Advanced Synthetic Route for High-Purity Cyano-formamide Compounds Using Novel Activating Reagents
Introduction to Next-Generation Cyano-formamide Synthesis
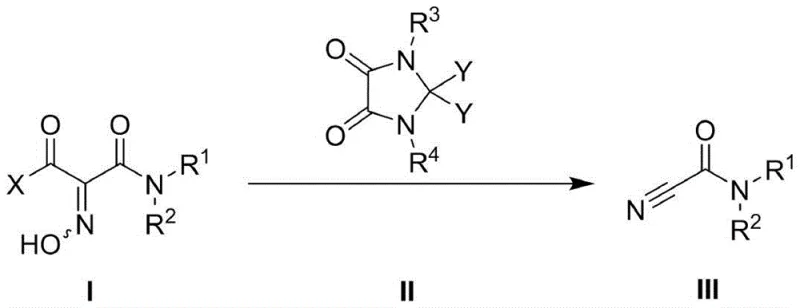
The landscape of organic synthesis is constantly evolving towards greener, safer, and more efficient methodologies, particularly for critical building blocks like cyano-formamide compounds. A pivotal advancement in this domain is detailed in patent CN109810020B, which discloses a novel method for synthesizing cyano-formamide derivatives using 1-acyl-1-carbamoyl oxime precursors mediated by a unique class of activating reagents. This technology represents a significant departure from traditional, hazardous protocols, offering a robust pathway for constructing bioactive molecules such as ureas, acrylonitriles, and tetrazoles. By leveraging 2,2-dihaloimidazolidinedione compounds as mediators, this approach achieves high yields under remarkably mild conditions, addressing long-standing challenges in impurity control and operational safety.
For global procurement leaders and R&D directors, the implications of this patent extend beyond mere academic interest; it offers a tangible solution for cost reduction in pharmaceutical intermediate manufacturing. The ability to utilize structurally tunable activators allows for precise optimization of reaction kinetics, ensuring consistent quality across batches. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is crucial for securing supply chains that are both resilient and compliant with increasingly stringent environmental regulations. The following analysis dissects the technical merits and commercial viability of this innovative synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyano-formamide compounds has relied heavily on reagents that pose severe safety and environmental risks. Traditional protocols often employ phosphorus oxychloride (POCl3), carbonyl cyanide, or 4-chloro-5H-1,2,3-dithiazol-5-one, all of which are characterized by high toxicity, corrosivity, and potential explosiveness. These reagents not only endanger laboratory personnel and plant operators but also generate substantial quantities of hazardous waste, complicating disposal and increasing the overall carbon footprint of the manufacturing process. Furthermore, many conventional activators lack structural versatility, making it difficult to fine-tune steric and electronic properties to accommodate sensitive substrates. This rigidity often leads to side reactions, lower yields, and complex purification workflows, ultimately driving up the cost of goods sold (COGS) for downstream API manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in CN109810020B introduces a paradigm shift by utilizing 2,2-dihaloimidazolidinedione compounds (Formula II) as activating agents. These reagents are inherently stable, easy to synthesize from cheap raw materials, and possess a modular structure that allows for extensive regulation of steric and electronic environments. The reaction proceeds smoothly at temperatures ranging from 25°C to 80°C, eliminating the need for energy-intensive heating or cryogenic cooling. This mildness translates directly into enhanced functional group tolerance, enabling the successful transformation of substrates bearing sensitive moieties such as boronic acids, nitro groups, and halogens without degradation. The result is a cleaner reaction profile with higher isolated yields, simplifying the downstream purification process and significantly reducing solvent consumption.
Mechanistic Insights into 2,2-Dihaloimidazolidinedione Mediated Activation
The core of this technological breakthrough lies in the unique reactivity of the 2,2-dihaloimidazolidinedione scaffold. Mechanistically, the halogen atoms at the 2-position of the imidazolidinedione ring act as potent electrophilic centers, facilitating the activation of the hydroxyl group in the 1-acyl-1-carbamoyl oxime (Formula I). This activation promotes an elimination sequence that efficiently converts the oxime functionality into the desired nitrile group while preserving the amide bond. The cyclic urea structure of the activator provides a rigid framework that stabilizes the transition state, thereby lowering the activation energy required for the dehydration step. This precise control over the reaction pathway minimizes the formation of by-products, such as hydrolyzed acids or polymerized species, which are common pitfalls in harsher acidic or basic conditions.
