Advanced Manufacturing of Olopatadine Deuterium-Labeled Metabolites for Clinical Pharmacokinetics
Advanced Manufacturing of Olopatadine Deuterium-Labeled Metabolites for Clinical Pharmacokinetics
The pharmaceutical industry's relentless pursuit of precision medicine has elevated the importance of stable isotope-labeled standards, particularly for complex antihistamines like olopatadine. Patent CN114380783A, published in April 2022, discloses a highly efficient five-step preparation method for an olopatadine deuterium-labeled metabolite, specifically targeting the N-methyl position with deuterium atoms. This technical breakthrough addresses the critical need for reliable internal standards in LC-MS/MS bioanalysis, enabling precise quantification of drug levels in biological matrices. As a leading entity in fine chemical manufacturing, we recognize that the ability to synthesize such specialized intermediates with high isotopic enrichment and chemical purity is a cornerstone of modern drug development support. The disclosed route transforms a hydroxypropyl precursor into the final deuterated acid through a logical sequence of activation, amination, and functional group manipulation, offering a blueprint for scalable production.
For R&D directors overseeing pharmacokinetic (PK) studies, the availability of high-quality deuterated metabolites is non-negotiable. These compounds serve as the gold standard for correcting matrix effects and ionization variability in mass spectrometry. The methodology outlined in the patent ensures that the deuterium label remains stable throughout the synthetic sequence, minimizing isotopic dilution which could otherwise compromise data integrity. By adopting this streamlined approach, manufacturers can significantly reduce the lead time for high-purity pharmaceutical intermediates required for IND-enabling toxicology studies. Furthermore, the process avoids harsh conditions that might degrade the sensitive dibenzoxepin core, thereby preserving the structural integrity essential for accurate metabolic tracking.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterium-labeled olopatadine metabolites has been plagued by inefficiencies associated with late-stage isotopic incorporation. Conventional routes often rely on reductive amination using deuterated reducing agents, which can suffer from incomplete deuteration or scrambling of the label due to proton exchange with the solvent. Additionally, traditional methods frequently employ expensive transition metal catalysts that require rigorous removal steps to meet residual metal specifications for biological testing. These legacy processes often result in lower overall yields and generate significant quantities of hazardous waste, complicating the environmental compliance profile of the manufacturing campaign. For procurement managers, these inefficiencies translate into higher costs per gram and extended supply chain vulnerabilities, as specialized reagents may have long lead times.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by introducing the deuterium label early in the synthesis via a nucleophilic substitution reaction. By converting the hydroxyl group of the starting material into a good leaving group (bromide), the process enables a clean SN2 displacement using a commercially available deuterated amine. This strategy not only ensures high isotopic purity but also simplifies the purification workflow, as the byproducts are easily removed via aqueous workup or standard chromatography. The subsequent steps involve a clever protection-deprotection sequence using chloroformates, which masks the secondary amine functionality to prevent side reactions during the final ester hydrolysis. This logical disconnection of the molecule allows for cost reduction in pharmaceutical intermediates manufacturing by maximizing atom economy and minimizing the number of purification cycles required.
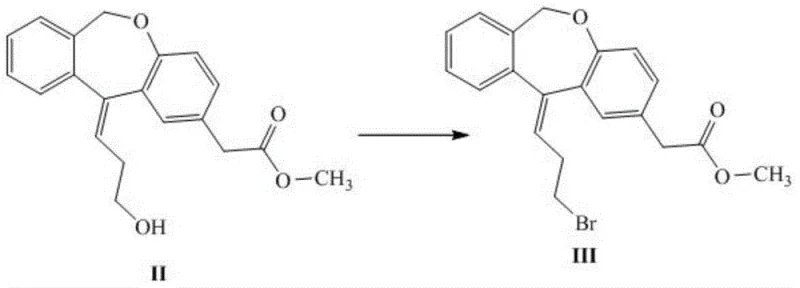
Mechanistic Insights into Nucleophilic Substitution and Protecting Group Strategy
The core of this synthetic innovation lies in the activation of the primary alcohol and its subsequent displacement. In the first step, the hydroxypropyl side chain of the dibenzoxepin scaffold is treated with a brominating reagent such as hydrobromic acid or phosphorus tribromide. This transformation converts the poor leaving group (-OH) into a highly reactive alkyl bromide, priming the molecule for nucleophilic attack. The reaction conditions are remarkably mild, typically proceeding at temperatures between 50°C and 100°C, which prevents thermal degradation of the tricyclic system. Following isolation, the alkyl bromide undergoes substitution with methyl-d3-benzylamine in the presence of a base like potassium carbonate or sodium hydroxide. This step is critical as it installs the isotopic label with high fidelity, leveraging the strong nucleophilicity of the amine to drive the reaction to completion.
