Advanced Synthesis of N-furanone Arylsulfonyl Hydrazones for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking novel molecular scaffolds that offer enhanced bioactivity while maintaining synthetic feasibility. Patent CN110511197B introduces a significant advancement in this domain by disclosing a new class of N-furanone arylsulfonyl hydrazone compounds. These molecules integrate a 2(5H)-furanone structural unit with an arylsulfonylhydrazone moiety, creating a unique pharmacophore with promising antitumor properties. The innovation lies not only in the biological potential but also in the streamlined synthetic methodology that allows for the efficient construction of these complex architectures. By leveraging a one-pot catalytic system, the process minimizes intermediate isolation steps, which is a critical factor for industrial viability. 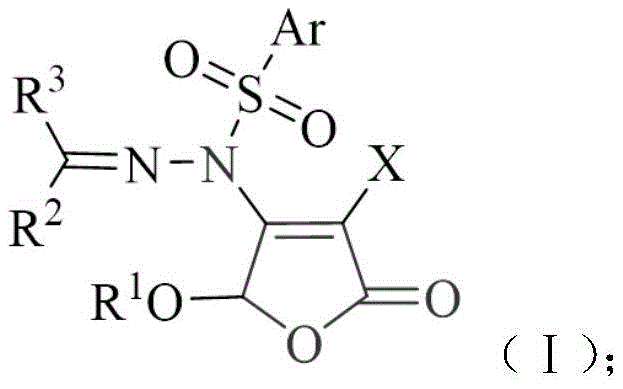 This structural versatility enables the generation of a diverse library of derivatives, allowing medicinal chemists to fine-tune physicochemical properties for optimal drug performance. The patent highlights the broad substrate scope, accommodating various aryl and heteroaryl groups, which is essential for structure-activity relationship (SAR) studies. For procurement and supply chain professionals, understanding the underlying chemistry of such intermediates is vital for assessing long-term availability and cost structures. The ability to synthesize these compounds under mild conditions suggests a robust manufacturing process that can be reliably scaled to meet commercial demand without excessive energy consumption or hazardous waste generation.
This structural versatility enables the generation of a diverse library of derivatives, allowing medicinal chemists to fine-tune physicochemical properties for optimal drug performance. The patent highlights the broad substrate scope, accommodating various aryl and heteroaryl groups, which is essential for structure-activity relationship (SAR) studies. For procurement and supply chain professionals, understanding the underlying chemistry of such intermediates is vital for assessing long-term availability and cost structures. The ability to synthesize these compounds under mild conditions suggests a robust manufacturing process that can be reliably scaled to meet commercial demand without excessive energy consumption or hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of arylsulfonylhydrazone-based bioactive molecules has been constrained by structural limitations in the existing literature. Most reported compounds in this class feature an N-methyl substitution at the 1-position or remain unsubstituted, which restricts the chemical space available for optimization. 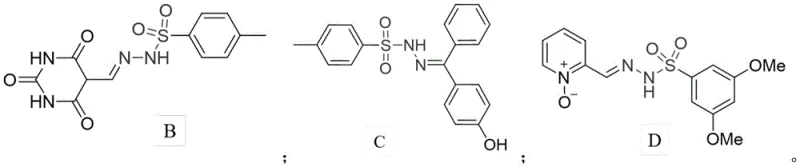 These conventional structures, while biologically active, often suffer from metabolic instability or limited solubility profiles that hinder their progression through clinical development pipelines. Furthermore, traditional synthetic routes to modify these scaffolds frequently involve multi-step sequences with harsh reaction conditions, leading to lower overall yields and increased production costs. The reliance on specific N-substitutions limits the ability to conjugate these pharmacophores with other functional groups, such as the furanone unit, which is known for its own distinct biological activities. Consequently, there has been a persistent need for a methodology that can overcome these structural bottlenecks and provide access to novel N-substituted derivatives with enhanced therapeutic potential.
These conventional structures, while biologically active, often suffer from metabolic instability or limited solubility profiles that hinder their progression through clinical development pipelines. Furthermore, traditional synthetic routes to modify these scaffolds frequently involve multi-step sequences with harsh reaction conditions, leading to lower overall yields and increased production costs. The reliance on specific N-substitutions limits the ability to conjugate these pharmacophores with other functional groups, such as the furanone unit, which is known for its own distinct biological activities. Consequently, there has been a persistent need for a methodology that can overcome these structural bottlenecks and provide access to novel N-substituted derivatives with enhanced therapeutic potential.
