Advanced Synthesis of Stable Cis-Cyclohexyl Amino Nitrile Salts for Scalable Agrochemical Production
Advanced Synthesis of Stable Cis-Cyclohexyl Amino Nitrile Salts for Scalable Agrochemical Production
The agrochemical industry continuously demands higher purity intermediates to ensure the efficacy and safety of final pesticide products. Patent CN110691771A introduces a significant technological breakthrough in the preparation of cis-para-substituted cyclohexylaminonitrile salts, which serve as critical precursors for the synthesis of spirotetramat, a potent insecticide and acaricide. This innovation addresses long-standing challenges regarding isomer separation and salt stability that have historically plagued the manufacturing of this key agrochemical intermediate. By utilizing specific organic acids such as maleic acid or glycolic acid, the disclosed method achieves exceptional stereo-selectivity, isolating the biologically active cis-isomer with high precision. This development is particularly relevant for R&D directors and procurement specialists seeking reliable agrochemical intermediate suppliers who can guarantee consistent quality and supply continuity. The transition from unstable formate salts or non-selective inorganic acid salts to these novel stable crystalline forms represents a paradigm shift in process chemistry, offering a robust foundation for commercial scale-up of complex agrochemical intermediates.
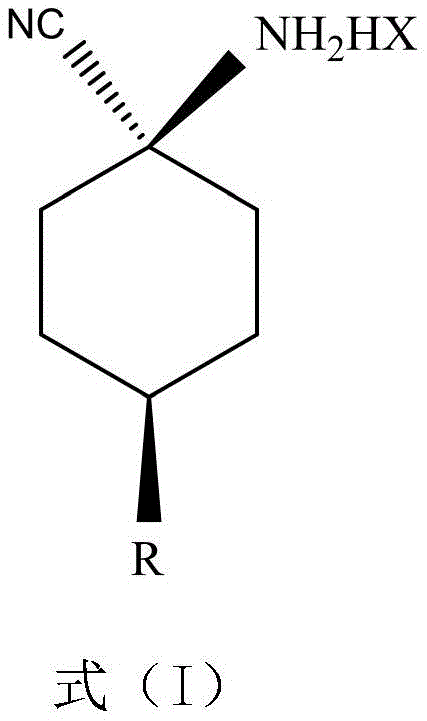
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in Chinese patent CN103270020B, relied heavily on inorganic acids like hydrochloric, sulfuric, and phosphoric acid, or organic acids like formic and acetic acid, to form salts with cyclohexylaminonitrile compounds. However, these conventional approaches suffered from severe deficiencies that hindered industrial application. Inorganic acids lacked the necessary stereo-selectivity, failing to distinguish between cis and trans isomers during crystallization, which resulted in mixtures that were difficult to purify further. Acetic acid salts simply failed to crystallize under standard conditions, rendering the isolation process impossible. Furthermore, while formic acid could form salts, the resulting products were notoriously unstable; they tended to agglomerate, change color from white to gray or brown, and decompose significantly over time, with purity dropping drastically within months. These stability issues posed significant risks for supply chain heads, as stored inventory could degrade before use, leading to substantial material waste and production delays.
The Novel Approach
The novel approach detailed in CN110691771A overcomes these historical barriers by employing maleic acid or glycolic acid as the salt-forming agents. This strategic selection of dicarboxylic and hydroxy acids facilitates the formation of a stable crystal lattice that preferentially incorporates the cis-isomer. The process involves dissolving the cis/trans mixture in solvents like ethyl acetate or methyl tert-butyl ether and adding the specific acid at controlled temperatures. Upon cooling to 1-10°C, the target cis-para-substituted cyclohexylaminonitrile salt crystallizes out with remarkable purity, often achieving cis/trans ratios greater than 95:5. Unlike the formate salts, these maleate and glycolate salts remain white and stable during storage, maintaining their chemical integrity and potency. This improvement not only simplifies the purification workflow but also ensures that the intermediate delivered to the next synthesis stage is of consistently high quality, thereby reducing lead time for high-purity agrochemical intermediates and minimizing the need for reprocessing.
