Advanced Synthesis of Herbicidal Sulfonylurea Intermediates via Novel Diazotization Route
Advanced Synthesis of Herbicidal Sulfonylurea Intermediates via Novel Diazotization Route
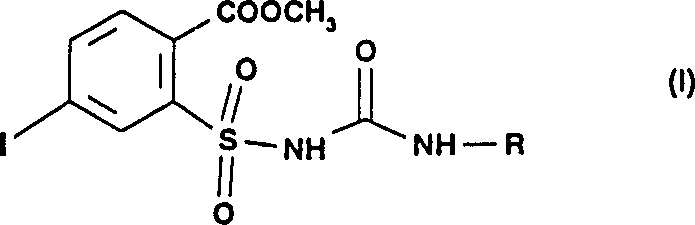
The global agrochemical industry constantly seeks more efficient pathways to produce active ingredients, particularly herbicidal sulfonylureas which are critical for modern crop protection. A pivotal advancement in this domain is documented in patent CN1078589C, which discloses a robust method for preparing 4-iodo-2-[N-(N-alkylaminocarbonyl)-aminosulfonyl]benzoic acid methyl ester and its derivatives. These compounds serve as essential building blocks for a wide range of potent herbicides. The technical breakthrough lies in a novel three-step sequence that bypasses the low-yielding limitations of traditional esterification and amidation routes. By leveraging a specific diazotization strategy followed by iodination, manufacturers can achieve unprecedented purity and volumetric efficiency. This report analyzes the technical merits of this process and its implications for supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonylurea intermediates has been plagued by inefficiencies that drive up costs and complicate waste management. Traditional approaches often involve reacting aryl-sulfonyl chlorides with ammonia to form sulfonamides, a step that frequently suffers from yields around 75% due to the formation of unwanted polysaccharide derivatives and ring-closure byproducts. Furthermore, alternative routes attempting to form the urea linkage directly from isopropyl esters and isocyanates have demonstrated even poorer performance, with documented yields as low as 44% of theoretical value. These low conversion rates necessitate extensive downstream purification, increased solvent consumption, and result in significant loss of valuable raw materials. For procurement managers, these inefficiencies translate directly into higher unit costs and unpredictable batch-to-batch consistency, creating bottlenecks in the supply of high-purity agrochemical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN1078589C introduces a paradigm shift by utilizing an amino-sulfonamide precursor as the starting point. Instead of struggling with difficult chlorosulfonyl substitutions, the process employs a diazotization reaction to install the critical iodine atom with exceptional precision. This route allows for the isolation of the key intermediate, methyl 4-iodo-2-aminosulfonylbenzoate, in yields exceeding 94% of theory. The subsequent conversion to the final urea derivative proceeds with near-quantitative efficiency, reaching up to 99.3% yield under optimized conditions. This dramatic improvement in mass balance not only reduces the cost of goods sold but also simplifies the environmental footprint by minimizing waste streams. The ability to access the target molecule through such a high-yielding pathway represents a significant competitive advantage for manufacturers aiming to reduce lead time for high-purity agrochemical intermediates.
Mechanistic Insights into Diazotization and Iodination Strategy
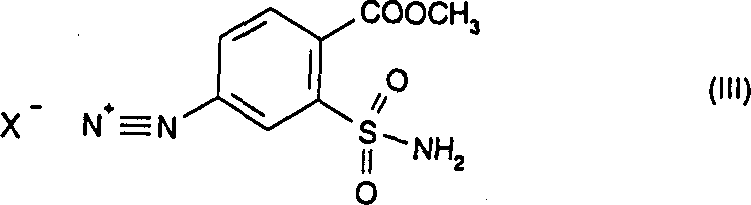
The core innovation of this synthesis lies in the careful manipulation of the diazonium species derived from methyl 4-amino-2-aminosulfonylbenzoate. In the first stage, the primary amine is treated with sodium nitrite in the presence of a strong mineral acid, such as hydrochloric acid, at a controlled temperature range of 15 to 20°C. This generates the corresponding diazonium salt intermediate in situ. Unlike simple aniline derivatives which may decompose rapidly, the presence of the ortho-sulfonamide group appears to stabilize the reaction environment, allowing for the subsequent substitution without significant degradation. The mechanistic pathway ensures that the reactive diazo group is immediately available for nucleophilic attack by iodide ions, preventing side reactions that typically plague aromatic substitution chemistry.
Following the formation of the diazonium salt, the introduction of iodide ions, typically via potassium iodide, triggers a Sandmeyer-type replacement where the nitrogen gas is expelled and the iodine atom is installed at the para-position relative to the ester group. This step is crucial because direct iodination of the aromatic ring via electrophilic substitution would lack regioselectivity and require harsher conditions. The patent data indicates that performing this reaction in an aqueous medium at 15 to 20°C results in the precipitation of the iodo-sulfonamide product, which can be easily filtered and washed. This phase separation behavior is mechanically advantageous for scale-up, as it avoids complex extraction procedures. The final step involves the nucleophilic attack of the sulfonamide nitrogen on an alkyl isocyanate, catalyzed by a mild base like potassium carbonate, to form the stable urea linkage found in the final herbicidal structure.
