Advanced N-Trifluoromethylation Technology for High-Purity Pharmaceutical Intermediates
Introduction to Next-Generation Trifluoromethylation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate trifluoromethyl groups into complex molecular architectures, driven by the profound impact of fluorine on metabolic stability and bioavailability. Patent CN111302968B discloses a groundbreaking synthetic method for producing amide nitrogen trifluoromethyl compounds, addressing critical bottlenecks in current manufacturing workflows. This technology enables the direct construction of the N-CF3 bond from readily available amide or sulfonamide precursors through an oxidative trifluoromethylation reaction. By leveraging a sophisticated catalytic system involving silver salts, pyridine ligands, and mild oxidants, this approach eliminates the need for hazardous reagents and harsh conditions that have historically plagued this chemical transformation. The innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to enhance their portfolio of fluorinated building blocks.
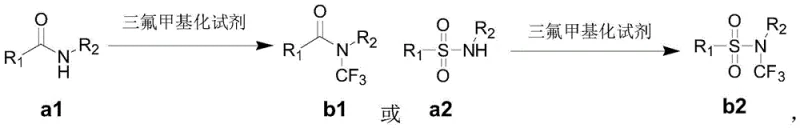
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
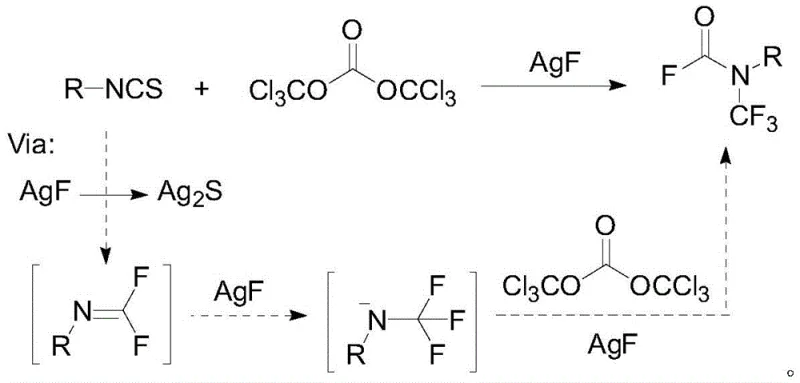
Historically, the synthesis of N-trifluoromethyl amides has been fraught with significant operational and economic challenges that hinder widespread industrial adoption. Early methodologies, such as those reported by the Rozen group, relied on the use of bromine trifluoride (BrF3), an extremely aggressive and hazardous fluorinating agent that requires specialized handling infrastructure and poses severe safety risks to personnel and equipment. Furthermore, more recent advancements, while safer, introduced prohibitive cost barriers; for instance, the Schoenebeck group reported a route utilizing bis(trichloromethyl) carbonate and isothiocyanates which indirectly realized the synthesis but necessitated the use of five equivalents of expensive silver fluoride (AgF). This stoichiometric reliance on precious metal salts not only inflates the raw material costs drastically but also generates substantial heavy metal waste, complicating downstream purification and environmental compliance efforts for any agrochemical intermediate supplier attempting to scale these processes.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a catalytic manifold that fundamentally reshapes the economic and safety profile of the reaction. By employing a combination of trimethylsilyl trifluoromethane (TMSCF3) as the trifluoromethyl source, Selectfluor as a mild oxidant, and a catalytic amount of silver trifluoromethanesulfonate paired with a pyridine ligand, the reaction proceeds efficiently at room temperature. This method allows for the direct conversion of amide substrates into the desired N-trifluoromethyl products without the need for multi-step indirect pathways or excessive reagent loading. The operational simplicity is further enhanced by the use of common organic solvents like dichloromethane and chlorobenzene, facilitating easy workup and isolation. This streamlined protocol offers a viable pathway for cost reduction in API manufacturing by minimizing reagent consumption and simplifying the overall process flow.
Mechanistic Insights into Silver-Catalyzed Oxidative Trifluoromethylation
The success of this transformation hinges on the intricate interplay between the silver catalyst, the pyridine ligand, and the oxidant, which together facilitate the generation of the active trifluoromethylating species under mild conditions. Mechanistically, the silver salt acts as a Lewis acid and a redox mediator, activating the silicon-based trifluoromethyl reagent while the pyridine ligand stabilizes the silver center, preventing premature aggregation or deactivation. The oxidant, Selectfluor, plays a crucial role in regenerating the active silver species and driving the thermodynamic equilibrium towards the formation of the N-CF3 bond. This catalytic cycle ensures that the silver component is turned over multiple times, thereby maximizing atom economy and reducing the burden of heavy metal residues in the final product. Understanding this mechanism is vital for R&D teams looking to adapt this chemistry to novel substrates, as it highlights the importance of ligand selection and oxidant compatibility in maintaining high reaction efficiency.
