Advanced One-Pot Synthesis of Sulfonylaminocarbonyl Triazolinone Salts for Global Agrochemical Supply Chains
Introduction to Advanced Herbicide Intermediate Manufacturing
The global demand for high-efficiency herbicides necessitates robust and scalable manufacturing processes for key active ingredients. Patent CN1301696A introduces a significant technological advancement in the production of sulfonylaminocarbonyl triazolinone salts, which serve as critical intermediates for herbicidally active compounds such as Flucarbazone-sodium and Propoxycarbazone-sodium. This intellectual property outlines a refined one-pot synthesis methodology that eliminates the need for isolating unstable or difficult-to-handle intermediate products, thereby streamlining the production workflow. By integrating the formation of the sulfonylaminocarbonyl triazolinone intermediate and its subsequent conversion into a salt within a single reaction vessel, the process addresses longstanding challenges in yield optimization and impurity control. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for evaluating supply chain resilience and cost-effectiveness in agrochemical manufacturing. The following analysis dissects the technical merits and commercial implications of this innovative approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for sulfonylaminocarbonyl triazolinones often involve multi-step sequences where the intermediate compound must be isolated, purified, and dried before proceeding to the salt formation stage. This conventional batch processing introduces several inefficiencies, including increased exposure of sensitive intermediates to environmental factors that may degrade product quality. Furthermore, the physical handling of intermediates requires additional unit operations such as filtration, washing, and secondary drying, which escalate both capital expenditure and operational costs. From a safety perspective, isolating reactive intermediates can pose hazards, particularly when dealing with large-scale batches where heat dissipation and containment are critical. The cumulative effect of these discrete steps often results in lower overall yields due to mechanical losses during transfer and purification, creating a bottleneck for manufacturers aiming to meet high-volume agricultural demands efficiently.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a telescoped one-pot strategy that seamlessly connects the acylation of the triazolinone ring with the subsequent neutralization to form the stable salt. 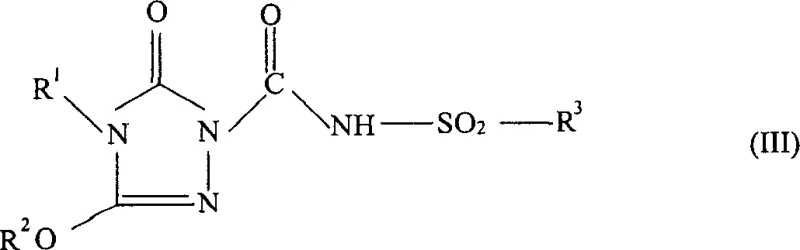 As illustrated in the reaction scheme, the process allows for the direct treatment of the reaction mixture containing the intermediate (Formula III) with a base to generate the final salt (Formula IV) without prior separation. This continuity not only minimizes the total processing time but also leverages the thermal energy of the initial reaction to drive the subsequent salt formation, potentially reducing external heating requirements. By maintaining the intermediate in solution, the risk of decomposition is mitigated, and the overall material throughput is maximized. This approach is particularly advantageous for producing compounds like Flucarbazone-sodium, where maintaining high chemical purity is paramount for herbicidal efficacy.
As illustrated in the reaction scheme, the process allows for the direct treatment of the reaction mixture containing the intermediate (Formula III) with a base to generate the final salt (Formula IV) without prior separation. This continuity not only minimizes the total processing time but also leverages the thermal energy of the initial reaction to drive the subsequent salt formation, potentially reducing external heating requirements. By maintaining the intermediate in solution, the risk of decomposition is mitigated, and the overall material throughput is maximized. This approach is particularly advantageous for producing compounds like Flucarbazone-sodium, where maintaining high chemical purity is paramount for herbicidal efficacy.
Mechanistic Insights into One-Pot Salt Formation
The core chemical transformation involves the nucleophilic attack of the substituted triazolinone (Formula I) on the sulfonyl isocyanate (Formula II), resulting in the formation of the sulfonylaminocarbonyl linkage.  This initial step typically proceeds at moderate temperatures ranging from 0°C to 45°C in solvents such as methyl isobutyl ketone (MIBK) or xylene. The reaction kinetics are favorable under these conditions, allowing for complete conversion within 1 to 8 hours. Crucially, the patent emphasizes that the intermediate does not need to be crystallized or filtered; instead, the reaction mixture is directly subjected to basic conditions. The addition of bases like sodium hydroxide or alkoxides facilitates the deprotonation of the acidic NH proton in the urea-like linkage, generating the anionic species that associates with the metal cation to form the stable salt. This mechanism ensures that the final product precipitates or remains in a manageable phase for isolation, depending on the specific solvent system employed.
This initial step typically proceeds at moderate temperatures ranging from 0°C to 45°C in solvents such as methyl isobutyl ketone (MIBK) or xylene. The reaction kinetics are favorable under these conditions, allowing for complete conversion within 1 to 8 hours. Crucially, the patent emphasizes that the intermediate does not need to be crystallized or filtered; instead, the reaction mixture is directly subjected to basic conditions. The addition of bases like sodium hydroxide or alkoxides facilitates the deprotonation of the acidic NH proton in the urea-like linkage, generating the anionic species that associates with the metal cation to form the stable salt. This mechanism ensures that the final product precipitates or remains in a manageable phase for isolation, depending on the specific solvent system employed.
