Advanced Synthesis of Substituted Benzenesulfonylureas for Commercial Herbicide Production
The chemical landscape of modern agrochemical manufacturing is constantly evolving, driven by the need for higher purity intermediates and more sustainable production methodologies. Patent CN1630647A represents a significant technological leap in the synthesis of substituted benzenesulfonylureas, a critical class of compounds widely utilized as herbicides and plant growth regulators. This intellectual property addresses long-standing challenges in the art, specifically focusing on the preparation of compounds containing carboxyl or carboxylic acid derivative groups on the benzene ring, which have historically been difficult to synthesize with high efficiency. The core innovation lies in a meticulously engineered solvent system and a novel catalytic approach that collectively enhance reaction kinetics and product integrity. By shifting away from traditional single-solvent systems, this method achieves superior control over side reactions, particularly the formation of saccharin derivatives during ammonolysis. For R&D directors and technical procurement specialists, understanding the nuances of this patent is essential, as it offers a pathway to more reliable supply chains and cost-effective manufacturing of high-value agrochemical intermediates. The technical depth of this disclosure provides a robust foundation for scaling complex synthetic routes while maintaining stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzenesulfonylureas has relied heavily on methods disclosed in earlier patents such as WO-A-92/13845, which typically utilize tetrahydrofuran (THF) as a standalone solvent for the ammonolysis of sulfonyl chlorides. While effective on a laboratory scale, these conventional approaches suffer from significant drawbacks when translated to industrial manufacturing environments. The primary issue is the unsatisfactory chemical yield, often falling below 80% of the theoretical value, largely due to the formation of persistent by-products like saccharin derivatives through intramolecular reactions. Furthermore, the subsequent phosgenation steps in traditional methods frequently require the use of hindered amine bases such as DABCO or DBU as catalysts. The presence of these bases introduces a complex purification burden, as they must be rigorously removed from the final product to meet purity specifications, adding both time and cost to the production cycle. Additionally, the reaction times for phosgenation in these legacy processes are often excessively long, particularly during the start-up phase, which negatively impacts space-time yields and overall equipment efficiency. These technical bottlenecks create substantial risks for supply chain continuity and cost management, making the adoption of improved methodologies not just a technical preference but a commercial necessity for competitive manufacturers.
The Novel Approach
The methodology outlined in CN1630647A introduces a paradigm shift by employing a specific mixture of solvents rather than a single polar aprotic solvent. This novel approach utilizes a blend of optionally halogenated aromatic hydrocarbons, such as xylene or toluene, combined with polar aprotic solvents like acetonitrile or ethyl acetate in precise weight ratios. This solvent engineering strategy effectively suppresses the formation of unwanted saccharin by-products during the ammonolysis step, thereby significantly boosting the yield of the desired sulfonamide intermediate. Moreover, the patent discloses a refined phosgenation process that replaces traditional amine base catalysts with alkyl isocyanates, such as n-butyl isocyanate. This catalytic innovation not only accelerates the reaction rate but also eliminates the need for complex base removal steps, streamlining the downstream processing. The coupling reaction with heterocyclic amines is further optimized by conducting the reaction in a high-boiling solvent mixture, ensuring complete conversion without the need for large excesses of reagents. This holistic improvement in the synthetic route translates directly into enhanced process robustness, making it an ideal candidate for large-scale commercial production where consistency and efficiency are paramount.