Furthermore, the reaction conditions are specifically tuned to suppress common side reactions that typically plague nucleophilic trifluoromethylation attempts, such as O-trifluoromethylation or decomposition of the sensitive N-CF3 bond. The use of a non-coordinating solvent system helps to maintain the reactivity of the electrophilic intermediates while minimizing competitive solvation effects that could retard the reaction rate. Impurity control is inherently built into the design, as the mild room temperature conditions prevent thermal degradation of sensitive functional groups often present in complex pharmaceutical intermediates. This level of selectivity ensures that the resulting crude product profiles are cleaner, reducing the load on purification units and contributing to higher overall yields. For quality assurance teams, this translates to more consistent batch-to-batch reproducibility and easier adherence to stringent purity specifications required for clinical grade materials.
How to Synthesize N-Trifluoromethyl Amides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the order of addition and the maintenance of an inert atmosphere to ensure optimal catalyst performance. The general procedure involves charging the reactor with the amide substrate, the oxidant, the silver salt, and the fluorinating agent, followed by the addition of the solvent system and the trifluoromethylating reagent under nitrogen or argon protection. The reaction is then allowed to stir at ambient temperature, monitoring progress via standard analytical techniques until conversion is complete. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction vessel under inert gas protection and add the amide substrate, oxidant (Selectfluor), silver salt (AgOTf), and fluorinating agent (CsF).
- Introduce the organic solvent mixture (DCM/Chlorobenzene), trifluoromethylating agent (TMSCF3), and pyridine ligand to the system.
- Stir the mixture at room temperature until completion, then quench with water, extract, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers compelling advantages that directly address the pain points of sourcing complex fluorinated intermediates. The shift from stoichiometric to catalytic silver usage represents a massive reduction in the bill of materials, as silver salts are among the most expensive components in organometallic synthesis. By eliminating the need for five equivalents of silver fluoride, the process significantly lowers the entry barrier for production, making high-value N-trifluoromethyl compounds more accessible for drug development programs. Additionally, the use of commercially available and stable reagents like TMSCF3 and Selectfluor ensures a reliable supply chain, mitigating the risks associated with sourcing exotic or hazardous chemicals that often face regulatory shipping restrictions.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the drastic reduction in precious metal consumption. Traditional routes requiring stoichiometric silver fluoride impose a heavy financial burden due to the high market price of silver and the loss of material during workup. In contrast, this catalytic system utilizes silver salts in substoichiometric quantities, effectively recycling the metal within the reaction cycle. This efficiency gain translates directly to lower production costs per kilogram, allowing for more competitive pricing strategies in the global market. Furthermore, the simplified workup procedure reduces solvent usage and labor hours, contributing to additional operational savings that accumulate significantly over large-scale campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals rather than specialized reagents. Reagents such as cesium fluoride, silver triflate, and pyridine derivatives are widely produced by multiple global vendors, ensuring that production is not held hostage by single-source suppliers. The stability of these materials also allows for longer shelf-life and easier storage requirements, reducing inventory turnover pressure. For supply chain heads, this means reduced lead time for high-purity pharmaceutical intermediates, as the risk of raw material stockouts is minimized. The robustness of the reaction conditions further ensures that manufacturing can proceed without interruption due to sensitive environmental controls, guaranteeing consistent delivery schedules to downstream clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for commercial scale-up of complex polymer additives and fine chemicals. The absence of cryogenic requirements and the use of ambient temperature conditions simplify reactor design and energy consumption profiles. Moreover, the reduction in heavy metal waste aligns with increasingly stringent environmental regulations regarding effluent discharge. By generating less hazardous waste, facilities can lower their waste treatment costs and reduce their environmental footprint. This sustainability angle is increasingly important for corporate social responsibility goals and facilitates smoother regulatory approvals for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the scope and limitations of the method. Understanding these details is essential for project managers evaluating the feasibility of integrating this chemistry into existing production lines.
Q: What are the primary advantages of this silver-catalyzed method over traditional stoichiometric silver fluoride routes?
A: The primary advantage is the drastic reduction in reagent cost and waste. Traditional methods often require five equivalents of expensive silver fluoride, whereas this catalytic system uses substoichiometric amounts of silver salts alongside readily available oxidants, significantly lowering the bill of materials.
Q: Is this trifluoromethylation process suitable for large-scale manufacturing?
A: Yes, the process is designed for scalability. It operates at room temperature, avoiding the need for cryogenic cooling or high-pressure equipment, and utilizes common organic solvents, making the transition from laboratory to commercial production straightforward and safe.
Q: What types of substrates are compatible with this N-trifluoromethylation protocol?
A: The method demonstrates broad substrate scope, successfully converting various amides and sulfonamides bearing different functional groups, including halogens, ethers, and esters, into their corresponding N-CF3 derivatives with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Trifluoromethyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this silver-catalyzed trifluoromethylation route for the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including specialized containment systems and rigorous QC labs capable of verifying stringent purity specifications for sensitive N-CF3 containing molecules. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. Our technical team is ready to perform a Customized Cost-Saving Analysis tailored to your specific target molecule, identifying opportunities to leverage this efficient synthesis method. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us help you accelerate your timeline to market with reliable, cost-effective, and scalable chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
