Advanced Silver-Catalyzed Synthesis of 9,10-Phenanthrenedicarboxylates for Commercial Scale-Up
Introduction to Novel Phenanthrene Synthesis Technology
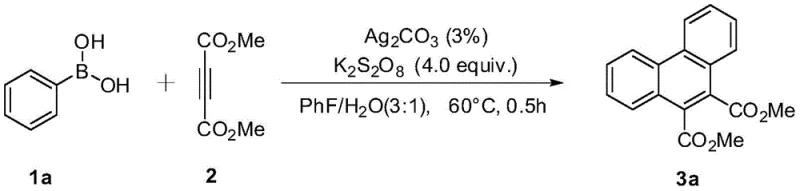
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing polycyclic aromatic hydrocarbons, particularly the phenanthrene scaffold, which serves as a critical backbone in numerous bioactive molecules and advanced materials. Patent CN110683949A introduces a groundbreaking approach to synthesizing 9,10-phenanthrenedicarboxylate compounds, addressing long-standing challenges in efficiency and scalability. This technology leverages a silver-catalyzed oxidative cyclization strategy that operates under remarkably mild conditions, specifically utilizing a fluorobenzene and water biphasic system at 60°C. Unlike traditional methods that often require toxic heavy metals or extreme thermal energy, this innovation utilizes readily available phenylboronic acids and dimethyl acetylenedicarboxylate as starting materials. The significance of this development lies in its ability to streamline the supply chain for high-purity pharmaceutical intermediates, offering a reliable phenanthrene dicarboxylate supplier pathway that minimizes environmental impact while maximizing yield. By shifting the paradigm from multi-step sequences to a concise one-pot reaction, this patent provides a compelling solution for manufacturers aiming to reduce lead time for high-purity intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the phenanthrene core has relied heavily on transition metal-catalyzed cross-coupling reactions, such as those employing palladium, rhodium, or iron complexes. While effective in academic settings, these conventional routes present significant hurdles for industrial adoption, primarily due to the high cost and scarcity of noble metal catalysts like palladium and rhodium. Furthermore, many established protocols necessitate the pre-functionalization of substrates, requiring multi-step synthetic sequences to generate specialized aryl halides or organometallic reagents before the cyclization event can occur. These additional steps not only increase the overall production cost but also introduce opportunities for yield loss and impurity generation at each stage. The harsh reaction conditions often associated with these methods, including high temperatures and strong bases, can lead to poor atom economy and complicate downstream purification processes. Consequently, the commercial scale-up of complex phenanthrene derivatives via these traditional pathways is often hindered by economic inefficiencies and regulatory concerns regarding heavy metal residues in final products.
The Novel Approach
In stark contrast, the methodology disclosed in CN110683949A represents a significant leap forward by utilizing an inexpensive silver carbonate catalyst in conjunction with potassium persulfate as a green oxidant. This novel approach eliminates the need for pre-functionalized substrates, allowing for the direct use of commercially abundant phenylboronic acids and alkynes in a single reaction vessel. The reaction proceeds efficiently at a moderate temperature of 60°C, drastically reducing energy consumption compared to high-thermal processes. Optimization studies within the patent reveal that a specific solvent mixture of fluorobenzene and water in a 3:1 ratio is critical for achieving optimal conversion rates, as demonstrated by the improved yields in specific experimental examples. This one-pot strategy not only simplifies the operational workflow but also enhances the overall sustainability of the manufacturing process by minimizing solvent waste and avoiding toxic reagents. The result is a streamlined protocol that offers substantial cost savings and operational simplicity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Silver-Catalyzed Oxidative Cyclization
The underlying mechanism of this transformation involves a sophisticated interplay between the silver catalyst and the persulfate oxidant to facilitate the formation of new carbon-carbon bonds. It is hypothesized that the silver(I) species interacts with the alkyne moiety of the dimethyl acetylenedicarboxylate, activating it towards nucleophilic attack or radical addition. Simultaneously, the persulfate oxidant likely generates sulfate radical anions that assist in the oxidation of the silver center or the direct activation of the boronic acid species, promoting the crucial C-H activation step required for ring closure. This radical-mediated pathway allows for the construction of the rigid phenanthrene skeleton under mild conditions that would otherwise be inaccessible via ionic mechanisms. The presence of water in the solvent system plays a dual role, acting both as a co-solvent to dissolve the inorganic oxidant and potentially participating in the proton transfer steps necessary for rearomatization. Understanding these mechanistic nuances is vital for R&D directors aiming to further optimize the process or adapt it for novel substrate classes, ensuring that the reaction remains robust against variations in raw material quality.