Furthermore, the substituent diversity on the nitrogen atoms of the activator (R3 and R4) plays a critical role in modulating solubility and reactivity. As demonstrated in the patent examples, varying these groups from simple alkyl chains to bulky adamantyl or silyl groups allows chemists to tailor the reagent for specific substrate classes. For instance, electron-withdrawing groups on the activator can enhance its electrophilicity, accelerating the reaction for less reactive substrates, while bulky groups can prevent unwanted nucleophilic attacks on the activator itself. This level of tunability ensures that the process remains robust across a wide array of chemical spaces, making it a versatile tool for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Cyano-formamide Compounds Efficiently
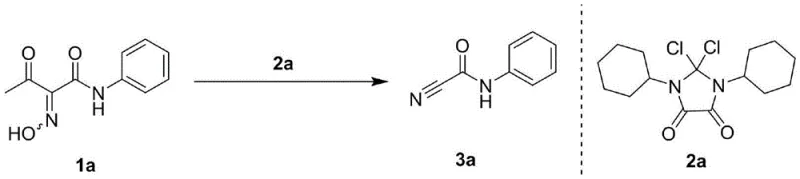
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize efficiency and safety. The process is designed to be operationally simple, typically involving the dissolution of the oxime substrate and the activating reagent in a common organic solvent such as acetonitrile, dichloromethane, or ethyl acetate. The reaction mixture is then stirred at moderate temperatures, typically around 60°C, until conversion is complete as monitored by TLC or HPLC. Workup involves standard extraction and washing procedures, followed by purification via column chromatography or recrystallization. The detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Dissolve the 1-acyl-1-carbamoyl oxime compound (Formula I) and the 2,2-dihaloimidazolidinedione activating reagent (Formula II) in a suitable organic solvent such as acetonitrile or dichloromethane.
- Stir the reaction mixture at a mild temperature ranging from 25°C to 80°C, maintaining a molar ratio of substrate to activator between 1: 1 and 1:1.5.
- Upon completion, extract the product with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. By replacing hazardous reagents like POCl3 with stable, solid 2,2-dihaloimidazolidinediones, manufacturers can drastically reduce the costs associated with specialized corrosion-resistant equipment and hazardous waste disposal. The mild reaction conditions also lower energy consumption, contributing to a more sustainable manufacturing profile that aligns with modern ESG (Environmental, Social, and Governance) goals. Moreover, the high functional group compatibility reduces the risk of batch failures due to side reactions, ensuring a more predictable and reliable supply of high-quality intermediates for downstream drug development.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents significantly lowers raw material costs and safety overheads. The simplified workup procedure reduces solvent usage and processing time, leading to substantial operational savings. Additionally, the high yields reported in the patent minimize material loss, further enhancing the overall economic efficiency of the production process.
- Enhanced Supply Chain Reliability: The activating reagents used in this method are synthesized from readily available starting materials, reducing the risk of supply bottlenecks associated with exotic or regulated chemicals. The stability of these reagents allows for long-term storage without degradation, enabling manufacturers to maintain strategic stockpiles and ensure continuous production even during market fluctuations.
- Scalability and Environmental Compliance: The absence of toxic gases and corrosive liquids simplifies the engineering controls required for scale-up, making it easier to transition from pilot plant to full commercial production. The greener nature of the process facilitates regulatory approval and reduces the environmental liability of the manufacturing site, positioning the supplier as a preferred partner for eco-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using 2,2-dihaloimidazolidinedione over traditional reagents like POCl3?
A: Unlike phosphorus oxychloride (POCl3), which is highly toxic, corrosive, and generates hazardous waste, 2,2-dihaloimidazolidinedione reagents are solid, user-friendly, and operate under mild conditions (25-80°C), significantly improving operator safety and environmental compliance.
Q: Does this synthesis method tolerate sensitive functional groups?
A: Yes, the method exhibits excellent functional group compatibility. It successfully tolerates diverse substituents including halogens, trifluoromethyl groups, nitro groups, cyano groups, hydroxyls, alkoxy groups, and even sensitive boronic acid esters, making it ideal for complex API intermediate synthesis.
Q: Is this process scalable for industrial production?
A: Absolutely. The reaction uses commercially available solvents like acetonitrile and operates at near-ambient temperatures without requiring cryogenic conditions or high pressure, facilitating straightforward scale-up from laboratory grams to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyano-formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical industry. Our team of expert chemists has extensively evaluated the methodology described in CN109810020B and possesses the technical capability to execute this route with precision and scalability. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from development to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyano-formamide intermediate delivered meets the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable, high-performance chemical solutions.