The Novel Approach
The methodology described in the patent data represents a paradigm shift by introducing a 2(5H)-furanone structural unit directly onto the sulfonyl hydrazide nitrogen. This innovative coupling strategy creates a new chemical entity that combines the biological benefits of both structural motifs. The synthesis is achieved through a concise reaction sequence that mixes arylsulfonylhydrazide, 3,4-dihalo-5-hydrocarbyloxy-2(5H)-furanone, and an aldehyde or ketone in a single vessel. This one-pot approach significantly reduces the operational complexity compared to stepwise synthesis, eliminating the need for intermediate purification and reducing solvent consumption. The use of readily available starting materials ensures that the supply chain remains resilient against raw material shortages. Additionally, the reaction proceeds with high efficiency, producing the target compounds in substantial yields without the formation of complex byproduct mixtures. This simplicity translates directly into commercial advantages, as it lowers the barrier for manufacturing scale-up and reduces the overall cost of goods sold for the final pharmaceutical intermediate.
Mechanistic Insights into DMAP-Catalyzed Condensation
The core of this synthetic breakthrough lies in the dual catalytic system employing an organic base and an acid to drive the condensation reaction. Specifically, 4-Dimethylaminopyridine (DMAP) acts as a nucleophilic catalyst, facilitating the activation of the electrophilic centers within the furanone and aldehyde components. 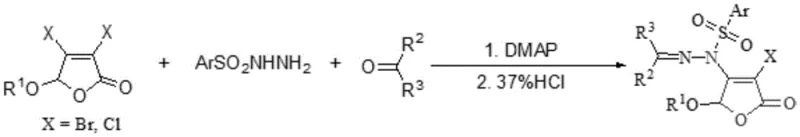 Simultaneously, the addition of hydrochloric acid creates an acidic environment that promotes the formation of the hydrazone linkage through dehydration. This synergistic catalytic effect allows the reaction to proceed smoothly at room temperature, avoiding the thermal degradation often associated with high-temperature processes. The mechanism ensures high regioselectivity, minimizing the formation of isomeric impurities that could complicate downstream purification. For R&D directors, this level of control over the reaction pathway is crucial for ensuring batch-to-batch consistency and meeting stringent purity specifications required for regulatory filings. The mild conditions also preserve sensitive functional groups on the aromatic rings, allowing for a wider range of substituents to be incorporated without protection-deprotection strategies. This mechanistic efficiency is the foundation upon which the commercial viability of the process is built, ensuring that the chemistry is robust enough for multi-kilogram production.
Simultaneously, the addition of hydrochloric acid creates an acidic environment that promotes the formation of the hydrazone linkage through dehydration. This synergistic catalytic effect allows the reaction to proceed smoothly at room temperature, avoiding the thermal degradation often associated with high-temperature processes. The mechanism ensures high regioselectivity, minimizing the formation of isomeric impurities that could complicate downstream purification. For R&D directors, this level of control over the reaction pathway is crucial for ensuring batch-to-batch consistency and meeting stringent purity specifications required for regulatory filings. The mild conditions also preserve sensitive functional groups on the aromatic rings, allowing for a wider range of substituents to be incorporated without protection-deprotection strategies. This mechanistic efficiency is the foundation upon which the commercial viability of the process is built, ensuring that the chemistry is robust enough for multi-kilogram production.
Impurity control is another critical aspect addressed by this mechanistic design. The use of specific halogenated furanone precursors ensures that the reaction pathway is directed towards the desired N-substituted product rather than competing O-alkylation or polymerization side reactions. The stoichiometry of the reagents is carefully balanced, with a slight excess of the sulfonyl hydrazide and aldehyde components to drive the equilibrium towards completion. This strategic excess ensures that the limiting reagent, the furanone derivative, is fully consumed, maximizing the atom economy of the process. The workup procedure involves a simple aqueous extraction followed by organic solvent separation, which effectively removes inorganic salts and catalyst residues. This straightforward isolation method reduces the reliance on expensive chromatographic media for bulk purification, further enhancing the cost-effectiveness of the manufacturing process. The resulting solid products exhibit sharp melting points and consistent spectral data, indicating a high degree of crystalline purity suitable for direct use in subsequent drug synthesis steps.