Mechanistic Insights into Isocyanate-Catalyzed Phosgenation
The mechanistic elegance of this patent lies in the substitution of hindered amine bases with alkyl isocyanates during the phosgenation of the sulfonamide intermediate. In conventional pathways, the conversion of sulfonamides to sulfonyl isocyanates typically requires stoichiometric or catalytic amounts of bases like pyridine or DABCO to scavenge the hydrogen chloride generated during the reaction. However, these bases often form stable salts or complexes that are difficult to separate, leading to purity issues in the final agrochemical active ingredient. The patented process circumvents this by utilizing an isocyanate of the formula R'-NCO as a catalyst, which facilitates the reaction with phosgene without generating non-volatile salt by-products. This mechanism allows for the reaction to proceed smoothly at elevated temperatures, typically between 100°C and 180°C, with the catalyst being easily removed via distillation along with excess phosgene. The result is a phenylsulfonyl isocyanate intermediate of exceptional purity, ready for the subsequent coupling step without extensive work-up. This mechanistic refinement is critical for R&D teams focused on impurity profiling, as it minimizes the risk of carry-over contaminants that could affect the biological efficacy or regulatory approval of the final herbicide product.
Furthermore, the patent details a sophisticated strategy for controlling impurities during the final coupling stage with heterocyclic amines. The reaction between the phenylsulfonyl isocyanate and the amine component is highly sensitive to solvent choice and reaction conditions. By employing a solvent mixture with a boiling point above 110°C, such as xylene combined with ethyl acetate, the process ensures that the reaction proceeds at an optimal rate without requiring excessive amounts of the amine reactant. This is particularly important when dealing with sterically hindered substrates, where conventional methods often struggle to achieve complete conversion. The use of this specific solvent system promotes a homogeneous reaction environment that facilitates the nucleophilic attack of the amine on the isocyanate group, leading to the formation of the sulfonylurea bond with high fidelity. 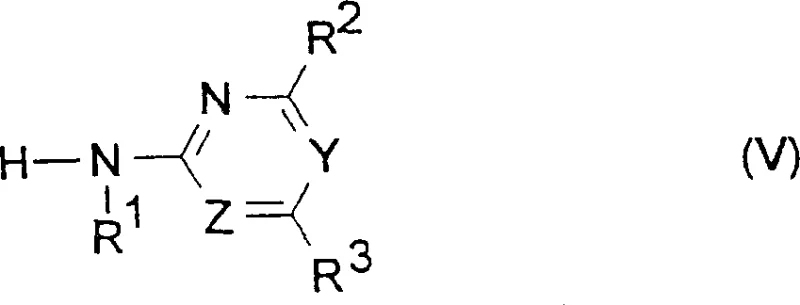 As illustrated in the structural representation, the heterocyclic amine component plays a pivotal role in defining the biological activity of the final molecule. The ability to couple this component efficiently and cleanly is a testament to the robustness of the patented solvent engineering, ensuring that the final product meets the stringent purity requirements demanded by the agrochemical industry.
As illustrated in the structural representation, the heterocyclic amine component plays a pivotal role in defining the biological activity of the final molecule. The ability to couple this component efficiently and cleanly is a testament to the robustness of the patented solvent engineering, ensuring that the final product meets the stringent purity requirements demanded by the agrochemical industry.
How to Synthesize Substituted Benzenesulfonylureas Efficiently
The implementation of this synthesis route requires careful attention to solvent ratios and reaction parameters to fully realize the benefits described in the patent. The process begins with the ammonolysis of the sulfonyl halide in a mixed solvent system, followed by the isocyanate-catalyzed phosgenation, and concludes with the coupling of the resulting isocyanate with the appropriate heterocyclic amine. Each step is designed to maximize yield and minimize waste, creating a streamlined workflow that is suitable for both pilot-scale validation and full commercial production. The detailed standardized synthesis steps provided in the technical documentation below offer a comprehensive guide for process chemists looking to adopt this methodology. These steps outline the specific temperatures, pressure conditions, and reagent addition rates necessary to achieve the reported high yields and purity levels. By adhering to these protocols, manufacturing teams can ensure consistent product quality while optimizing resource utilization.
- Perform ammonolysis of sulfonyl halides in a specific mixture of aromatic hydrocarbons and polar aprotic solvents to maximize yield and minimize saccharin by-products.
- Execute phosgenation of the resulting sulfonamide using an alkyl isocyanate catalyst instead of hindered amine bases to simplify purification.
