Advanced Nickel-Catalyzed Cross-Dimerization for High-Purity Methyl 4-Pentenoate Production
The chemical landscape for producing valuable intermediates is constantly evolving, driven by the need for higher selectivity and more sustainable catalytic processes. Patent CN1154630C introduces a groundbreaking methodology for the production of chemical intermediates through the cross-dimerization of two compounds containing carbon-carbon double bonds. Specifically, this intellectual property details a highly selective route to synthesize methyl 4-pentenoate (M4P), a crucial intermediate for the production of adipic acid and subsequently Nylon 6,6. The core innovation lies in the utilization of a specialized nickel(II) catalyst complex, structured as [QNiL1L2]+X-, which operates under remarkably mild conditions compared to traditional oligomerization methods. This technology represents a significant leap forward for manufacturers seeking a reliable fine chemical intermediates supplier capable of delivering high-purity precursors with reduced environmental impact and enhanced process efficiency.
The significance of this patent extends beyond mere academic interest; it addresses critical pain points in industrial synthesis where selectivity often competes with yield. By employing a catalyst system that incorporates an eta-3 carbon-based allyl group, a trisubstituted phosphine ligand, and a weakly coordinating ligand, the process achieves exceptional control over the reaction pathway. This level of control is paramount for R&D directors focused on impurity profiles, as it minimizes the formation of unwanted branched isomers or homopolymerization byproducts. For procurement managers and supply chain heads, the implication is clear: a more robust, scalable, and cost-effective manufacturing route for a high-demand commodity chemical derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the dimerization, oligomerization, and polymerization of olefins using nickel catalysts have been well-documented, yet they often suffer from significant drawbacks regarding selectivity and operational complexity. Traditional methods frequently rely on neutral nickel complexes or systems that lack the precise electronic tuning necessary to differentiate between cross-dimerization and homopolymerization. In many conventional processes, the reaction of ethylene with functionalized olefins like methyl acrylate results in a complex mixture of products, including linear and branched dimers, trimers, and substantial amounts of polymeric waste. This lack of specificity necessitates extensive and costly downstream purification steps to isolate the desired methyl 4-pentenoate, thereby inflating the overall cost of production and reducing the effective yield. Furthermore, older catalytic systems often require harsher reaction conditions, such as elevated temperatures and pressures, which can degrade sensitive functional groups and pose safety risks in large-scale commercial operations.
The Novel Approach
The novel approach disclosed in CN1154630C fundamentally alters the catalytic landscape by introducing a cationic nickel(II) species stabilized by weakly coordinating anions. This structural modification significantly enhances the electrophilicity of the metal center, facilitating the selective insertion of ethylene and methyl acrylate into the growing chain while suppressing unwanted side reactions. The use of bulky, weakly coordinating anions such as [B(3,5-(CF3)2C6H3)4]- prevents the anion from binding tightly to the nickel, leaving the coordination sites available for the substrates. This results in a highly selective transformation where methyl 4-pentenoate is formed with superior regioselectivity compared to prior art. The process operates efficiently at temperatures between 15-60°C and moderate pressures, offering a safer and more energy-efficient alternative to traditional high-pressure oligomerization. This breakthrough enables cost reduction in fine chemical intermediates manufacturing by streamlining the synthesis workflow and minimizing waste generation.
Mechanistic Insights into Nickel-Catalyzed Cross-Dimerization
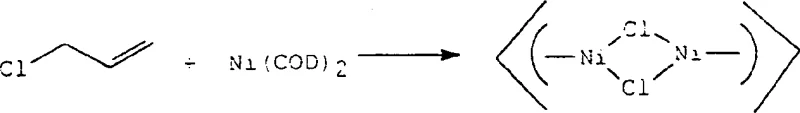
The mechanistic elegance of this process lies in the precise orchestration of ligand effects around the nickel center. The catalyst precursor is typically generated from nickel(0) sources like Ni(COD)2 reacting with allyl halides to form a diallyl-dichloro-dinickel(II) complex. This intermediate is then further modified with specific phosphine ligands, such as tricyclohexylphosphine (PCy3), and treated with a boron compound to abstract a halide and generate the active cationic species. The presence of the weakly coordinating ligand L2, which can be derived from the solvent itself (e.g., diethyl ether or acetonitrile), ensures that the catalyst remains soluble and active without blocking the substrate coordination sites. This delicate balance allows for the continuous turnover of the catalyst, achieving high turnover numbers (TO) as demonstrated in the patent examples, where values exceeding 300 were observed under optimized conditions.
