Advanced Synthesis of 2-Heterocyclyl Methyl Benzoic Acid Derivatives for Pharmaceutical Manufacturing
Advanced Synthesis of 2-Heterocyclyl Methyl Benzoic Acid Derivatives for Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, selective, and scalable synthetic routes. Patent CN1358176A introduces a groundbreaking methodology for the production of 2-heterocyclyl methyl benzoic acid derivatives, a class of compounds serving as critical building blocks for various bioactive agents. This technology addresses long-standing challenges in regioselectivity and purification, offering a robust pathway for generating high-purity intermediates essential for drug discovery and development. By leveraging the unique reactivity of 2-benzo[c]furanones (phthalides), this process bypasses traditional multi-step sequences, directly delivering the target carboxylic acid functionality with exceptional control over impurity profiles.
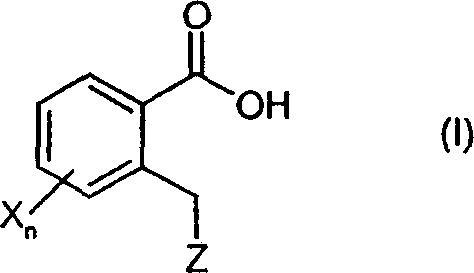
These derivatives, characterized by the general formula (I) where the heterocyclic moiety Z is linked via a methylene bridge to the ortho-position of a benzoic acid scaffold, represent a versatile chemical space. The ability to introduce diverse heterocyclic systems—ranging from triazoles to complex fused rings—allows medicinal chemists to rapidly explore structure-activity relationships (SAR). For R&D directors focused on pipeline acceleration, the availability of such a flexible synthetic platform means faster iteration cycles and reduced time-to-market for new therapeutic candidates. Furthermore, the structural integrity of sensitive functional groups is maintained throughout the synthesis, ensuring that the final intermediates meet the stringent quality standards required for downstream coupling reactions in API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of analogous structures often relied on the alkylation of heterocycles with 2-halomethyl benzoates. This conventional approach suffers from significant drawbacks, primarily the necessity of a subsequent ester hydrolysis step to reveal the free carboxylic acid. This hydrolysis is not merely an additional operational unit; it poses severe chemical compatibility issues, as many sensitive substituents on the aromatic ring or the heterocycle may degrade under the harsh acidic or basic conditions required for saponification. Moreover, competing nucleophilic attacks can lead to the formation of N-heterocyclyl-2-hydroxymethyl-benzamide derivatives, a persistent side reaction that complicates purification and drastically reduces overall yield. The accumulation of these difficult-to-remove impurities often necessitates expensive chromatographic separations, rendering the process economically unviable for large-scale commercial production.
The Novel Approach
In stark contrast, the method disclosed in CN1358176A utilizes 2-benzo[c]furanone (phthalide) derivatives as the electrophilic partner, reacting them directly with nitrogen heterocycles or their metal salts. ![General Formula II of 2-benzo[c]furanone (phthalide)](/insights/img/2-heterocyclyl-methyl-benzoic-acid-pharma-intermediate-supplier-20260306234146-02.png)
This innovative strategy capitalizes on the inherent ring strain and electrophilicity of the lactone carbonyl in the phthalide structure. Upon nucleophilic attack by the nitrogen heterocycle, the lactone ring opens spontaneously to generate the carboxylate anion, which upon workup yields the desired benzoic acid directly. This telescoped transformation effectively merges the alkylation and hydrolysis steps into a single operational sequence. Surprisingly, despite the theoretical possibility of forming amide byproducts similar to those seen in prior art, this process demonstrates remarkable chemoselectivity, favoring the formation of the C-N bond at the methylene position while preserving the carboxylic acid functionality. This selectivity is a game-changer for process chemistry, eliminating the need for protecting group strategies and significantly streamlining the manufacturing workflow.
Mechanistic Insights into Nucleophilic Ring-Opening Alkylation
The core of this technology lies in the nucleophilic ring-opening of the phthalide system. The reaction initiates with the generation of a nucleophilic nitrogen species, typically achieved by deprotonating the heterocycle (H-Z) with a suitable base such as potassium hydroxide, sodium hydride, or alkali metal carbonates. 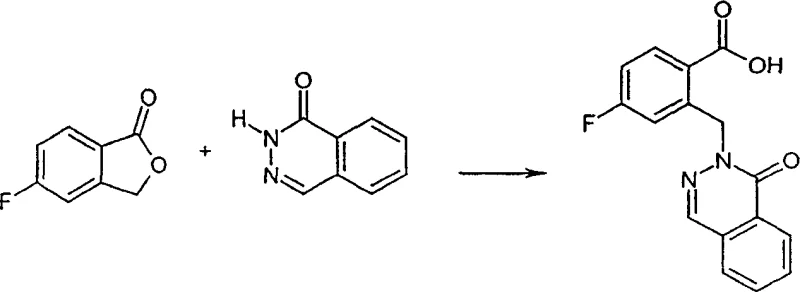
Once formed, this nitrogen anion attacks the methylene carbon adjacent to the lactone oxygen in the phthalide molecule. This attack triggers the cleavage of the C-O bond within the lactone ring, resulting in the formation of a stable carboxylate intermediate. The choice of solvent plays a pivotal role in this mechanism; aprotic polar diluents like N,N-dimethylformamide (DMF) or methyl isobutyl ketone (MIBK) are essential. These solvents effectively solvate the cationic counter-ions (e.g., K+, Na+) without hydrogen bonding to the nucleophile, thereby enhancing its reactivity and ensuring a clean substitution profile. The reaction temperature, typically ranging from 0°C to 200°C depending on the specific substrates, allows for fine-tuning of the kinetics to maximize conversion while minimizing thermal degradation of sensitive heterocyclic motifs.