Impurity control is another critical aspect of this mechanistic pathway, heavily influenced by the pH of the reaction medium during the salt formation step. Experimental data within the patent indicates that maintaining the pH between 6 and 7 is optimal for achieving purities around 98%. Deviations towards highly alkaline conditions (pH > 10) can lead to the formation of byproducts or degradation of the sensitive triazolinone ring, resulting in lower purity profiles. Therefore, the precise dosing of the base, often monitored via in-process controls, is vital for ensuring the quality of the final agrochemical intermediate. This level of control allows manufacturers to consistently produce material that meets stringent regulatory specifications for residue limits and active content, which is a key concern for downstream formulators.
How to Synthesize Sulfonylaminocarbonyl Triazolinone Salts Efficiently
Implementing this synthesis route requires careful attention to solvent selection and moisture control, particularly during the initial coupling reaction. The process begins with the preparation of a slurry or solution of the substituted triazolinone in a dry organic solvent, often achieved through azeotropic distillation to remove trace water that could hydrolyze the isocyanate reagent. Once the reaction environment is optimized, the sulfonyl isocyanate is added gradually to manage exotherms and ensure uniform mixing. Following the completion of the coupling reaction, confirmed by chromatographic monitoring, the system is transitioned to the salt formation phase by introducing water and the base. The detailed standardized synthesis steps for replicating this high-yield process are outlined below.
- React substituted triazolinone (Formula I) with sulfonyl isocyanate (Formula II) in a solvent like MIBK at 0°C to 45°C to form the intermediate.
- Without isolating the intermediate, add water and a base such as sodium hydroxide to the reaction mixture.
- Control the pH of the mixture between 6 and 7 to ensure high purity conversion to the final salt (Formula IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot technology offers tangible benefits that extend beyond simple chemical yield improvements. The elimination of the intermediate isolation step fundamentally alters the cost structure of manufacturing these herbicide intermediates. By removing the need for separate filtration, drying, and re-dissolution equipment, facilities can reduce their fixed asset requirements and lower maintenance overheads. Additionally, the reduction in unit operations translates to decreased labor intensity and shorter cycle times, allowing for faster turnover of production batches. This efficiency gain is crucial for meeting seasonal demand spikes in the agrochemical sector without requiring massive expansions in plant capacity. The ability to produce high-purity material in fewer steps also reduces the consumption of auxiliary materials such as filter aids and washing solvents, contributing to a leaner and more sustainable operation.
- Cost Reduction in Manufacturing: The streamlined nature of the one-pot process directly impacts the cost of goods sold by minimizing solvent usage and energy consumption. Since the intermediate is not isolated, there is no need for the extensive solvent exchanges or drying cycles that typically characterize multi-step syntheses. This reduction in solvent volume not only lowers raw material costs but also decreases the load on solvent recovery systems, leading to significant operational savings. Furthermore, the improved yield consistency reduces the amount of starting material required per kilogram of final product, optimizing the utilization of expensive reagents like sulfonyl isocyanates. These cumulative efficiencies result in a more competitive pricing structure for the final agrochemical intermediate.
- Enhanced Supply Chain Reliability: Simplifying the manufacturing process inherently reduces the number of potential failure points in the supply chain. With fewer transfer steps and unit operations, the risk of cross-contamination or mechanical delays is substantially diminished. This robustness ensures a more predictable delivery schedule, which is vital for downstream customers who rely on just-in-time inventory models. Moreover, the use of common industrial solvents like MIBK and xylene ensures that raw material sourcing remains stable and less susceptible to market volatility compared to specialized or exotic reagents. This stability allows supply chain planners to forecast production capacities with greater confidence and secure long-term contracts with key agricultural clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to commercial-scale reactors. The moderate temperature range (0°C to 45°C) reduces the energy burden associated with extreme heating or cooling, aligning with modern green chemistry principles. Additionally, the reduced generation of solid waste from filtration steps lowers the environmental footprint of the manufacturing site. By minimizing the release of volatile organic compounds through efficient solvent recycling and reduced handling, the process supports compliance with increasingly stringent environmental regulations. This alignment with sustainability goals enhances the corporate reputation of manufacturers and meets the growing demand for eco-friendly agrochemical production methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of adopting this technology for their own production lines or sourcing strategies. The answers reflect the balance between chemical precision and operational practicality required in the fine chemical industry.
Q: What is the primary advantage of the one-pot method described in CN1301696A?
A: The primary advantage is the elimination of the intermediate isolation step. By converting the sulfonylaminocarbonyl triazolinone intermediate directly to its salt in the same vessel, the process significantly reduces processing time, solvent usage, and potential product loss associated with filtration and drying steps.
Q: How does pH control impact the purity of the final herbicide salt?
A: Strict pH control during the salt formation step is critical. The patent data indicates that maintaining a pH between 6 and 7 yields products with approximately 98% purity. Deviating to higher pH levels (above 10) can lead to a decrease in purity, necessitating careful monitoring during base addition.
Q: Which solvents are preferred for this synthesis scale-up?
A: Methyl isobutyl ketone (MIBK) and xylene mixtures are identified as preferred solvents. These solvents facilitate the reaction at moderate temperatures (0°C to 45°C) and allow for effective azeotropic drying of starting materials, which is essential for high yield conversion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylaminocarbonyl Triazolinone Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global agrochemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the one-pot sulfonylaminocarbonyl triazolinone synthesis are executed with precision. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our facility is designed to handle the specific solvent systems and reaction conditions required for this process, guaranteeing consistent quality and supply continuity for our partners.
We invite you to collaborate with us to optimize your supply chain for herbicide intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your product development and commercialization goals. Let us be your trusted partner in navigating the complexities of agrochemical intermediate production.