Mechanistic Insights into Stereo-selective Crystallization and Acylation
The core of this technological advancement lies in the specific molecular interactions between the cyclohexylaminonitrile substrate and the chosen acid counter-ions. Maleic acid and glycolic acid possess unique structural features that enable them to form strong, directional hydrogen bonds with the amino group of the cis-isomer. This interaction stabilizes the cis-conformation within the crystal lattice more effectively than the trans-isomer, driving the equilibrium towards the precipitation of the desired stereoisomer. The thermodynamic stability of these salts is superior, preventing the degradation pathways observed with formic acid salts, such as hydrolysis or oxidative discoloration. For R&D teams, understanding this mechanism is crucial for optimizing solvent systems and cooling profiles to maximize yield. The ability to selectively crystallize the cis-isomer from a roughly 55:45 mixture without requiring complex chromatographic separation significantly streamlines the manufacturing process, making it highly attractive for cost reduction in agrochemical intermediate manufacturing.
In the subsequent conversion of this salt to Compound II, the patent highlights another critical mechanistic improvement regarding the choice of acid binding agent. Traditional methods often utilized tertiary amines like triethylamine to scavenge the hydrochloric acid generated during the acylation with 2,5-dimethylphenylacetyl chloride. However, strong organic bases can promote the unwanted hydrolysis of the sensitive nitrile group, leading to impurity formation and reduced yields. The disclosed method replaces these organic amines with inorganic bases such as sodium carbonate or sodium bicarbonate. These weaker bases are sufficiently alkaline to neutralize the acid byproduct but mild enough to preserve the nitrile functionality. This subtle yet powerful modification prevents side reactions, ensuring that the final amide product retains its structural integrity. Additionally, the use of inorganic bases simplifies the workup procedure, as the resulting salts are water-soluble and easily removed, avoiding the complex distillation steps required to recover volatile organic amines.
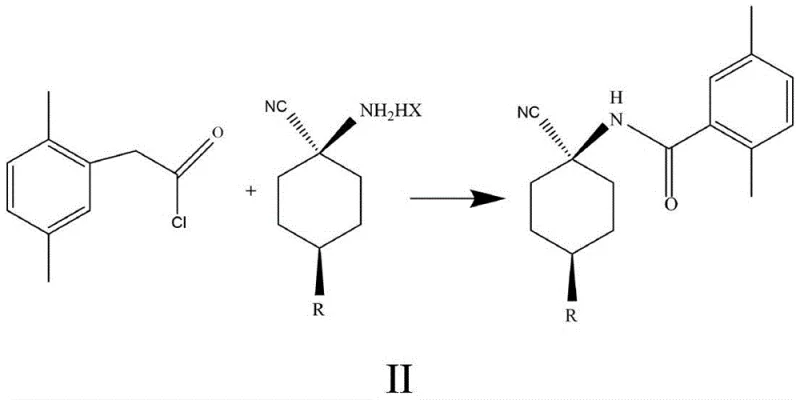
How to Synthesize Cis-para-substituted Cyclohexylaminonitrile Salt Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for producing the stable cis-isomer salt, suitable for both laboratory optimization and industrial manufacturing. The process begins with the preparation of a solution containing the crude cis/trans mixture of the aminonitrile compound in a suitable organic solvent. The choice of solvent, such as ethyl acetate or isopropyl acetate, is critical for solubility and crystal habit. Once the solution is prepared, the specific acidifying agent, either maleic acid or glycolic acid, is added in a controlled stoichiometric ratio, typically between 0.3 and 0.55 molar equivalents relative to the amine. This sub-stoichiometric addition is key to driving the selective crystallization of the cis-isomer while leaving the trans-isomer in the mother liquor. The mixture is then subjected to a controlled cooling regimen, typically bringing the temperature down to between 1 and 10 degrees Celsius. Detailed standardized synthesis steps see the guide below.
- Dissolve the cis/trans mixture of para-substituted cyclohexylaminonitrile in an organic solvent such as ethyl acetate or methyl tert-butyl ether.
- Add maleic acid or glycolic acid (0.3-0.55 molar equivalents) to the solution at 25-30°C and stir for 8-12 hours to initiate salt formation.
