Advanced Solvent-Based Purification Technology for High-Purity Allopurinol Manufacturing
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and economic viability of critical active pharmaceutical ingredients (APIs). A significant advancement in this domain is detailed in patent CN103896944A, which introduces a novel purification strategy for allopurinol, a widely used xanthine oxidase inhibitor essential for treating hyperuricemia and gout. Traditional purification techniques have long struggled with the physicochemical limitations of allopurinol, particularly its poor solubility profile which necessitates energy-intensive processes. This new technical disclosure offers a transformative approach by leveraging high-boiling-point polar organic solvents to overcome these solubility barriers. By shifting away from aqueous systems to optimized organic media, the process not only drastically improves the yield and purity profiles but also addresses critical environmental concerns related to wastewater generation. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms for high-volume pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of allopurinol crude product has been plagued by significant inefficiencies inherent to its molecular structure. The primary conventional method involves aqueous recrystallization, which is severely hampered by the fact that allopurinol is only slightly soluble in water, with a maximum solubility of merely 1% even at elevated temperatures of 90-100°C. To dissolve a practical amount of crude material, manufacturers are forced to use up to 200 times the weight of the product in water, creating a massive thermal load that requires enormous amounts of steam for heating. Furthermore, the subsequent cooling crystallization step demands extensive cooling water resources to precipitate the product from such a dilute solution. This results in an exorbitant energy footprint and generates vast volumes of wastewater that require complex treatment before discharge. Alternatively, acid-alkali refining methods attempt to increase solubility by converting allopurinol into sodium salts; however, this introduces severe operational risks. The harsh pH swings required for salt formation and subsequent acidification can lead to product degradation, while the multi-step nature of the process complicates scale-up and increases the likelihood of introducing inorganic salt impurities that are difficult to remove.
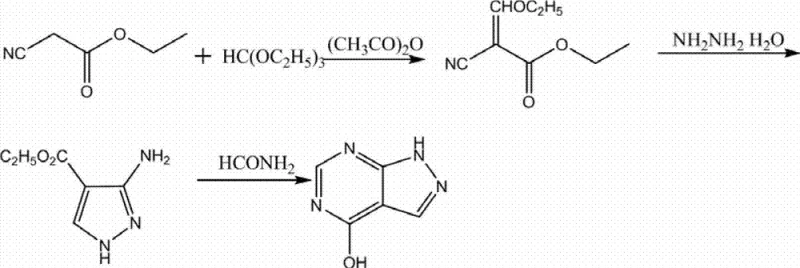
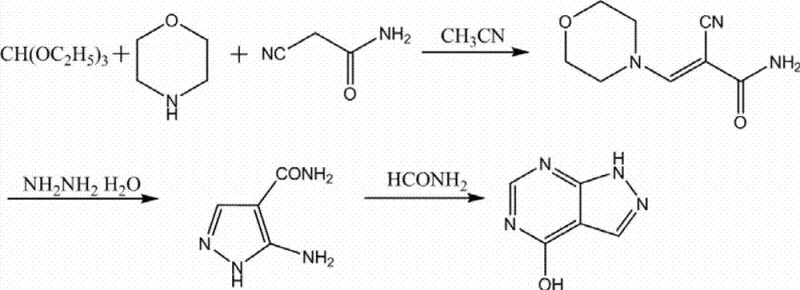
The Novel Approach
In stark contrast to these legacy techniques, the novel approach described in the patent utilizes a sophisticated solvent engineering strategy that fundamentally alters the thermodynamics of the purification process. By employing high-boiling-point polar organic solvents such as dimethyl sulfoxide (DMSO), dimethylformamide (DMF), or ethylene glycol, the process exploits the strong polarity of these media to achieve significantly higher solubility for allopurinol at temperatures ranging from 100°C to 190°C. This allows for the dissolution of crude material in a much more concentrated state, with solvent-to-product weight ratios as low as 4:1 to 30:1, compared to the 200:1 ratio required in water. The procedure involves dissolving the crude product under heat, followed by a critical decolorization step using activated carbon to adsorb colored impurities and trace organics. The solution is then filtered while hot to remove the carbon and any insoluble particulates, ensuring a clear mother liquor. Upon controlled cooling to room temperature, the solubility of allopurinol drops sharply in these specific solvents, inducing the formation of high-quality crystals. This method effectively bypasses the need for massive water volumes and avoids the chemical stress of acid-base conversions, resulting in a streamlined, single-phase purification workflow that is inherently safer and more efficient.
