Accelerating Organometallic Production: Microwave-Assisted Synthesis of Arene Ruthenium(II) Complexes
The landscape of organometallic chemistry is undergoing a significant transformation driven by the need for greener, faster, and more efficient synthetic methodologies. Patent CN102898480A introduces a groundbreaking microwave-assisted synthesis method for arene ruthenium(II) compounds, addressing critical bottlenecks in the production of these valuable pharmaceutical intermediates. Traditionally, the coordination of ruthenium centers with complex ligands like phenanthroimidazole derivatives required prolonged reflux periods, often spanning several hours to days, with inconsistent results. This new technology leverages dielectric heating to accelerate molecular motion and collision frequency, enabling the completion of reactions in mere minutes. The core innovation lies in the ability to achieve yields exceeding 90% within a timeframe of 20 to 30 minutes, a stark contrast to the sluggish kinetics of conventional thermal heating. 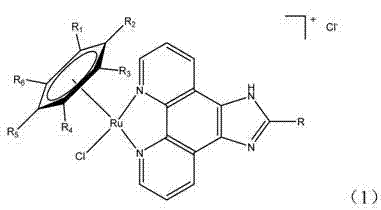 . For R&D directors and process chemists, this represents a paradigm shift, offering a robust pathway to access high-purity organometallic scaffolds essential for anticancer drug development and catalytic applications.
. For R&D directors and process chemists, this represents a paradigm shift, offering a robust pathway to access high-purity organometallic scaffolds essential for anticancer drug development and catalytic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arene ruthenium(II) complexes has been plagued by inefficiencies inherent to conductive heating methods. As documented in prior art by researchers such as Himeda and Sadler, standard protocols typically involve refluxing reactants in solvents like ethanol or dichloromethane for extended durations. For instance, comparative data indicates that attempting to synthesize phenanthroimidazole derivatives or their ruthenium complexes via oil-bath heating at 100°C often requires 3 to 8 hours to reach completion. Even after such extensive thermal exposure, the resulting yields are frequently suboptimal, hovering between 45% and 58% for certain derivatives. Furthermore, some specific arene ruthenium(II) precursors simply fail to form under these traditional conditions, leading to dead ends in synthetic routes. This not only wastes valuable raw materials but also creates significant bottlenecks in supply chains where time-to-market is critical. The energy consumption associated with maintaining high temperatures for hours also contradicts modern principles of green chemistry, adding unnecessary operational costs and environmental burden to the manufacturing process.
The Novel Approach
The microwave-assisted methodology described in the patent offers a decisive solution to these longstanding challenges by utilizing direct molecular excitation. Instead of relying on the slow transfer of heat from a vessel wall to the reaction mixture, microwave irradiation energizes the molecules directly, leading to rapid and uniform heating throughout the bulk solution. This results in a dramatic acceleration of reaction kinetics, allowing the synthesis of target arene ruthenium(II) compounds to be completed in just 0.5 to 30 minutes at moderate temperatures of 50°C to 70°C. The impact on yield is equally profound; experimental data shows that yields consistently surpass 90%, effectively doubling the output compared to conventional methods. Moreover, this technique enables the successful preparation of precursor compounds that were previously inaccessible, expanding the chemical space available for drug discovery. By drastically shortening the reaction cycle, this approach facilitates rapid iteration in R&D and supports scalable manufacturing processes that are both economically and environmentally superior.
Mechanistic Insights into Microwave-Accelerated Coordination Chemistry
The efficacy of this synthesis relies on the unique interaction between microwave energy and the polar transition states involved in ruthenium coordination. In the formation of the arene ruthenium(II) precursor, ruthenium trichloride reacts with cyclohexadiene derivatives to form a dimeric chloro-bridged structure. 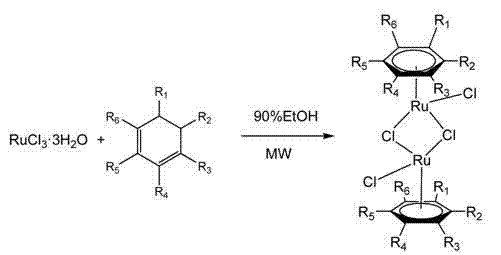 . Under microwave irradiation, the dipolar polarization of the solvent and reactants enhances the rate of ligand substitution and oxidative addition steps. The rapid heating prevents the decomposition of sensitive intermediates that might occur during prolonged conventional heating. Subsequently, when this precursor reacts with the phenanthroimidazole ligand, the microwave field facilitates the displacement of chloride bridges by the bidentate nitrogen donor. This ligand exchange is crucial for forming the stable cationic ruthenium complex. The precise control over temperature (50-70°C) ensures that the delicate balance between reaction rate and product stability is maintained, minimizing side reactions such as ligand degradation or metal precipitation. This mechanistic understanding allows chemists to fine-tune reaction parameters for different substituents on the arene or imidazole ring, ensuring consistent quality across diverse compound libraries.
. Under microwave irradiation, the dipolar polarization of the solvent and reactants enhances the rate of ligand substitution and oxidative addition steps. The rapid heating prevents the decomposition of sensitive intermediates that might occur during prolonged conventional heating. Subsequently, when this precursor reacts with the phenanthroimidazole ligand, the microwave field facilitates the displacement of chloride bridges by the bidentate nitrogen donor. This ligand exchange is crucial for forming the stable cationic ruthenium complex. The precise control over temperature (50-70°C) ensures that the delicate balance between reaction rate and product stability is maintained, minimizing side reactions such as ligand degradation or metal precipitation. This mechanistic understanding allows chemists to fine-tune reaction parameters for different substituents on the arene or imidazole ring, ensuring consistent quality across diverse compound libraries.