- Couple the phenylsulfonyl isocyanate intermediate with heterocyclic amines in a high-boiling solvent system to ensure complete conversion and high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the process described in CN1630647A offers substantial advantages for procurement managers and supply chain leaders focused on cost optimization and reliability. The elimination of hindered amine bases from the phosgenation step removes a significant cost center associated with reagent procurement and waste disposal. Additionally, the simplified purification process reduces the consumption of solvents and energy required for downstream processing, leading to a more sustainable and economically viable manufacturing operation. The robustness of the solvent system also enhances supply chain reliability by reducing the risk of batch failures due to side reactions or incomplete conversions. This stability is crucial for maintaining consistent delivery schedules to downstream formulators and ensuring that production targets are met without interruption. For organizations seeking a reliable agrochemical intermediate supplier, this technology represents a strategic asset that aligns with goals for cost reduction in herbicide manufacturing and operational excellence.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by eliminating the need for expensive hindered amine catalysts and reducing the complexity of the purification workflow. By avoiding the formation of difficult-to-remove by-products, the method minimizes the loss of valuable starting materials and reduces the volume of waste generated. This efficiency translates into direct savings on raw material procurement and waste treatment, making the overall manufacturing process more economically attractive. Furthermore, the ability to recover and reuse solvents like xylene and ethyl acetate contributes to long-term cost stability and environmental compliance.
- Enhanced Supply Chain Reliability: The high yields and consistent purity achieved through this method ensure a stable supply of high-quality intermediates. The robustness of the reaction conditions reduces the likelihood of batch-to-batch variability, which is a common cause of supply chain disruptions. By implementing this process, manufacturers can offer more reliable lead times and guarantee the availability of critical materials for their clients. This reliability is essential for maintaining trust with partners and securing long-term contracts in the competitive agrochemical market.
- Scalability and Environmental Compliance: The use of common industrial solvents and the avoidance of problematic reagents make this process highly scalable from laboratory to commercial production. The reduced generation of hazardous waste and the ability to recycle solvents align with increasingly strict environmental regulations. This compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer. The process is designed to handle large volumes efficiently, ensuring that production can be ramped up to meet market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process advantages and operational requirements. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this method for their production needs. The information provided here serves as a foundational resource for further technical discussions and feasibility assessments.
Q: How does the new solvent system improve yield compared to conventional THF methods?
A: The patented mixed solvent system comprising aromatic hydrocarbons and polar aprotic solvents significantly suppresses the formation of saccharin derivatives, a common by-product in conventional THF-based ammonolysis, thereby increasing theoretical yields from below 80% to over 90%.
Q: Why is the isocyanate catalyst preferred over hindered amine bases for phosgenation?
A: Using alkyl isocyanates as catalysts eliminates the need for hindered amine bases like DABCO, which are difficult and costly to remove from the final product. This simplifies the work-up process and enhances the overall purity of the sulfonyl isocyanate intermediate.
Q: What are the scalability advantages of this process for industrial manufacturing?
A: The process utilizes robust solvent systems like xylene and ethyl acetate which are easily recoverable and reusable. The high conversion rates and reduced by-product formation minimize waste treatment costs and facilitate seamless scale-up from pilot to commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Benzenesulfonylureas Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthesis technologies in delivering high-quality agrochemical intermediates to the global market. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in CN1630647A can be seamlessly integrated into our manufacturing operations. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of substituted benzenesulfonylureas meets the exacting standards required by our partners. Our infrastructure is designed to support the complex solvent systems and reaction conditions outlined in the patent, allowing us to deliver consistent quality at scale.
We invite you to collaborate with us to optimize your supply chain and leverage the commercial benefits of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this technology can enhance your product portfolio. By partnering with us, you gain access to a reliable source of high-purity intermediates backed by a commitment to technical excellence and customer satisfaction.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
