Advanced Synthesis of 4-Alkoxy Acetoacetate Intermediates for Scalable Antiretroviral Drug Manufacturing
The pharmaceutical industry's relentless pursuit of effective antiretroviral therapies has placed significant demand on the supply chain of critical intermediates, particularly for integrase inhibitors like Dolutegravir. Patent CN111004121A, published in April 2020, introduces a groundbreaking preparation method for 4-alkoxy acetoacetate compounds, which serve as pivotal building blocks in the synthesis of next-generation HIV/AIDS medications. This technology addresses long-standing bottlenecks in the manufacturing of these esters by replacing expensive, scarce starting materials with commodity chemicals, thereby stabilizing the supply chain for global pharmaceutical manufacturers. The disclosed method not only simplifies the synthetic route but also significantly enhances the purity profile of the final intermediate, which is crucial for meeting the stringent regulatory requirements of active pharmaceutical ingredient (API) production. By shifting away from complex coupling reagents and heavy metal catalysts, this innovation represents a substantial leap forward in green chemistry and process efficiency for the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-alkoxy acetoacetates has been plagued by economic and logistical challenges that hinder large-scale production. One prevalent conventional method relies on the nucleophilic substitution of 4-chloroacetoacetate, as illustrated in the reaction scheme below. While chemically straightforward, this route is fundamentally flawed for commercial scale-up due to the prohibitive cost and limited availability of the 4-chloroacetoacetate starting material. The scarcity of suppliers for this specific chloro-ester creates a fragile supply chain, leading to volatile pricing and potential production delays for downstream drug manufacturers. Furthermore, alternative routes utilizing coupling agents such as CDI (carbonyldiimidazole) and magnesium chloride suffer from low atom economy and high reagent costs, rendering them economically unviable for tonnage-scale manufacturing. Another traditional approach involves the use of zinc powder in benzene, which generates substantial amounts of inorganic salt waste that is difficult and costly to treat, posing significant environmental compliance burdens for modern chemical plants.
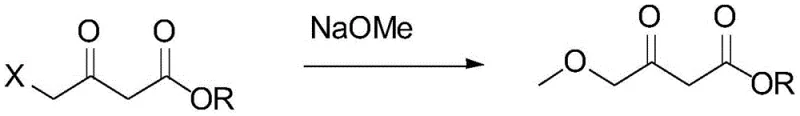
The Novel Approach
In stark contrast to these legacy methods, the novel process disclosed in the patent utilizes a readily available haloacetate, such as methyl chloroacetate, as the primary feedstock. This strategic shift to commodity raw materials drastically reduces the raw material cost basis and ensures a stable, continuous supply unaffected by niche market fluctuations. The new methodology employs a clever three-step cascade that avoids the use of hazardous heavy metals and expensive activating agents. Instead, it leverages standard base-catalyzed etherification followed by a controlled condensation and a final mild acid hydrolysis. This approach not only simplifies the operational complexity but also improves the overall yield and purity of the target 4-alkoxy acetoacetate. The reaction conditions are notably mild, operating at temperatures between 40°C and 100°C, which reduces energy consumption and enhances safety profiles compared to high-temperature or high-pressure alternatives. The specific reaction pathway is detailed in the schematic below, highlighting the transformation from simple esters to the complex beta-keto ester framework.
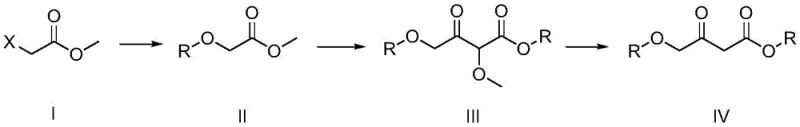
Mechanistic Insights into Base-Catalyzed Etherification and Hydrolysis
The core of this innovative synthesis lies in its sophisticated management of reactivity through a 'masking' strategy. The first step involves the nucleophilic substitution of a haloacetate (Compound I) with an alkoxide base to form an alkoxyacetate (Intermediate II). This etherification is conducted under controlled temperatures to prevent over-reaction or ester hydrolysis, ensuring high conversion rates. The subsequent step is a base-mediated condensation that constructs the beta-keto ester skeleton. Crucially, this condensation yields an alpha-alkoxy beta-keto ester (Intermediate III), where the alpha-position is protected by a methoxy group. This protection is vital because it prevents unwanted self-condensation or polymerization side reactions that typically plague direct acetoacetate syntheses. The final step involves a selective acid hydrolysis in an aqueous medium. Under these specific acidic conditions, the alpha-methoxy group is cleaved to reveal the methylene group, yielding the desired 4-alkoxy acetoacetate (Product IV). This hydrolysis is remarkably selective, leaving the ester functionalities intact while removing the masking group, which is a testament to the precise control over reaction kinetics achieved in this process.