From an impurity control perspective, the mildness of this silver-catalyzed system offers distinct advantages over harsher acidic or basic conditions. The selective nature of the silver catalyst minimizes side reactions such as polymerization of the alkyne or homocoupling of the boronic acid, which are common pitfalls in traditional coupling chemistries. The use of a biphasic solvent system further aids in impurity management, as inorganic byproducts and catalyst residues tend to partition into the aqueous phase, simplifying the isolation of the organic product. This inherent selectivity translates to a cleaner crude reaction profile, reducing the burden on downstream purification units like column chromatography or crystallization. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process, ultimately leading to a higher purity final product that meets stringent regulatory specifications for pharmaceutical applications.
How to Synthesize Dimethyl 9,10-phenanthrenedicarboxylate Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and readily accessible reagents. To achieve the high yields reported in the patent, precise control over the stoichiometry of the oxidant and the ratio of the biphasic solvent is essential. The process begins with the sequential addition of the phenylboronic acid derivative, the alkyne acceptor, the silver carbonate catalyst, and the potassium persulfate oxidant into a pressure-resistant reaction vessel. Following the addition of the fluorobenzene and water solvent mixture, the system is sealed and heated to maintain a constant temperature of 60°C for a duration of 0.5 hours. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results.
- Combine phenylboronic acid, dimethyl acetylenedicarboxylate, silver carbonate catalyst, and potassium persulfate oxidant in a pressure tube.
- Add a mixed solvent system of fluorobenzene and water (3: 1 ratio) to the reaction mixture.
- Heat the sealed tube to 60°C for 0.5 hours, then cool and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this silver-catalyzed technology presents a compelling value proposition centered around cost efficiency and supply security. The primary driver for cost reduction is the substitution of expensive noble metal catalysts with affordable silver salts, which significantly lowers the raw material cost per kilogram of product. Additionally, the elimination of multi-step substrate preparation reduces the number of unit operations required, thereby decreasing labor costs, utility consumption, and equipment occupancy time. The short reaction time of merely 0.5 hours allows for higher throughput in existing reactor fleets, effectively increasing production capacity without the need for capital investment in new infrastructure. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to fluctuations in the pricing of specialty chemicals.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the use of commodity-grade starting materials such as phenylboronic acids and dimethyl acetylenedicarboxylate, which are available in bulk quantities from multiple global suppliers. By avoiding the use of proprietary ligands or specialized palladium catalysts, the process removes significant cost barriers associated with intellectual property licensing and exclusive supply agreements. Furthermore, the simplified workup procedure, which relies on standard column chromatography or crystallization rather than complex extraction sequences, reduces solvent consumption and waste disposal costs. This holistic approach to cost optimization ensures that the final product can be offered at a competitive price point while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available reagents that are not subject to the same geopolitical or logistical constraints as rare earth metals or exotic catalysts. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material specifications, reducing the risk of batch failures due to supplier quality issues. Moreover, the scalability of the one-pot reaction allows for flexible production scheduling, enabling manufacturers to respond quickly to changes in market demand. This agility is crucial for maintaining reliable delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors, where just-in-time inventory management is often critical.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The use of water as a co-solvent and the absence of volatile organic chlorinated solvents in the reaction mixture reduce the environmental footprint of the manufacturing process. The mild reaction temperature minimizes energy usage, contributing to lower carbon emissions associated with production. These sustainability credentials are increasingly important for corporate social responsibility reporting and can provide a competitive advantage when bidding for contracts with environmentally conscious multinational corporations. The ease of scaling this process from gram to ton scale ensures that supply can grow in tandem with market demand without encountering significant technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the key advantages of this silver-catalyzed method over traditional palladium routes?
A: This method eliminates the need for expensive palladium catalysts and harsh conditions, utilizing cheap silver carbonate and mild temperatures (60°C) for a more cost-effective and environmentally friendly process.
Q: Can this synthesis tolerate various functional groups on the phenylboronic acid substrate?
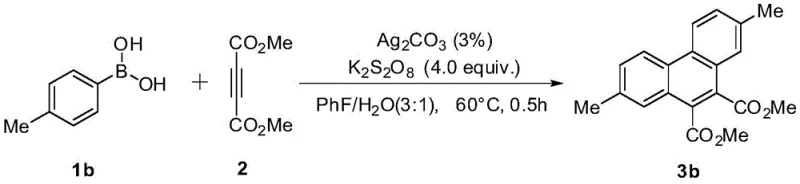
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like fluorine, yielding diverse derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The one-pot nature, short reaction time (0.5h), and simple aqueous workup make it highly scalable, reducing operational complexity and facilitating commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Phenanthrenedicarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value intermediates like 9,10-phenanthrenedicarboxylates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of silver-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to guarantee product quality. We are committed to delivering solutions that not only meet technical benchmarks but also drive value through operational excellence and supply chain stability.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline and optimize your manufacturing costs.