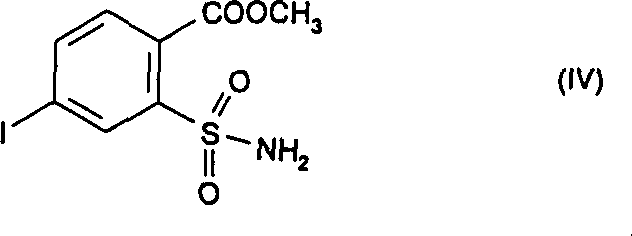
How to Synthesize 4-iodo-2-[N-(N-alkylaminocarbonyl)-aminosulfonyl]benzoic acid methyl ester Efficiently
Implementing this synthesis requires strict adherence to temperature controls and stoichiometric ratios to maximize the benefits of the novel pathway. The process begins with the preparation of the diazonium salt in an aqueous acidic medium, followed by the immediate addition of iodide to precipitate the solid intermediate. The final coupling with isocyanates is performed in organic solvents like chlorobenzene to ensure complete conversion. Detailed operational parameters, including specific molar ratios of sodium nitrite to amine and precise heating profiles for the urea formation step, are critical for reproducing the high yields reported in the patent literature. Operators must ensure that excess nitrite is destroyed before iodination to prevent oxidation of the iodide source.
- Diazotize methyl 4-amino-2-aminosulfonylbenzoate in aqueous acid (HCl) with sodium nitrite at 15-20°C to form the diazonium salt.
- React the diazonium salt solution with potassium iodide at 15-20°C to precipitate methyl 4-iodo-2-aminosulfonylbenzoate with >90% yield.
- Condense the iodo-sulfonamide intermediate with an alkyl isocyanate in chlorobenzene using potassium carbonate base at 55-90°C to obtain the final urea derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the drastic reduction in raw material waste, which directly correlates to lower manufacturing costs. By achieving yields near the theoretical maximum, facilities can produce more product per batch without increasing reactor volume or utility consumption. This efficiency creates a buffer against fluctuations in the pricing of key starting materials like amines and isocyanates. Furthermore, the use of water as the primary solvent for the initial steps reduces reliance on volatile organic compounds early in the process, aligning with increasingly stringent environmental regulations and reducing disposal costs associated with hazardous waste.
- Cost Reduction in Manufacturing: The elimination of low-yielding steps fundamentally alters the cost structure of the intermediate. Traditional methods losing over 50% of material to byproducts require significantly larger input quantities to achieve the same output. By contrast, this high-yield route minimizes the cost per kilogram of the final active ingredient. Additionally, the simplicity of the workup—often involving mere filtration and washing rather than column chromatography or distillation—reduces labor hours and energy consumption. The removal of expensive transition metal catalysts, which are sometimes required in alternative cross-coupling strategies, further lowers the bill of materials and eliminates the need for costly heavy metal scavenging processes.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as methyl 4-amino-2-aminosulfonylbenzoate and common alkyl isocyanates, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard industrial-grade reagents without requiring ultra-high purity inputs, ensures consistent production schedules. This reliability is crucial for maintaining continuous supply to downstream formulators who depend on just-in-time delivery of herbicide actives. The ability to store the stable iodo-intermediate also provides flexibility in production planning, allowing manufacturers to decouple the iodination step from the final urea formation based on demand signals.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in fine chemical plants, such as stirred tank reactors and filter presses. The aqueous nature of the diazotization and iodination steps facilitates heat transfer and safety management, mitigating the risks associated with exothermic reactions on a large tonnage scale. Moreover, the high selectivity of the reaction means fewer impurities are generated, simplifying the effluent treatment process. This aligns with green chemistry principles by maximizing atom economy and reducing the E-factor (mass of waste per mass of product), making it an attractive option for facilities aiming to improve their sustainability metrics and regulatory compliance posture.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific challenges identified in prior art and the solutions provided by the patented methodology. Understanding these nuances is essential for R&D teams evaluating technology transfer and for quality assurance teams establishing specification limits. The answers reflect the empirical data and process descriptions found within the intellectual property documentation.
Q: How does this new process improve yield compared to conventional sulfonylurea synthesis?
A: Conventional methods reacting chlorosulfonyl compounds with ammonia often yield only about 75% due to polysaccharide byproducts, while direct urea formation from isopropyl esters yields as low as 44%. This patented diazotization route achieves yields exceeding 94% for the iodo-intermediate and up to 99% for the final product.
Q: What are the critical temperature controls for the diazotization step?
A: To ensure stability of the diazonium salt intermediate and prevent decomposition, the reaction must be strictly maintained between 15°C and 20°C during the addition of sodium nitrite and the subsequent iodination step.
Q: Is this process scalable for commercial production of herbicide intermediates?
A: Yes, the process utilizes standard aqueous workups, simple filtration for isolation, and common solvents like chlorobenzene, making it highly suitable for large-scale commercial manufacturing without requiring exotic catalysts or complex purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-iodo-2-[N-(N-alkylaminocarbonyl)-aminosulfonyl]benzoic acid methyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields promised in patent CN1078589C are realized on an industrial scale. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of sulfonylurea intermediate meets the exacting standards required for agrochemical registration. We understand that consistency is key to your formulation success, and our process engineering team is dedicated to optimizing every parameter from diazotization temperature to final crystallization.
We invite you to collaborate with us to leverage this advanced synthesis route for your herbicide portfolio. Our technical sales team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield methodology for your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project timeline. Let us help you secure a sustainable and cost-effective supply chain for your next-generation crop protection solutions.
