Strategic Manufacturing of High-Purity p-Methoxyphenylacetic Acid via Renewable Anethole Oxidation
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to petrochemical-derived intermediates, particularly for high-volume compounds like p-methoxyphenylacetic acid, also known as homoanisic acid. Patent CN101298416B introduces a groundbreaking methodology that shifts the synthetic paradigm from toxic, fossil-fuel-based precursors to renewable natural resources. This technical disclosure outlines a robust three-step sequence starting from natural anethole (methyl allylphenol), proceeding through oxidative cleavage to p-methoxybenzaldehyde, followed by a carbene insertion-like haloform reaction, and concluding with a selective reduction. For R&D directors and procurement strategists, this patent represents a significant opportunity to decouple supply chains from volatile petrochemical markets while adhering to stricter environmental regulations regarding cyanide and heavy metal usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of homoanisic acid has relied on several chemically aggressive and environmentally hazardous pathways that pose substantial operational risks. The traditional cyanide process, for instance, utilizes highly toxic potassium cyanide to generate cyanohydrins, creating severe safety liabilities for operators and necessitating complex wastewater treatment systems to neutralize cyanide residues before discharge. Similarly, the benzyl cyanide hydrolysis method suffers from low reaction yields and the inherent dangers of handling sodium cyanide on a commercial scale. Other methods, such as the Grignard reagent approach, require stringent anhydrous conditions and cryogenic temperatures that are energy-intensive and difficult to maintain in large-scale reactors. Furthermore, the oxo synthesis route depends on expensive carbonylation catalysts that are prone to poisoning and deactivation, alongside the use of toxic carbon monoxide gas under pressure, which complicates plant safety protocols and increases capital expenditure for specialized high-pressure equipment.
The Novel Approach
In stark contrast, the methodology described in the patent leverages the abundant availability of natural anethole to create a cleaner, more efficient synthetic trajectory. By utilizing oxidative cleavage, either via ozonolysis or permanganate oxidation, the process efficiently converts the alkene functionality of anethole directly into the requisite aldehyde intermediate with high atom economy. The subsequent transformation employs a phase-transfer catalyzed haloform reaction, which operates under mild alkaline conditions (30-80°C) and eliminates the need for hazardous cyanide reagents entirely. The final reduction step utilizes accessible reducing agents like stannous chloride or sodium pyrosulfite under acidic conditions, avoiding the complex catalytic systems of previous art. This holistic approach not only simplifies the operational workflow by allowing for telescoped steps but also drastically reduces the generation of hazardous byproducts, aligning perfectly with modern green chemistry principles and reducing the overall cost of goods sold through simplified waste management.
Mechanistic Insights into Anethole Oxidative Cleavage and Haloform Conversion
The core of this synthetic strategy lies in the precise control of the oxidative cleavage of the propenyl side chain of anethole. In the ozonolysis variant, ozone reacts with the carbon-carbon double bond to form a molozonide, which rearranges to a stable ozonide; this intermediate is then subjected to reductive decomposition using agents like sodium bisulfite at controlled temperatures (60-90°C) to yield p-methoxybenzaldehyde selectively. Alternatively, the use of potassium permanganate or manganese dioxide offers a non-ozonolytic pathway where the oxidant directly attacks the alkene, though careful stoichiometry (e.g., a 1:2.5 molar ratio for KMnO4) is required to prevent over-oxidation to the carboxylic acid at this stage. The selectivity of this initial oxidation is paramount, as it sets the purity profile for the entire downstream process, minimizing the formation of polymeric tars or over-oxidized acids that are difficult to separate.
Following the formation of the aldehyde, the mechanism shifts to a base-mediated haloform-type reaction facilitated by phase-transfer catalysts (PTC) such as tetrabutylammonium bromide (TBAB). Under strong alkaline conditions (using NaOH or KOH), the PTC transports hydroxide ions into the organic phase, promoting the formation of a trihalomethyl intermediate which subsequently undergoes nucleophilic attack to insert an oxygenated carbon unit, effectively generating the p-methoxymandelic acid salt. This step is critical for extending the carbon chain while introducing the alpha-hydroxy functionality. The final mechanistic step involves the reductive deoxygenation of the benzylic hydroxyl group. Using reducing agents like stannous chloride in concentrated hydrochloric acid at elevated temperatures (80-120°C), the C-O bond is cleaved to yield the methylene group of the acetic acid side chain. This reduction is highly chemoselective, preserving the aromatic methoxy group and the carboxylic acid functionality while removing the oxygen atom, resulting in the target p-methoxyphenylacetic acid with high structural fidelity.
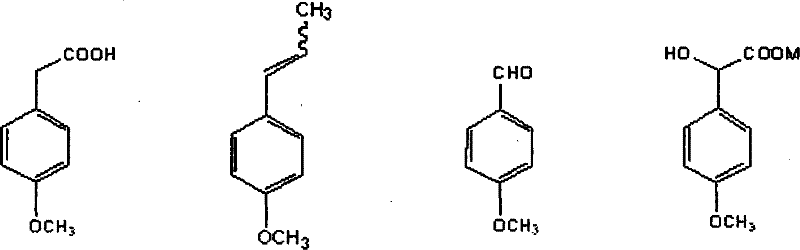
How to Synthesize p-Methoxyphenylacetic Acid Efficiently
The synthesis of this valuable pharmaceutical intermediate requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process begins with the preparation of the oxidation reactor, where natural anethole is dissolved in a suitable solvent system such as a mixture of cyclohexane and ethyl acetate to facilitate mass transfer during the ozonolysis or permanganate oxidation. Following the isolation of the aldehyde intermediate, the reaction mixture is adjusted to a strongly alkaline pH, and the phase-transfer catalyst is introduced to drive the haloform reaction to completion over a period of 2 to 12 hours. Detailed standardized operating procedures regarding temperature ramping, reagent addition rates, and workup protocols are essential for reproducibility. For the complete, step-by-step laboratory and pilot-scale instructions, please refer to the structured guide below.