From an impurity control perspective, the mechanism inherently suppresses the formation of the hydroxymethyl-benzamide byproduct. In conventional aminolysis of esters, the amine attacks the carbonyl carbon; however, in this phthalide system, the steric and electronic environment favors attack at the benzylic methylene position. This regioselectivity is crucial for maintaining high purity levels, as it prevents the generation of isomeric impurities that share similar physical properties with the target molecule. For quality control teams, this means simpler crystallization protocols and higher confidence in the identity of the bulk material, reducing the analytical burden and ensuring consistent batch-to-batch reproducibility essential for regulatory filings.
How to Synthesize 2-Heterocyclyl Methyl Benzoic Acid Efficiently
Implementing this synthesis requires careful attention to the preparation of the heterocycle salt and the selection of the reaction medium. The process generally begins with the suspension of the nitrogen heterocycle in a solvent like methyl isobutyl ketone, followed by the portion-wise addition of a base such as 85% potassium hydroxide. After stirring to ensure complete salt formation, the phthalide derivative is introduced, often in a co-solvent system like DMF to facilitate dissolution and heat transfer. The mixture is then heated to reflux, driving the ring-opening reaction to completion. Following the reaction, standard workup procedures involving concentration, aqueous washes, and acidification allow for the isolation of the pure product.
- Prepare the nitrogen heterocycle metal salt by treating the heterocycle with a base like potassium hydroxide or sodium hydride in a solvent such as methyl isobutyl ketone.
- React the prepared metal salt with 2-benzo[c]furanone (phthalide) in an aprotic polar diluent like N,N-dimethylformamide under reflux conditions.
- Isolate the final 2-heterocyclyl methyl benzoic acid derivative by concentrating the mixture, washing with ether, and precipitating with acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phthalide-based route offers substantial strategic benefits beyond mere technical elegance. The elimination of the ester hydrolysis step translates directly into reduced processing time and lower utility consumption, as there is no need for extended heating under strongly acidic or basic conditions followed by neutralization. This streamlining of the process flow significantly lowers the operational expenditure (OPEX) associated with manufacturing these intermediates. Furthermore, the avoidance of harsh hydrolysis conditions means that a broader range of starting materials can be utilized without fear of decomposition, expanding the sourcing options for raw materials and mitigating supply risk. This flexibility is critical in a volatile global market where the availability of specific halogenated precursors can fluctuate.
- Cost Reduction in Manufacturing: The most significant economic driver is the reduction in unit operations. By combining alkylation and hydrolysis into a single pot, the process eliminates the need for intermediate isolation, drying, and re-dissolution steps. This consolidation reduces solvent usage, waste generation, and labor hours. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, which often require costly chromatographic purification or multiple recrystallizations. The ability to isolate the product via simple filtration after acidification further drives down downstream processing costs, making the overall cost of goods sold (COGS) highly competitive compared to traditional halomethyl benzoate routes.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically 2-benzo[c]furanones and common nitrogen heterocycles, are commodity chemicals available from multiple global suppliers. Unlike specialized halomethyl esters which may have limited supply chains or long lead times, phthalides are robust and stable, allowing for long-term storage and bulk purchasing strategies. This stability ensures a continuous supply of intermediates, preventing production bottlenecks. Moreover, the reaction conditions are compatible with standard stainless steel reactors, meaning that existing manufacturing infrastructure can be utilized without the need for specialized glass-lined or Hastelloy equipment, further securing supply continuity.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The use of aprotic polar solvents like MIBK and DMF is well-established in industrial settings, with established recovery and recycling protocols. The absence of strong mineral acids or bases in the final workup (only mild acidification is needed) reduces the corrosive load on equipment and simplifies wastewater treatment. The high atom economy of the ring-opening reaction means less chemical waste is generated per kilogram of product. This aligns with modern green chemistry principles and helps pharmaceutical companies meet their sustainability goals, reducing the environmental footprint of their supply chain while ensuring compliance with increasingly strict regulatory standards.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is vital for stakeholders evaluating its integration into their supply chain. The following questions address common technical and commercial inquiries regarding the synthesis of 2-heterocyclyl methyl benzoic acid derivatives. These answers are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making.
Q: What is the primary advantage of using phthalides over halomethyl benzoates?
A: Using phthalides eliminates the need for a separate ester hydrolysis step, which can be unstable for certain substituents, thereby simplifying the workflow and improving overall yield.
Q: Which solvents are preferred for this nucleophilic ring-opening reaction?
A: Aprotic polar diluents are critical, with N,N-dimethylformamide (DMF), N,N-dimethylacetamide, and methyl isobutyl ketone (MIBK) being particularly effective for stabilizing the reaction intermediates.
Q: Does this method prevent the formation of amide byproducts?
A: Yes, surprisingly, this method selectively yields the desired benzoic acid derivatives with high purity, avoiding the N-heterocyclyl-2-hydroxymethyl-benzamide side reactions common in prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Heterocyclyl Methyl Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We are committed to delivering these complex 2-heterocyclyl methyl benzoic acid derivatives with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this patented technology, guaranteeing a reliable supply of material for your clinical and commercial needs.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and budgetary goals. Let us be your partner in turning innovative chemistry into commercial reality.