How to Synthesize N-furanone Arylsulfonyl Hydrazone Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to ensure safety and quality. The process begins with the dissolution of the furanone precursor and sulfonyl hydrazide in a compatible organic solvent such as dichloromethane. The detailed standardized synthesis steps see the guide below. Following the reaction, the mixture is quenched with water and extracted to isolate the crude product. Final purification is achieved through silica gel column chromatography or recrystallization, yielding the target compound as a high-purity solid. This protocol is designed to be adaptable, allowing process chemists to optimize solvent volumes and stirring rates based on reactor geometry.
- Mix 3,4-dihalo-5-hydrocarbyloxy-2(5H)-furanone with arylsulfonylhydrazide and DMAP in solvent.
- Add concentrated hydrochloric acid and aldehyde/ketone compound at room temperature.
- Stir, extract with dichloromethane, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in the patent offers substantial opportunities for cost optimization and supply chain stabilization. The reliance on commercially available starting materials means that sourcing risks are minimized, as these chemicals are produced by multiple vendors globally. Cost Reduction in Manufacturing: The elimination of extreme temperature requirements significantly lowers energy consumption, as the reaction proceeds efficiently at ambient conditions. This reduction in thermal load translates to lower utility costs and reduced wear on reactor equipment over time. Furthermore, the one-pot nature of the synthesis reduces labor hours associated with intermediate handling and transfer, leading to substantial operational savings. The high yield profile ensures that raw material waste is minimized, improving the overall material balance and reducing the cost per kilogram of the active intermediate. These factors combine to create a highly competitive cost structure that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts or cryogenic cooling systems, which are often cost drivers in fine chemical synthesis. By utilizing simple organic bases and mineral acids, the reagent costs are kept to a minimum while maintaining high reaction efficiency. The simplified workup procedure reduces the volume of solvents required for extraction and purification, lowering both procurement costs and waste disposal fees. This lean manufacturing approach ensures that the production of these intermediates remains economically viable even at large scales.
- Enhanced Supply Chain Reliability: The use of common organic solvents and widely available reagents ensures that the supply chain is not dependent on single-source suppliers for exotic chemicals. This diversification of the supply base mitigates the risk of production delays caused by logistical bottlenecks or vendor shortages. The robustness of the reaction conditions means that manufacturing can be easily transferred between different facilities without significant re-validation efforts. This flexibility is crucial for maintaining continuous supply to downstream pharmaceutical customers who require just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste, as the byproducts are primarily inorganic salts that can be easily treated in standard wastewater facilities. The absence of heavy metals in the catalyst system simplifies the regulatory compliance process for environmental discharge. The process is inherently scalable, as the heat generation is manageable under ambient conditions, reducing the need for complex cooling infrastructure in large reactors. This environmental profile aligns with modern green chemistry principles, making the process attractive for companies with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. 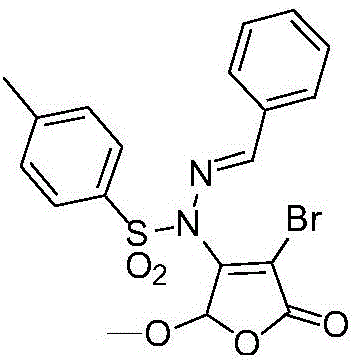 Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their drug development pipelines. The information provided here serves as a foundational reference for further discussions on customization and scale-up.
Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their drug development pipelines. The information provided here serves as a foundational reference for further discussions on customization and scale-up.
Q: What are the key advantages of this synthesis method?
A: The method features mild room temperature conditions, readily available raw materials, and a simplified one-pot procedure that eliminates complex purification steps.
Q: What is the primary application of these compounds?
A: These N-furanone arylsulfonyl hydrazone derivatives exhibit significant inhibitory activity against tumor cells, specifically for antitumor drug development.
Q: Is this process scalable for industrial production?
A: Yes, the use of common organic solvents and ambient reaction conditions facilitates straightforward scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-furanone Arylsulfonyl Hydrazone Supplier
NINGBO INNO PHARMCHEM stands ready to support your pharmaceutical development needs with our expertise in complex intermediate synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee the quality of every batch. We understand the critical nature of antitumor drug development and are committed to providing intermediates that meet the highest industry standards. Our team of expert chemists can assist in optimizing the synthesis route to further enhance yield and reduce costs specific to your volume needs.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain economics. Let us help you accelerate your drug development timeline with our reliable manufacturing capabilities and deep technical knowledge. Reach out today to discuss how we can support your next breakthrough in oncology research.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