- Cool the reaction mixture to 1-10°C using an ice-water bath to induce crystallization, then filter and dry the stable cis-isomer salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the enhanced stability of the intermediate salt. By eliminating the instability issues associated with formate salts, manufacturers can significantly reduce material loss due to degradation during storage and transportation. This stability ensures that inventory remains usable for longer periods, providing greater flexibility in production scheduling and reducing the pressure on just-in-time delivery models. Furthermore, the high selectivity of the crystallization process means that less raw material is wasted in trying to separate isomers, leading to a more efficient utilization of starting materials. This efficiency translates directly into better resource management and a more predictable supply of high-quality intermediates for downstream pesticide production.
- Cost Reduction in Manufacturing: The switch from organic amine acid binders to inorganic carbonates or bicarbonates in the acylation step represents a significant opportunity for cost optimization. Organic amines like triethylamine are not only expensive but also require energy-intensive recovery processes, such as distillation, to be reused or disposed of safely. In contrast, inorganic bases are inexpensive, readily available commodity chemicals that do not require complex recovery systems. Additionally, the avoidance of nitrile hydrolysis means higher reaction yields and less waste generation, further driving down the cost per kilogram of the final product. The elimination of expensive heavy metal catalysts or complex purification steps in favor of simple crystallization also contributes to substantial cost savings in the overall manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the maleate and glycolate salts ensures a more reliable supply chain. Since these salts do not degrade or clump during storage, the risk of batch rejection due to quality failure is minimized. This reliability is crucial for maintaining continuous production lines for end-products like spirotetramat. Moreover, the raw materials required for this process, including the specific organic acids and inorganic bases, are widely available in the global chemical market, reducing the risk of supply bottlenecks. The ability to recycle mother liquor, as suggested in the patent, further enhances material efficiency, ensuring that the supply of the intermediate can be scaled up to meet demand without proportional increases in raw material consumption.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is highly favorable. The use of inorganic bases generates aqueous waste streams that are easier to treat compared to the organic waste generated by amine-based processes. This simplifies wastewater treatment and helps facilities meet stringent environmental regulations. The crystallization process itself is inherently scalable, as it relies on standard unit operations like mixing, cooling, and filtration, which are easily transferred from pilot plants to large-scale commercial reactors. The high purity of the isolated salt reduces the burden on downstream purification steps, allowing for a more streamlined and environmentally friendly production workflow that aligns with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications provided in patent CN110691771A, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of adopting this technology for your specific manufacturing needs. The insights provided here cover aspects of stability, selectivity, and process safety, which are critical for decision-making in R&D and operations.
Q: Why are maleate or glycolate salts preferred over formate salts for this intermediate?
A: According to patent CN110691771A, formate salts exhibit poor stability, prone to agglomeration, discoloration, and decomposition during storage. In contrast, maleate and glycolate salts form stable, white crystalline solids that maintain high purity over extended periods, ensuring consistent quality for downstream synthesis.
Q: How does this process improve the selectivity of the cis-isomer?
A: Traditional acids like hydrochloric or sulfuric acid lack the stereo-selectivity required to separate the cis-isomer effectively. The specific hydrogen bonding interactions formed with maleic or glycolic acid allow for the selective crystallization of the cis-para-substituted cyclohexylaminonitrile, achieving cis/trans ratios exceeding 95:5.
Q: What are the advantages of using inorganic bases in the subsequent acylation step?
A: Replacing organic amines like triethylamine with alkali metal carbonates or bicarbonates eliminates the risk of nitrile group hydrolysis caused by strong organic bases. This substitution also reduces costs associated with amine recovery and wastewater treatment, enhancing overall process safety and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-para-substituted Cyclohexylaminonitrile Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful production of advanced agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110691771A are fully realized in practical, large-scale operations. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex stereo-selective crystallizations and moisture-sensitive reactions allows us to provide a consistent supply of stable cis-amine salts, mitigating the risks associated with intermediate degradation and supply interruptions.
We invite you to collaborate with us to optimize your supply chain for spirotetramat and related pesticide intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to not just a product, but a comprehensive solution that includes specific COA data and route feasibility assessments to ensure seamless integration into your existing manufacturing processes. Contact us today to discuss how our advanced synthesis capabilities can enhance your production efficiency and product quality.