- Oxidative Cleavage: Convert natural anethole to p-methoxybenzaldehyde using ozone or potassium permanganate under controlled temperatures.
- Haloform Reaction: React the aldehyde with alkali and a phase-transfer catalyst to form p-methoxymandelic acid salt.
- Reductive Deoxygenation: Reduce the mandelic acid derivative using stannous chloride or sodium pyrosulfite under acidic conditions to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anethole-based route offers compelling strategic advantages that extend beyond simple chemical yield. By shifting the raw material base from petrochemical derivatives like benzyl chloride to natural anethole, manufacturers can insulate their supply chains from the volatility of crude oil prices and the geopolitical instability often associated with fossil fuel sourcing. The elimination of highly toxic cyanide salts removes a significant regulatory burden, reducing the costs associated with hazardous material storage, transportation, and specialized waste disposal permits. Furthermore, the mild reaction conditions—operating largely at atmospheric pressure and moderate temperatures—reduce the energy consumption profile of the manufacturing plant, contributing to lower utility costs and a smaller carbon footprint, which is increasingly a requirement for Tier 1 suppliers in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the purification train and the avoidance of expensive, single-use catalysts. Unlike the oxo synthesis method which relies on precious metal catalysts that are difficult to recover and prone to deactivation, this method utilizes inexpensive, commodity-grade reagents like sodium hydroxide and stannous chloride. The ability to telescope the reaction steps without rigorous intermediate purification, particularly between the haloform reaction and the final reduction, significantly reduces solvent consumption and processing time. Additionally, the use of renewable natural feedstocks often provides a more stable long-term pricing structure compared to fluctuating petrochemical markets, leading to substantial and sustained cost savings in the final active pharmaceutical ingredient (API) production.
- Enhanced Supply Chain Reliability: Sourcing natural anethole, which is abundantly available from essential oil industries (such as star anise or fennel), diversifies the supplier base and reduces dependency on a limited number of petrochemical producers. The robustness of the chemical process, which tolerates a wider range of operating conditions compared to sensitive Grignard or high-pressure carbonylation reactions, ensures higher batch success rates and consistent output volumes. This reliability is crucial for maintaining continuous production schedules for downstream drugs like venlafaxine, preventing costly stockouts and ensuring that contractual delivery obligations to major pharmaceutical partners are consistently met without interruption due to process upsets or raw material shortages.
- Scalability and Environmental Compliance: From a scale-up perspective, the absence of high-pressure gas handling (carbon monoxide) and cryogenic requirements makes this technology readily adaptable to existing multipurpose chemical reactors without the need for massive capital investment in new infrastructure. The environmental profile is significantly improved by the elimination of cyanide-containing effluents and hydrogen sulfide byproducts, which are common in older synthetic routes. This compliance with stringent environmental standards facilitates faster regulatory approvals for new manufacturing sites and reduces the risk of production shutdowns due to environmental non-compliance. The simplified waste stream, consisting mainly of aqueous salts and organic solvents that are easier to treat or recycle, further enhances the sustainability credentials of the manufacturing site, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route for p-methoxyphenylacetic acid. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that potential partners have a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or for assessing the quality of the supplied intermediates.
Q: Why is the anethole route preferred over the cyanide process for homoanisic acid?
A: The traditional cyanide process involves highly toxic raw materials like potassium cyanide, posing severe environmental and safety risks. The patented anethole route utilizes renewable natural feedstocks and avoids cyanide entirely, significantly reducing hazardous waste treatment costs and operator exposure risks while maintaining high selectivity.
Q: What are the critical parameters for the oxidative cleavage step?
A: Temperature control is vital, typically maintained between 0°C and 50°C depending on the oxidant used (ozone vs. permanganate). The molar ratio of oxidant to substrate must be carefully balanced (e.g., 1:1.2 for ozone) to prevent over-oxidation, ensuring high yields of the intermediate p-methoxybenzaldehyde without forming excessive carboxylic acid byproducts.
Q: How does this method impact commercial scalability?
A: The process eliminates the need for high-pressure carbon monoxide equipment required in oxo synthesis and avoids the difficult-to-remove heavy metal catalysts associated with other methods. The use of standard phase-transfer catalysis and ambient pressure reactions simplifies reactor requirements, making the transition from pilot scale to multi-ton commercial production more straightforward and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Methoxyphenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of complex pharmaceutical agents. Our technical team has extensively analyzed the pathway described in CN101298416B and possesses the expertise to optimize this renewable route for industrial application. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of p-methoxyphenylacetic acid meets the exacting standards required for API synthesis, thereby mitigating the risk of downstream impurity propagation.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chains through this advanced, eco-friendly technology. By leveraging our process engineering capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our commitment to sustainable chemistry can drive value for your organization. Let us be your strategic partner in delivering high-quality chemical solutions that power the next generation of life-saving medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