Impurity control is another critical aspect where this method excels, particularly for pharmaceutical applications requiring stringent purity specifications. Conventional long-duration heating often promotes the formation of thermal byproducts and oligomeric species due to localized overheating or extended exposure to reactive conditions. In contrast, the rapid and uniform heating profile of microwave synthesis limits the time window available for secondary reactions to occur. The patent data demonstrates that products obtained via this method exhibit clean spectral profiles, with ESI-MS and NMR data confirming the expected structures with minimal impurities. For example, the synthesis of [(η6-C6H6)Ru(PIP)Cl]Cl yielded a product with a distinct mass spectrum peak at m/z 511.1, corresponding to the [M-Cl]+ ion, with high fidelity. This inherent purity reduces the burden on downstream purification steps, such as chromatography or repeated recrystallization, thereby streamlining the overall production workflow. For quality assurance teams, this translates to more reliable batch-to-batch consistency and a lower risk of failing regulatory compliance tests due to unknown impurities.
How to Synthesize Arene Ruthenium(II) Compounds Efficiently
The implementation of this microwave-assisted protocol requires careful attention to reaction conditions to maximize the benefits of rapid heating. The process begins with the preparation of the necessary precursors, followed by the coordination step in a specialized microwave reactor. Detailed procedural nuances, such as the specific molar ratios of ruthenium precursor to ligand (typically 1:1.2 to 1:5) and the choice of solvent (dichloromethane), are critical for success.
- Prepare the reaction mixture by combining the arene ruthenium(II) precursor, phenanthroimidazole derivative, and dichloromethane in a microwave reaction tube under nitrogen atmosphere.
- Subject the mixture to microwave irradiation at a controlled temperature between 50°C and 70°C for a duration of 0.5 to 30 minutes to facilitate rapid ligand exchange.
- Upon completion, remove the solvent via rotary evaporation, dissolve the residue in water, filter, and recrystallize the final product from methanol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this microwave synthesis technology presents compelling economic and operational advantages. The most immediate benefit is the drastic reduction in processing time, which directly translates to increased throughput and asset utilization. By reducing reaction times from hours to minutes, manufacturing facilities can run significantly more batches within the same operational window, effectively expanding capacity without the need for capital investment in new reactors. This agility is crucial for responding to fluctuating market demands and ensuring the continuous supply of critical intermediates. Furthermore, the substantial improvement in yield means that less raw material is required to produce the same amount of final product, leading to significant cost savings on expensive ruthenium salts and specialized ligands.
- Cost Reduction in Manufacturing: The elimination of prolonged heating cycles results in a marked decrease in energy consumption, aligning with sustainability goals and reducing utility costs. Additionally, the higher yields minimize waste generation, lowering the costs associated with waste disposal and solvent recovery. The ability to synthesize previously inaccessible precursors also removes the need for costly alternative synthetic routes or external sourcing of hard-to-find materials.
- Enhanced Supply Chain Reliability: Shorter lead times for synthesis allow for a more responsive supply chain, reducing the need for large safety stocks of finished goods. The robustness of the microwave method ensures consistent production schedules, mitigating the risk of delays caused by failed batches or extended reaction times common in traditional methods. This reliability is essential for maintaining uninterrupted production lines in downstream pharmaceutical manufacturing.
- Scalability and Environmental Compliance: While microwave chemistry was once limited to small scales, modern industrial microwave reactors now allow for kilogram-scale production, making this technology viable for commercial manufacturing. The reduced solvent usage and energy footprint contribute to a greener manufacturing profile, helping companies meet increasingly strict environmental regulations and corporate social responsibility targets.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis method is vital for stakeholders evaluating its potential integration into their operations. The following questions address common inquiries regarding the feasibility, scope, and benefits of this microwave-assisted approach.
Q: What are the primary advantages of using microwave synthesis for arene ruthenium(II) compounds compared to conventional heating?
A: Microwave synthesis significantly reduces reaction time from several hours or days to merely minutes while simultaneously increasing reaction yields from approximately 50% to over 90%, as demonstrated in comparative studies within the patent data.
Q: Can this microwave method be applied to synthesize precursors that are difficult to obtain via traditional methods?
A: Yes, the patent highlights that certain arene ruthenium(II) precursors, which fail to form or yield poorly under conventional reflux conditions, can be successfully synthesized with high efficiency using this microwave-assisted protocol.
Q: What specific ligands are compatible with this ruthenium coordination chemistry?
A: The method is specifically optimized for phenanthroimidazole derivatives, including various substituted forms such as 2-(phenyl)imidazo[4,5-f][1,10]phenanthroline and its methoxy-substituted analogs, ensuring versatile application in drug design.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arene Ruthenium(II) Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic technologies like microwave-assisted coordination chemistry in delivering high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the lab can be successfully translated to industrial manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of arene ruthenium(II) compound meets the exacting standards required for drug development. Our infrastructure is designed to handle complex organometallic synthesis with precision, safety, and reproducibility.
We invite global partners to collaborate with us to leverage these technological advancements for their specific project needs. Whether you require custom synthesis of novel ruthenium complexes or scale-up of existing protocols, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with high-performance organometallic solutions.
