Scalable Synthesis of Optically Active Beta-Aryl Homoallylic Amines for DPPIV Inhibitors
The pharmaceutical industry's relentless pursuit of effective treatments for Type II diabetes has placed significant emphasis on the development of Dipeptidyl Peptidase IV (DPPIV) inhibitors. As detailed in patent CN101962350A, a breakthrough methodology has been established for the synthesis of optically active beta-aryl homoallylic amine compounds, which serve as pivotal chiral building blocks in this therapeutic class. This technology leverages a metal-promoted asymmetric aryl allylation reaction, specifically utilizing chiral N-tert-butylsulfinyl imines as the electrophilic partners. The innovation addresses a critical bottleneck in the supply chain for high-purity pharmaceutical intermediates by offering a route that operates under mild conditions while delivering exceptional stereochemical fidelity. For R&D teams and procurement specialists alike, understanding the nuances of this zinc or indium-promoted transformation is essential for securing a reliable supply of next-generation antidiabetic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the beta-aryl homoallylic amine scaffold has relied on several distinct strategies, each fraught with significant operational and economic drawbacks that hinder large-scale manufacturing. Traditional approaches often include aza-Barbier type aryl allylation reactions of imines, which frequently suffer from moderate yields and poor stereocontrol, necessitating costly and wasteful purification steps to isolate the desired enantiomer. Furthermore, palladium-catalyzed aryl allylation methods, while effective in some contexts, introduce the persistent challenge of residual heavy metal contamination, requiring rigorous and expensive scavenging processes to meet stringent regulatory limits for Active Pharmaceutical Ingredients (APIs). Additionally, acid-catalyzed three-component coupling reactions involving aldehydes, amines, and allyl tributyltin reagents have been reported, but these often exhibit only medium levels of cis-selectivity and enantioselectivity, typically ranging between 52% and 78% ee, which is insufficient for the production of high-value chiral drugs without extensive recycling or resolution protocols.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a zinc or indium-promoted asymmetric allylation of chiral N-tert-butylsulfinyl imines. This methodology represents a paradigm shift by combining the use of inexpensive, non-toxic metallic powders with a robust chiral auxiliary strategy. The reaction proceeds efficiently at room temperature in polar aprotic solvents like DMF, eliminating the need for cryogenic conditions or specialized high-pressure equipment. Crucially, this process demonstrates remarkable substrate tolerance, accommodating a wide array of aryl and heteroaryl groups while consistently delivering products with diastereomeric excess (de) values exceeding 95%. By shifting away from precious metal catalysis and tin reagents, this route not only enhances the environmental profile of the synthesis but also drastically simplifies the downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Zinc-Promoted Asymmetric Allylation
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the chiral tert-butylsulfinyl group and the activated metal surface. When the N-tert-butylsulfinyl imine reacts with the aryl allyl halide in the presence of zinc, an organozinc species is generated in situ, which then undergoes a highly organized addition to the imine double bond. The bulky tert-butylsulfinyl moiety exerts profound steric influence, directing the incoming allyl group to attack from a specific face of the imine, thereby establishing the new chiral centers with high precision. Interestingly, the reaction exhibits a strong preference for the formation of the syn-diastereomer, a feature that remains consistent regardless of whether the starting allyl halide possesses a cis or trans olefin geometry. This geometric independence is a significant advantage for process chemistry, as it allows manufacturers to utilize the most commercially available and cost-effective isomer of the allyl halide starting material without compromising the stereochemical outcome of the final product.
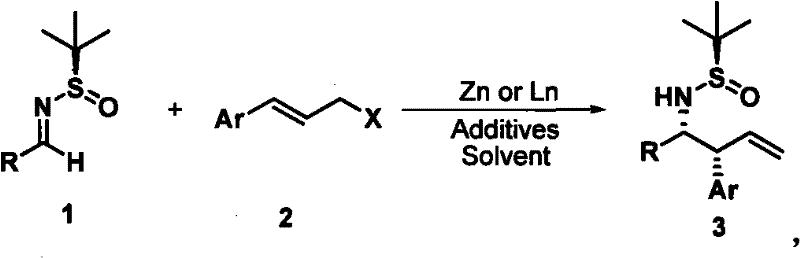
From an impurity control perspective, the high diastereoselectivity of this reaction serves as a built-in purification mechanism. Because the formation of the unwanted anti-diastereomer is suppressed to levels below 5%, the burden on chromatographic separation is significantly reduced. This inherent selectivity minimizes the generation of difficult-to-remove stereoisomeric impurities that could otherwise persist through subsequent synthetic steps and compromise the safety profile of the final drug substance. Furthermore, the use of additives such as lithium chloride or other halide salts has been shown to enhance the reactivity of the metal surface and stabilize the transition state, further pushing the yields towards quantitative levels (often exceeding 90%) while maintaining the rigorous stereochemical standards required for regulatory approval.
