Industrial Scale Production of Tetraalkoxypropane: A Safer Route for Pharma and Agrochemical Intermediates
The chemical industry constantly seeks methods to enhance safety and efficiency, particularly when dealing with volatile reagents. Patent CN1764623A introduces a transformative approach to the production of tetraalkoxypropanes, specifically targeting the synthesis of 1,1,3,3-tetraalkoxypropane and its derivatives. Historically, the synthesis of these valuable intermediates relied heavily on methyl vinyl ether or ethyl vinyl ether. However, methyl vinyl ether exists as a gas under standard conditions, posing significant logistical challenges for industrial containment and reaction control. Similarly, ethyl vinyl ether has a very low boiling point of 36-37°C, rendering it highly flammable and difficult to manage in large-scale operations. This patent elegantly solves these issues by utilizing propoxy vinyl ether, a liquid reagent with a significantly higher boiling point, thereby enabling robust industrial scalability without the need for specialized high-pressure gas handling infrastructure.
This technological advancement is particularly relevant for manufacturers of high-purity tetraalkoxypropane intended for use as skeleton forming agents in the production of pyrazole and pyrimidine derivatives. These derivatives are foundational building blocks in the pharmaceutical and agrochemical sectors. By shifting the feedstock from gaseous or highly volatile liquids to a stable liquid vinyl ether, the process described in CN1764623A not only mitigates safety risks but also streamlines the supply chain. For a reliable pharmaceutical intermediate supplier, adopting such a route means reducing the complexity of raw material procurement and storage, ultimately leading to more consistent production schedules and enhanced operational continuity for downstream clients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
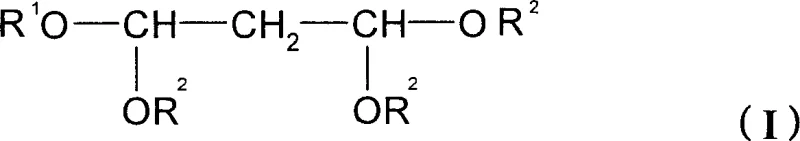
Traditional synthetic routes for 1,1,3,3-tetraalkoxypropanes, such as 1,1,3,3-tetramethoxypropane, have long been hindered by the physical properties of the requisite vinyl ethers. The conventional method typically involves the reaction of an orthoformate ester with a corresponding vinyl ether. For the methoxy variant, this necessitates the use of methyl vinyl ether. Since methyl vinyl ether is gaseous, its application in industrial settings requires complex pressurized systems, specialized gas feeding lines, and rigorous leak detection protocols, all of which drive up capital expenditure and operational costs. Furthermore, the use of ethyl vinyl ether for the ethoxy variant presents a different set of hazards; its low boiling point makes it extremely volatile and prone to ignition, creating a substantial fire risk in large reactors. These factors collectively limit the feasibility of scaling these reactions to the multi-ton levels required by the global demand for commercial scale-up of complex heterocyclic intermediates.
The Novel Approach
The methodology disclosed in CN1764623A circumvents these inherent limitations by substituting the problematic vinyl ethers with propoxy vinyl ether or butoxy vinyl ether. These reagents are liquids at ambient temperatures and possess higher boiling points, which drastically simplifies the reaction engineering. The process allows for the synthesis of both symmetric tetraalkoxypropanes, like 1,1,3,3-tetramethoxypropane, and novel asymmetric variants, such as 1,3,3-trimethoxy-1-propoxypropane. This flexibility is achieved through an alkoxy exchange reaction mechanism where the propoxy group from the vinyl ether can be exchanged or retained depending on the stoichiometry and reaction conditions. This innovation effectively decouples the production of critical pharma intermediates from the constraints of gaseous reagents, offering a pathway that is inherently safer, easier to control, and more amenable to standard batch processing equipment found in most fine chemical facilities.
Mechanistic Insights into Lewis Acid-Catalyzed Alkoxy Exchange
The core of this synthesis lies in the Lewis acid-catalyzed reaction between a trialkyl orthoformate and a propoxy vinyl ether. The mechanism initiates with the activation of the orthoformate or the vinyl ether by a Lewis acid catalyst, such as anhydrous iron(III) chloride (FeCl3), boron trifluoride, or aluminum chloride. Among these, iron(III) chloride is often preferred due to its balance of catalytic activity, safety profile, and cost-effectiveness. The catalyst facilitates the nucleophilic attack of the alkoxy group on the vinyl double bond, leading to the formation of a mixed acetal intermediate. This intermediate subsequently undergoes rearrangement and further alkoxy exchange to yield the final tetraalkoxypropane structure. The reaction is typically conducted at mild temperatures ranging from -30°C to 50°C, which helps in minimizing side reactions and thermal degradation of the sensitive acetal products.