Mechanistic Insights into Solvent-Mediated Recrystallization
The efficacy of this purification method relies heavily on the specific interaction between the allopurinol molecule and the chosen polar aprotic or protic solvents. Allopurinol contains multiple hydrogen bond donors and acceptors within its pyrazolo-pyrimidine ring system, which interact strongly with the highly polar sulfoxide group of DMSO or the amide group of DMF. At elevated temperatures (120-150°C), these interactions overcome the lattice energy of the solid allopurinol, facilitating rapid dissolution. The choice of solvent is critical; DMSO is particularly effective due to its high dielectric constant and ability to stabilize the transition states of impurities, keeping them in solution even as the temperature drops. The decolorization mechanism involves the physical adsorption of large conjugated organic molecules onto the high-surface-area porous structure of the activated carbon. By maintaining the solution at high temperature during filtration, the process prevents premature nucleation of allopurinol on the filter cake, which would otherwise trap impurities and reduce yield. The subsequent cooling phase is governed by supersaturation kinetics; as the temperature decreases, the solvent's capacity to hold the solute diminishes, driving the nucleation and growth of pure allopurinol crystals while leaving soluble impurities in the mother liquor. This precise control over the solubility curve ensures that the crystalline lattice forms with minimal defect sites, thereby excluding impurity molecules from the crystal structure.
Impurity control is further enhanced by the neutral nature of the solvent system. Unlike acid-alkali methods where pH fluctuations can catalyze hydrolysis or decomposition of sensitive functional groups, the organic solvent environment maintains a neutral pH throughout the process. This stability is crucial for preventing the formation of degradation byproducts such as biuret or other urea derivatives that often plague allopurinol synthesis. The hot filtration step acts as a physical barrier, removing not only the decolorizing agent but also any high-molecular-weight polymeric byproducts that may have formed during the upstream synthesis (as seen in the condensation reactions of ethyl cyanoacetate or malonamide nitrile). The washing step post-filtration, typically using small amounts of purified water, serves to remove residual solvent adhering to the crystal surface without redissolving the product, thanks to allopurinol's low water solubility at ambient temperatures. This multi-layered approach to impurity management—combining chemical solubility selectivity, physical adsorption, and thermal fractionation—results in a final product with exceptional purity metrics, consistently achieving single impurity levels below 0.1% and total impurities well under pharmacopoeia limits.
How to Synthesize High-Purity Allopurinol Efficiently
Implementing this purification protocol requires precise control over thermal parameters and solvent ratios to maximize recovery while maintaining crystal quality. The process begins with the careful selection of the solvent system, where DMSO is often preferred for its optimal balance of solubility and ease of recovery, though DMF and ethylene glycol are viable alternatives depending on facility capabilities. Operators must ensure that the dissolution temperature is maintained strictly within the 100-190°C range to guarantee complete solubilization of the crude feedstock without risking thermal degradation of the API. The addition of activated carbon must be timed correctly after dissolution, allowing sufficient contact time (typically 15 minutes) for effective adsorption of chromophores. Following hot filtration, the cooling rate becomes a critical process parameter; too rapid cooling may lead to oiling out or the formation of fine, hard-to-filter crystals, while too slow cooling may reduce throughput. The detailed standardized synthesis steps, including specific weight ratios and drying conditions, are outlined in the technical guide below to ensure reproducibility across different manufacturing scales.