How to Synthesize Optically Active Beta-Aryl Homoallylic Amines Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction efficiency with operational simplicity, making it highly attractive for scale-up. The process begins with the preparation of the reaction vessel under an inert atmosphere, followed by the sequential addition of the chiral imine, the aryl allyl halide, activated zinc powder, and a halide additive in a dry solvent system. The reaction mixture is stirred at ambient temperature, allowing the transformation to proceed to completion within a few hours, after which standard aqueous workup procedures are employed to isolate the crude product. This streamlined workflow eliminates the need for complex temperature cycling or hazardous reagents, aligning perfectly with modern green chemistry principles. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- React N-tert-butylsulfinyl imine with aryl allyl halide reagent in the presence of activated zinc powder and additives like lithium chloride in DMF solvent at room temperature.
- Quench the reaction mixture and perform aqueous workup using hydrochloric acid, sodium bicarbonate, and brine to isolate the crude sulfinyl-protected amine.
- Purify the product via rapid column chromatography to obtain the optically active beta-aryl homoallylic amine with high diastereomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-promoted synthesis route offers tangible strategic benefits that extend far beyond the laboratory bench. The primary advantage lies in the substantial cost optimization achieved by replacing expensive transition metal catalysts and toxic organotin reagents with commodity-grade zinc powder. This substitution not only lowers the direct material cost but also mitigates the financial risks associated with the volatility of precious metal markets. Moreover, the elimination of heavy metals from the process flow removes the necessity for specialized metal scavenging resins and the associated validation testing, resulting in a leaner, more agile manufacturing process that can respond rapidly to market demand fluctuations without being bogged down by complex purification bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the use of earth-abundant metals and the high atom economy of the reaction. By avoiding the use of palladium or indium (where zinc is sufficient), the raw material expenditure is drastically simplified. Additionally, the high yields and stereoselectivity mean that less starting material is wasted on correcting isomeric errors, leading to a more efficient utilization of resources. The simplified workup procedure further reduces solvent consumption and waste disposal costs, contributing to a lower overall cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable starting materials. Unlike specialized chiral catalysts that may have long lead times or single-source dependencies, activated zinc powder and aryl allyl halides are commodity chemicals available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of failed batches that could disrupt inventory levels and delay deliveries to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton production is facilitated by the benign nature of the reagents and the absence of exothermic hazards typically associated with cryogenic organometallic reactions. The operation at room temperature reduces energy consumption for heating or cooling, aligning with sustainability goals. From a regulatory standpoint, the absence of tin and low levels of zinc residues simplify the environmental permitting process and ensure easier compliance with increasingly strict guidelines regarding heavy metal content in pharmaceutical products, thereby future-proofing the manufacturing asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing pipelines, we have compiled answers to common inquiries regarding the scope and limitations of the patent. These insights are derived directly from the experimental data and claims presented in the intellectual property documentation, ensuring accuracy and relevance for process development teams. Understanding these technical specifics is crucial for assessing the fit of this methodology within your specific project timelines and quality targets.
Q: What is the stereoselectivity of this zinc-promoted allylation method?
A: The method achieves excellent stereocontrol, typically providing syn-diastereomers with ratios exceeding 95:5 and diastereomeric excess (de) values ranging from 95% to 99%, regardless of the initial alkene geometry.
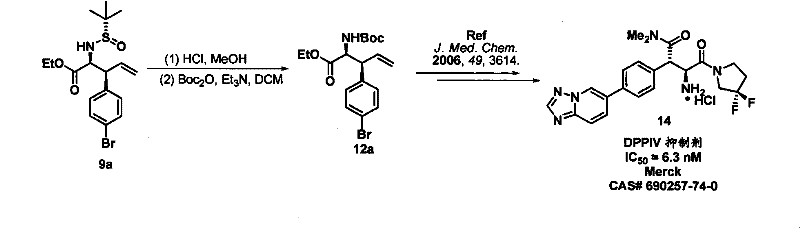
Q: Can this method be applied to glyoxylic ester imines for API synthesis?
A: Yes, the protocol is highly effective for glyoxylic ester imines, yielding protected amino acid derivatives that serve as critical precursors for DPPIV inhibitors like Sitagliptin.
Q: What are the advantages of using zinc over palladium catalysts in this reaction?
A: Using zinc powder eliminates the need for expensive and toxic transition metal catalysts, significantly reducing raw material costs and simplifying the removal of heavy metal residues from the final pharmaceutical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aryl Homoallylic Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of optically active beta-aryl homoallylic amines meets the exacting standards required for DPPIV inhibitor synthesis. Our capability to handle chiral auxiliaries and manage stereoselective reactions positions us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this innovative zinc-promoted allylation technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data for our reference standards and to initiate a dialogue regarding route feasibility assessments for your target molecules, ensuring a secure and efficient supply chain for your critical drug development programs.