Impurity control in this process is managed primarily through the precise regulation of reactant ratios and post-reaction purification. By using a large excess of the orthoformate, the equilibrium can be shifted to favor the formation of symmetric products, while limiting the orthoformate allows for the isolation of asymmetric species. The patent highlights that unreacted orthoformate can be recovered and recycled, which is a crucial aspect of waste minimization. Following the reaction, the mixture is neutralized, often with a base like sodium carbonate, to quench the Lewis acid catalyst. The final product is then isolated via vacuum distillation. This purification step is critical for removing residual starting materials and any oligomeric byproducts, ensuring that the resulting high-purity tetraalkoxypropane meets the stringent specifications required for subsequent cyclization reactions into pyrazoles and pyrimidines.
How to Synthesize 1,1,3,3-Tetramethoxypropane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production. The process begins with the charging of a reactor with the orthoformate, followed by the addition of the catalyst under controlled conditions. The propoxy vinyl ether is then added slowly to manage the exotherm and ensure complete conversion. Detailed standardized synthesis steps see the guide below.
- Charge a reactor with trialkyl orthoformate and add a Lewis acid catalyst such as anhydrous iron(III) chloride under stirring.
- Maintain the reaction temperature between -30°C and 50°C while slowly adding propoxy vinyl ether over several hours.
- After aging the mixture, neutralize the catalyst and perform vacuum distillation to isolate the target tetraalkoxypropane or asymmetric derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to propoxy vinyl ether represents a significant strategic advantage. The primary benefit stems from the elimination of the need for specialized gas handling infrastructure. By utilizing liquid raw materials that are stable at ambient conditions, facilities can leverage existing tank farms and pumping systems, thereby avoiding the capital investment associated with pressurized gas storage and delivery systems. This transition directly contributes to cost reduction in agrochemical intermediate manufacturing by lowering both the initial setup costs and the ongoing maintenance expenses related to safety compliance for hazardous gases. Furthermore, the use of less volatile reagents reduces the risk of production stoppages due to safety incidents or regulatory inspections focused on high-risk volatile organic compounds.
- Cost Reduction in Manufacturing: The adoption of this process eliminates the requirement for expensive and complex equipment needed to handle gaseous methyl vinyl ether. Liquid propoxy vinyl ether can be transported and stored in standard drums or bulk tanks, significantly reducing logistics costs. Additionally, the ability to recover and recycle excess orthoformate improves the overall atom economy of the process, leading to lower raw material consumption per unit of product. The use of inexpensive and readily available Lewis acid catalysts like iron(III) chloride further drives down the operational expenditure compared to processes requiring precious metal catalysts or extreme conditions.
- Enhanced Supply Chain Reliability: Sourcing gaseous reagents often involves complex logistics and limited supplier bases, which can introduce vulnerabilities into the supply chain. In contrast, propoxy vinyl ether and orthoformates are commercially available liquid chemicals with established supply networks. This availability ensures a more stable and continuous flow of raw materials, reducing lead time for high-purity pyrazole precursors. The robustness of the liquid-phase reaction also means that production schedules are less susceptible to disruptions caused by weather conditions or transportation restrictions that often affect the shipment of compressed gases.
- Scalability and Environmental Compliance: The process is inherently scalable because it operates under mild conditions and uses standard chemical engineering unit operations. The ability to run the reaction at near-ambient temperatures reduces energy consumption for heating or cooling, contributing to a smaller carbon footprint. Moreover, the reduced volatility of the reagents minimizes fugitive emissions, aiding in compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The simplified waste stream, primarily consisting of recyclable orthoformates and neutralized salts, facilitates easier waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of tetraalkoxypropanes using the patented propoxy vinyl ether route. These insights are derived directly from the experimental data and claims presented in CN1764623A, providing clarity on the feasibility and advantages of this method for potential partners and technical stakeholders.
Q: Why is propoxy vinyl ether preferred over methyl vinyl ether for industrial synthesis?
A: Methyl vinyl ether is a gas at room temperature, making it difficult and hazardous to handle in large-scale industrial processes. Propoxy vinyl ether is a liquid with a higher boiling point, allowing for safer and easier handling using standard liquid processing equipment.
Q: What are the primary applications of 1,1,3,3-tetraalkoxypropane?
A: These compounds serve as highly reactive skeleton forming agents used primarily in the synthesis of pyrazole and pyrimidine derivatives, which are critical intermediates in the production of various pharmaceuticals and agricultural chemicals.
Q: Can this process produce asymmetric tetraalkoxypropanes?
A: Yes, by adjusting the molar ratio of propoxy vinyl ether to trialkyl orthoformate and controlling the reaction temperature, the process can selectively produce asymmetric forms like 1,3,3-trialkoxy-1-propoxypropane alongside symmetric products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,3,3-Tetramethoxypropane Supplier
The synthesis route detailed in CN1764623A exemplifies the kind of process innovation that drives efficiency in the fine chemical sector. At NINGBO INNO PHARMCHEM, we specialize in translating such patented methodologies into robust commercial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,1,3,3-tetramethoxypropane or its asymmetric derivatives meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in Lewis acid catalysis and acetal chemistry, we can help you secure a stable supply of high-quality skeleton forming agents. Please contact us to request specific COA data and route feasibility assessments for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