Impurity control is inherently built into the catalyst design. The steric bulk of the trisubstituted phosphine ligand and the electronic properties of the weakly coordinating anion work in tandem to favor the formation of the linear 4-pentenoate isomer over the thermodynamically stable internal isomers (2-pentenoate or 3-pentenoate). GC-MS analysis of the reaction mixtures confirms that the selectivity for methyl 4-pentenoate can exceed 96%, with minimal detection of branched or internal isomers. This high degree of chemoselectivity is critical for pharmaceutical and polymer applications where trace impurities can affect downstream processing or final product performance. By minimizing the formation of homopolymerization products like dimethyl hexenedioate, the process ensures a cleaner crude reaction product, simplifying the isolation of high-purity methyl 4-pentenoate and reducing the burden on purification infrastructure.
How to Synthesize Methyl 4-Pentenoate Efficiently
The synthesis of methyl 4-pentenoate via this patented route involves a straightforward yet technically precise sequence of operations designed to maximize catalyst activity and product selectivity. The process begins with the preparation of the active nickel catalyst under strictly anhydrous and anaerobic conditions, typically within a glovebox or using Schlenk line techniques to prevent catalyst deactivation by oxygen or moisture. Once the catalyst is generated, it is introduced to a reaction vessel containing the substrates—ethylene and methyl acrylate—in a suitable non-protic solvent such as dichloromethane or chlorobenzene. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and pressure settings required to replicate the high efficiency reported in the patent data.
- Prepare the cationic nickel(II) catalyst complex by reacting an allyl-nickel halide precursor with a trisubstituted phosphine ligand and a weakly coordinating anion source.
- Contact ethylene and methyl acrylate in the presence of the catalyst under an oxygen-free and water-free atmosphere at temperatures between 15-60°C.
- Recover the crude reaction product and optionally isolate high-purity methyl 4-pentenoate through standard separation techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers substantial strategic benefits that extend beyond simple yield improvements. The ability to utilize commodity chemicals like ethylene and methyl acrylate as feedstocks ensures a stable and cost-effective raw material base, mitigating the risks associated with volatile specialty chemical markets. The mild operating conditions reduce energy consumption and equipment stress, leading to lower operational expenditures and extended asset life. Furthermore, the high selectivity of the process minimizes the generation of hazardous waste and byproducts, aligning with increasingly stringent environmental regulations and sustainability goals. This translates to significant cost savings in waste disposal and compliance management, making the overall supply chain more resilient and economically viable.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals other than nickel, combined with the high turnover numbers achieved by the catalyst, drastically reduces the catalyst loading required per unit of product. This efficiency directly lowers the variable cost of goods sold. Additionally, the suppression of homopolymerization means that less feedstock is wasted on unwanted polymeric byproducts, maximizing the atom economy of the process. The simplified downstream processing due to high product purity further reduces the costs associated with distillation and purification, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals like ethylene and methyl acrylate, manufacturers can secure a consistent supply of raw materials without being dependent on niche suppliers. The robustness of the catalyst system allows for flexible production scheduling, as the reaction can be tuned by adjusting pressure and temperature within a broad range. This flexibility ensures that production targets can be met even in the face of minor fluctuations in utility availability or feedstock quality, thereby enhancing the reliability of delivery to downstream customers in the nylon and polymer industries.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch reactors with conditions that are easily translatable to continuous flow or larger batch vessels. The use of non-protic solvents and the absence of heavy metal contaminants in the final product simplify the regulatory approval process for new applications. Moreover, the reduced formation of polymeric waste aligns with green chemistry principles, lowering the environmental footprint of the manufacturing site. This compliance advantage facilitates smoother audits and certifications, positioning the manufacturer as a preferred partner for environmentally conscious global corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed cross-dimerization technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of adopting this route for commercial production. Understanding these nuances is essential for technical teams assessing the integration of this process into existing manufacturing lines.
Q: What is the primary advantage of the nickel catalyst described in CN1154630C?
A: The primary advantage is the high selectivity for the linear cross-dimer product (methyl 4-pentenoate) over homopolymerization products or branched isomers, achieved through the use of weakly coordinating anions and specific phosphine ligands.
Q: What are the typical reaction conditions for this cross-dimerization process?
A: The process operates under mild conditions, typically at temperatures ranging from 15°C to 60°C and pressures from atmospheric up to approximately 689.5 kPa, utilizing non-protic organic solvents.
Q: Why is methyl 4-pentenoate considered a valuable intermediate?
A: Methyl 4-pentenoate serves as a critical precursor for the production of adipic acid, which is a key starting material for manufacturing Nylon 6,6, making it essential for the polymer and engineering plastics industries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-Pentenoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic processes like the one described in CN1154630C for the production of high-value intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of methyl 4-pentenoate meets the exacting standards required for nylon 6,6 synthesis and other demanding applications. We understand that consistency and reliability are the cornerstones of a successful supply chain, and we are dedicated to delivering both.
We invite you to collaborate with us to explore how this technology can optimize your manufacturing costs and improve your product quality. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage in the global market for fine chemical intermediates through innovation and partnership.
