footprint, facilitating easier compliance with local and international environmental protection laws. The use of established oxidation chemistries ensures that safety protocols are well-understood and manageable, reducing the regulatory burden associated with novel or hazardous reagents. This combination of scalability and compliance makes the technology an ideal candidate for sustainable industrial expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent specifications and practical engineering considerations to assist decision-makers in evaluating the feasibility of this route. Understanding these details is crucial for assessing the potential impact on your current production capabilities and cost structures.
Q: How does the one-pot method improve yield compared to traditional stepwise synthesis?
A: The patented one-pot method combines aldol condensation and catalytic hydrogenation into a single operational unit, eliminating the need to isolate unstable intermediates. This integration reduces material loss during transfer and purification, resulting in a documented yield increase of 3-4% compared to conventional stepwise processes.
Q: What are the scalability advantages of this synthesis route for industrial production?
A: The process utilizes standard industrial conditions, such as hydrogen pressures between 2.0-5.0 MPa and temperatures up to 100°C, which are easily manageable in large-scale reactors. Furthermore, the requirement for only two rectification steps significantly lowers energy consumption and simplifies the downstream processing infrastructure.
Q: Can the catalyst be recycled to reduce manufacturing costs?
A: Yes, the protocol explicitly includes a filtration step at 60-70°C after the hydrogenation reaction, allowing for the recovery and reuse of the precious metal catalyst (Pd/C, Pt, or Ni). This recyclability is a critical factor in minimizing the cost of goods sold (COGS) for high-volume production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Delta-Dodecalactone Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex laboratory innovations like the one-pot tandem catalysis method into robust commercial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in large-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of delta-dodecalactone meets the highest standards required for food and fragrance applications. Our commitment to technical excellence allows us to navigate the nuances of pressure hydrogenation and oxidative ring expansion with precision, delivering consistent quality that our global partners rely on.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through advanced process engineering. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our capabilities can support your long-term growth and product development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