Following the amination, the synthesis employs a temporary protecting group strategy to manage the reactivity of the secondary amine. The intermediate is reacted with a chloroformate, such as 1-chloroethyl chloroformate, to form a carbamate derivative. This modification reduces the basicity of the nitrogen, preventing it from interfering with the subsequent hydrolysis of the methyl ester. The deprotection step is elegantly simple, involving reflux in a protic solvent like ethanol or isopropanol, which cleaves the carbamate to regenerate the free amine. Finally, the methyl ester is hydrolyzed under basic conditions to yield the target carboxylic acid. This multi-step choreography demonstrates a deep understanding of orthogonal reactivity, ensuring that each functional group is modified selectively without affecting the others, a key requirement for producing high-purity OLED material or pharmaceutical standards.
How to Synthesize Olopatadine Deuterium Labeled Metabolite Efficiently
The synthesis of this complex metabolite requires precise control over reaction parameters to ensure optimal yield and isotopic retention. The process begins with the activation of the alcohol, followed by the introduction of the deuterated amine, and concludes with ester hydrolysis. Each step has been optimized in the patent examples to demonstrate robustness across different scales.
- Bromination of the hydroxypropyl side chain using hydrobromic acid or phosphorus tribromide to activate the alcohol for nucleophilic attack.
- Nucleophilic substitution with methyl-d3-benzylamine in the presence of base (K2CO3 or NaOH) to introduce the deuterium label.
- Protection of the secondary amine using chloroformates followed by deprotection in protic solvents to prepare for final hydrolysis.
- Final saponification of the methyl ester using inorganic base under mild conditions to yield the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented methodology offers tangible benefits in terms of cost stability and operational reliability. The reliance on commodity chemicals such as hydrobromic acid, potassium carbonate, and common organic solvents (THF, toluene, ethanol) means that raw material sourcing is not dependent on single-source suppliers or geopolitical bottlenecks. This diversification of the supply base enhances supply chain reliability, ensuring that production campaigns can proceed without interruption even if one reagent becomes temporarily scarce. Furthermore, the elimination of precious metal catalysts removes the need for expensive scavenging resins and extensive analytical testing for heavy metals, directly contributing to cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The process achieves high yields in the critical bond-forming steps, with the amination reaction reporting yields exceeding 90% in optimized examples. High conversion rates minimize the loss of expensive deuterated starting materials, which are typically the cost driver in isotope-labeled synthesis. Additionally, the use of standard purification techniques like column chromatography and recrystallization avoids the need for preparative HPLC, which is costly and difficult to scale. By streamlining the workflow and reducing the number of unit operations, the overall cost of goods sold (COGS) is significantly lowered, making the final metabolite more accessible for routine QC applications.
- Enhanced Supply Chain Reliability: The synthetic route is designed for scalability, utilizing reactors and equipment standard in any multipurpose chemical plant. The absence of cryogenic conditions or high-pressure hydrogenation simplifies the engineering requirements, allowing for rapid technology transfer from lab to pilot to commercial scale. This flexibility ensures that suppliers can respond quickly to fluctuating demand from clinical trial sites. Moreover, the robustness of the chemistry means that batch-to-batch variability is minimized, providing customers with consistent quality and reducing the risk of failed batches that could delay critical drug development timelines.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates waste streams that are manageable and treatable using standard effluent treatment protocols. The solvents used are largely recyclable, and the avoidance of toxic heavy metals aligns with green chemistry principles increasingly demanded by regulatory bodies. This environmental compatibility facilitates smoother regulatory approvals for the manufacturing site and reduces the liability associated with hazardous waste disposal. The ability to scale this process from grams to kilograms without fundamental changes to the chemistry underscores its suitability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for their specific projects. The following questions address common concerns regarding purity, scalability, and application.
Q: What is the isotopic purity and labeling position of this metabolite?
A: The synthesis utilizes methyl-d3-benzylamine, ensuring three deuterium atoms are specifically located on the N-methyl group. This precise labeling allows for accurate mass spectrometric differentiation from the endogenous drug during metabolic profiling.
Q: Can this process be scaled for commercial GMP production?
A: Yes, the process relies on standard unit operations such as reflux, filtration, and solvent extraction without requiring cryogenic conditions or exotic catalysts. The high yields reported in the patent examples (up to 99% in intermediate steps) indicate excellent scalability for multi-kilogram campaigns.
Q: How does this method improve upon traditional metabolite synthesis?
A: Traditional methods often struggle with late-stage isotopic incorporation which can be low-yielding. This route introduces the label early via a robust SN2 reaction on an activated alkyl bromide, ensuring high isotopic retention and overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olopatadine Deuterium Labeled Metabolite Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with state-of-the-art manufacturing capabilities to deliver high-value intermediates like the olopatadine deuterium-labeled metabolite. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that in the realm of isotopic standards, stringent purity specifications are paramount; therefore, our rigorous QC labs utilize advanced analytical techniques to verify isotopic enrichment and chemical purity before every shipment. Whether you require milligram quantities for method development or kilogram scales for clinical trials, our infrastructure is ready to support your timeline.
We invite you to collaborate with us to optimize your supply chain for deuterated standards. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient synthetic routes can lower your overall project costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in accelerating drug development through superior chemical manufacturing solutions.