- Dissolve crude allopurinol in a high-boiling-point polar organic solvent (such as DMSO or DMF) at temperatures between 100-190°C.
- Perform decolorization treatment using activated carbon on the hot solution, followed immediately by hot filtration to remove insoluble impurities and carbon.
- Cool the filtrate to room temperature to induce crystallization, then separate, wash, and dry the purified allopurinol crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-based purification technology offers profound strategic advantages that extend far beyond simple yield improvements. The most immediate impact is observed in the drastic reduction of utility costs associated with the purification stage. By eliminating the need to heat and cool hundreds of liters of water per kilogram of product, facilities can realize substantial savings in steam and chilled water consumption. Furthermore, the ability to recycle the organic solvent completely creates a closed-loop system that minimizes raw material waste and reduces the burden on wastewater treatment plants. This shift from a linear, high-waste process to a circular, solvent-recovery model aligns perfectly with modern sustainability goals and regulatory pressures regarding industrial effluent. The simplified operational workflow also translates to reduced labor hours and lower maintenance costs, as the equipment is subjected to less corrosive stress compared to acid-alkali cycles.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the intensification of the production unit. Because the solvent volume required is significantly lower than that of water (reducing the liquid load by orders of magnitude), existing reactor vessels can process vastly larger batches of crude material without requiring capital investment in new infrastructure. This equipment capacity expansion, estimated to be between 5 to 40 times greater than water-based methods, allows manufacturers to spread fixed overhead costs over a much larger output volume. Additionally, the elimination of expensive acid and base reagents, along with the associated neutralization salts, removes a significant line item from the bill of materials. The high yield of approximately 90% ensures that valuable crude starting material is not lost to mother liquors or degradation, directly improving the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this purification method enhances overall supply security. The process is less sensitive to variations in crude quality compared to acid-alkali refining, which can fail if the impurity profile shifts unexpectedly. The use of common, commercially available solvents like DMSO and DMF ensures that raw material sourcing remains stable and unaffected by niche supply disruptions. Moreover, the shorter cycle time per batch—due to faster heating and cooling of smaller solvent volumes—increases the annual production capacity of the facility. This agility allows suppliers to respond more rapidly to fluctuations in market demand, reducing lead times for downstream pharmaceutical customers who rely on just-in-time delivery models for their formulation lines.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward because it relies on standard unit operations (dissolution, filtration, crystallization) that are well-understood in chemical engineering. The absence of complex pH control loops simplifies automation and reduces the risk of operator error during scale-up. Environmentally, the process is superior because the organic solvents can be distilled and reused indefinitely, virtually eliminating solvent discharge. The reduction in wastewater volume is particularly significant, easing the load on effluent treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this method in your own manufacturing context. Understanding these nuances is essential for R&D teams planning technology transfer and for quality assurance professionals establishing control strategies.
Q: Why is traditional water-based refining inefficient for allopurinol?
A: Traditional water refining requires massive amounts of water (200 times the weight of the product) due to allopurinol's low solubility, leading to excessive steam consumption for heating and significant wastewater generation during cooling.
Q: What solvents are optimal for this purification method?
A: The process utilizes high-boiling-point polar organic solvents such as dimethyl sulfoxide (DMSO), dimethylformamide (DMF), or ethylene glycol, which offer significantly higher solubility for allopurinol at elevated temperatures.
Q: What purity and yield levels can be achieved?
A: This method consistently achieves a yield of approximately 90% with a final product purity exceeding 99.88%, meeting stringent pharmacopoeia standards for single and total impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allopurinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced purification technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into robust industrial processes. Our facilities are equipped with state-of-the-art reactors and filtration systems capable of handling high-boiling solvents safely and efficiently. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of allopurinol meets or exceeds international pharmacopoeia standards, providing our clients with the confidence needed for regulatory filings and market entry.
We invite you to collaborate with us to leverage this cutting-edge purification method for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of this critical pharmaceutical ingredient.