From an impurity control perspective, this mechanism offers distinct advantages over direct alkylation methods. Traditional routes often struggle with poly-alkylation and the formation of regioisomers, which are difficult to separate and can compromise the safety profile of the final drug. By forming the carbon-carbon bond first in the presence of the protecting methoxy group, the process directs the reactivity exclusively towards the desired beta-keto structure. The final acid treatment acts as a purification step in itself, as many basic impurities formed during the condensation are neutralized or partitioned into the aqueous phase during workup. The result is a crude product with exceptionally high purity, often exceeding 98% after simple distillation, which significantly reduces the burden on downstream purification processes. This high level of chemical fidelity is essential for pharmaceutical intermediates, where trace impurities can trigger rigorous regulatory scrutiny and delay drug approval timelines.
How to Synthesize Methyl 4-Methoxyacetoacetate Efficiently
The practical implementation of this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and minimize byproduct formation. The process begins with the preparation of the alkoxyacetate intermediate, followed by the condensation reaction in a non-polar solvent like toluene to facilitate water removal and drive the equilibrium. The final hydrolysis step must be monitored closely to ensure complete deprotection without degrading the sensitive beta-keto ester motif. For a comprehensive guide on the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- React a haloacetate compound with a first base in a solvent to perform etherification and obtain an alkoxyacetate intermediate.
- Condense the alkoxyacetate intermediate with a second base in a second solvent to form an alpha-alkoxy beta-keto ester intermediate.
- Hydrolyze the intermediate in an aqueous acid solution to remove the alpha-alkoxy group and yield the final 4-alkoxy acetoacetate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The most significant advantage is the decoupling of production costs from volatile specialty chemical markets. By utilizing methyl chloroacetate and sodium methoxide—both of which are produced in massive quantities globally—the manufacturing process becomes insulated from the supply shocks that frequently affect niche intermediates like 4-chloroacetoacetate. This stability allows for more accurate long-term budgeting and contract negotiation, providing a competitive edge in the pricing of the final API. Furthermore, the elimination of expensive coupling reagents like CDI and heavy metals like zinc translates directly into a leaner cost structure, enabling significant cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the replacement of high-cost inputs with commodity chemicals. The avoidance of stoichiometric amounts of activating agents and the elimination of expensive metal powders remove major cost drivers from the bill of materials. Additionally, the simplified workup procedures reduce solvent consumption and waste disposal fees, further enhancing the overall cost efficiency. This structural cost advantage allows manufacturers to offer more competitive pricing to downstream pharmaceutical clients while maintaining healthy margins, creating a sustainable business model for long-term supply agreements.
- Enhanced Supply Chain Reliability: Supply chain resilience is dramatically improved by sourcing raw materials that have multiple global suppliers and established logistics networks. Unlike specialized reagents that may have single-source dependencies, the commodity nature of the starting materials ensures that production can continue uninterrupted even if one supplier faces difficulties. The robustness of the chemical process itself, with its tolerance for standard industrial equipment and mild conditions, further reduces the risk of unplanned downtime. This reliability is critical for pharmaceutical companies that require guaranteed continuity of supply to meet their own regulatory commitments and patient needs.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvents and conditions that are standard in multi-tonne chemical reactors. The absence of heavy metal waste streams simplifies environmental compliance and reduces the regulatory burden associated with wastewater treatment. This 'green' aspect of the synthesis not only aligns with corporate sustainability goals but also accelerates the permitting process for new manufacturing lines. The ability to scale from kilogram to metric tonne quantities without fundamental changes to the chemistry ensures that the supply can grow in lockstep with the market demand for the final antiretroviral medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of the method for potential adopters.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The new method utilizes cheap, commercially available haloacetates instead of expensive 4-chloroacetoacetate or coupling reagents like CDI. It avoids heavy metal waste (Zn) and offers higher purity suitable for API manufacturing.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process employs a unique 'masking' strategy where an alpha-methoxy group is introduced and subsequently removed via mild acid hydrolysis. This minimizes side reactions and decomposition common in direct condensation methods, resulting in purity levels exceeding 98%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses standard solvents like toluene and methanol and avoids hazardous reagents like zinc powder or expensive activators. The reaction conditions are mild (40-100°C), making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Alkoxy Acetoacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving medications. Our technical team has thoroughly analyzed the methodology described in CN111004121A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to market. We are committed to delivering 4-alkoxy acetoacetate compounds with stringent purity specifications, supported by our rigorous QC labs that ensure every batch meets the exacting standards of the global pharmaceutical industry. Our facility is equipped to handle the specific solvent systems and reaction conditions of this novel route, guaranteeing a consistent and reliable supply for your drug development programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
